Abstract
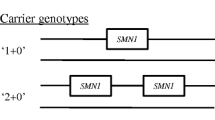
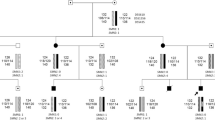
Spinal muscular atrophy (SMA) is an autosomal recessive, neurodegenerative disorder characterised commonly by proximal muscle weakness and wasting in the absence of sensory signs. Deletion or disruption of the SMN1 gene causes the disease. The SMN1 gene is located within an inverted duplication on chromosome 5q13 with the genes SMN2, NAIP and GTF2H2. MLPA analysis of 13 Cypriot SMA patients revealed that, 12 patients carried a homozygous SMN1 gene deletion and one patient carried two copies of the SMN1 gene. Two of 13 cases were a consequence of a paternally originating de novo mutation. Five genotypes were identified within the population, with the most frequent being a homozygous SMN1 and NAIP genes deletion. In conclusion, genotype–phenotype correlation revealed that SMN2 is inversely related to disease severity and that NAIP and GTF2H2 act as negative modifiers. This study provided, for the first time, a comprehensive overview of gene copy numbers and inheritance patterns within Cypriot SMA families.



Similar content being viewed by others
References
Ogino S, Wilson RB, Gold B (2004) New insights on the evolution of the SMN1 and SMN2 region: simulation and meta-analysis for allele and haplotype frequency calculations. Eur J Hum Genet 12(12):1015–1023. doi:10.1038/sj.ejhg.5201288
Markowitz JA, Singh P, Darras BT (2012) Spinal muscular atrophy: a clinical and research update. Pediatr Neurol 46(1):1–12. doi:10.1016/j.pediatrneurol.2011.09.001
D’Amico A, Mercuri E, Tiziano FD, Bertini E (2011) Spinal muscular atrophy. Orphanet J Rare Dis 6:71. doi:10.1186/1750-1172-6-71
Prior TW, Krainer AR, Hua Y, Swoboda KJ, Snyder PC, Bridgeman SJ, Burghes AH, Kissel JT (2009) A positive modifier of spinal muscular atrophy in the SMN2 gene. Am J Hum Genet 85(3):408–413. doi:10.1016/j.ajhg.2009.08.002
Clermont O, Burlet P, Burglen L, Lefebvre S, Pascal F, McPherson J, Wasmuth JJ, Cohen D, Le Paslier D, Weissenbach J et al (1994) Use of genetic and physical mapping to locate the spinal muscular atrophy locus between two new highly polymorphic DNA markers. Am J Hum Genet 54(4):687–694
Lefebvre S, Burglen L, Reboullet S, Clermont O, Burlet P, Viollet L, Benichou B, Cruaud C, Millasseau P, Zeviani M et al (1995) Identification and characterization of a spinal muscular atrophy-determining gene. Cell 80(1):155–165
Monani UR, Lorson CL, Parsons DW, Prior TW, Androphy EJ, Burghes AH, McPherson JD (1999) A single nucleotide difference that alters splicing patterns distinguishes the SMA gene SMN1 from the copy gene SMN2. Hum Mol Genet 8(7):1177–1183
Lorson CL, Hahnen E, Androphy EJ, Wirth B (1999) A single nucleotide in the SMN gene regulates splicing and is responsible for spinal muscular atrophy. Proc Natl Acad Sci USA 96(11):6307–6311
Feldkotter M, Schwarzer V, Wirth R, Wienker TF, Wirth B (2002) Quantitative analyses of SMN1 and SMN2 based on real-time lightCycler PCR: fast and highly reliable carrier testing and prediction of severity of spinal muscular atrophy. Am J Hum Genet 70(2):358–368. doi:10.1086/338627
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16(3):1215
Cusco I, Lopez E, Soler-Botija C, Jesus Barcelo M, Baiget M, Tizzano EF (2003) A genetic and phenotypic analysis in Spanish spinal muscular atrophy patients with c.399_402del AGAG, the most frequently found subtle mutation in the SMN1 gene. Hum Mutat 22(2):136–143. doi:10.1002/humu.10245
Kirwin SM, Vinette KM, Gonzalez IL, Abdulwahed HA, Al-Sannaa N, Funanage VL (2013) A homozygous double mutation in SMN1: a complicated genetic diagnosis of SMA. Mol Genet Genomic Med 1(2):113–117. doi:10.1002/mgg3.10
Wirth B, Brichta L, Hahnen E (2006) Spinal muscular atrophy: from gene to therapy. Semin Pediatr Neurol 13(2):121–131. doi:10.1016/j.spen.2006.06.008
Wirth B, Schmidt T, Hahnen E, Rudnik-Schoneborn S, Krawczak M, Muller-Myhsok B, Schonling J, Zerres K (1997) De novo rearrangements found in 2% of index patients with spinal muscular atrophy: mutational mechanisms, parental origin, mutation rate, and implications for genetic counseling. Am J Hum Genet 61(5):1102–1111. doi:10.1086/301608
Arkblad EL, Darin N, Berg K, Kimber E, Brandberg G, Lindberg C, Holmberg E, Tulinius M, Nordling M (2006) Multiplex ligation-dependent probe amplification improves diagnostics in spinal muscular atrophy. Neuromuscul Disord 16(12):830–838. doi:10.1016/j.nmd.2006.08.011
Rodrigues NR, Owen N, Talbot K, Patel S, Muntoni F, Ignatius J, Dubowitz V, Davies KE (1996) Gene deletions in spinal muscular atrophy. J Med Genet 33(2):93–96
Amara A, Adala L, Ben Charfeddine I, Mamai O, Mili A, Lazreg TB, H’Mida D, Amri F, Salem N, Boughammura L, Saad A, Gribaa M (2012) Correlation of SMN2, NAIP, p44, H4F5 and Occludin genes copy number with spinal muscular atrophy phenotype in Tunisian patients. Eur J Paediatr Neurol 16(2):167–174. doi:10.1016/j.ejpn.2011.07.007
Burlet P, Burglen L, Clermont O, Lefebvre S, Viollet L, Munnich A, Melki J (1996) Large scale deletions of the 5q13 region are specific to Werdnig–Hoffmann disease. J Med Genet 33(4):281–283
Derakhshandeh-Peykar P, Esmaili M, Ousati-Ashtiani Z, Rahmani M, Babrzadeh F, Farshidi S, Attaran E, Sajedifar MM, Farhud DD (2007) Molecular analysis of the SMN1 and NAIP genes in Iranian patients with spinal muscular atrophy. Ann Acad Med Singapore 36(11):937–941
Roy N, Mahadevan MS, McLean M, Shutler G, Yaraghi Z, Farahani R, Baird S, Besner-Johnston A, Lefebvre C, Kang X et al (1995) The gene for neuronal apoptosis inhibitory protein is partially deleted in individuals with spinal muscular atrophy. Cell 80(1):167–178
Savas S, Gokgoz N, Kayserili H, Ozkinay F, Yuksel-Apak M, Kirdar B (2000) Screening of deletions in SMN, NAIP and BTF2p44 genes in Turkish spinal muscular atrophy patients. Hum Hered 50(3):162–165. doi:10.1159/000022907
Watihayati MS, Zabidi AM, Tang TH, Nishio H, Zilfalil BA (2007) NAIP-deletion analysis in Malaysian patients with spinal muscular atrophy. Kobe J Med Sci 53(4):171–175
Jordanova A, Stoyanova V, Uzunova M, Litvinenko I, Kremensky I (1998) Deletion analysis of Bulgarian SMA families. Hum Mutat 12(1):33–38. doi:10.1002/(SICI)1098-1004(1998)12:1<33:AID-HUMU5>3.0.CO;2-Y
Miskovic M, Lalic T, Radivojevic D, Cirkovic S, Vlahovic G, Zamurovic D, Guc-Scekic M (2011) Lower incidence of deletions in the survival of motor neuron gene and the neuronal apoptosis inhibitory protein gene in children with spinal muscular atrophy from Serbia. Tohoku J Exp Med 225(3):153–159
Sertic J, Barisic N, Sostarko M, Bosnjak N, Culic V, Cvitanovic L, Ferencak G, Brzovic Z, Stavljenic-Rukavina A (1997) Deletions in the SMN and NAIP genes in patients with spinal muscular atrophy in Croatia. Coll Antropol 21(2):487–492
Burglen L, Seroz T, Miniou P, Lefebvre S, Burlet P, Munnich A, Pequignot EV, Egly JM, Melki J (1997) The gene encoding p44, a subunit of the transcription factor TFIIH, is involved in large-scale deletions associated with Werdnig–Hoffmann disease. Am J Hum Genet 60(1):72–79
He J, Zhang QJ, Lin QF, Chen YF, Lin XZ, Lin MT, Murong SX, Wang N, Chen WJ (2013) Molecular analysis of SMN1, SMN2, NAIP, GTF2H2, and H4F5 genes in 157 Chinese patients with spinal muscular atrophy. Gene 518(2):325–329. doi:10.1016/j.gene.2012.12.109
Chen WJ, He J, Zhang QJ, Lin QF, Chen YF, Lin XZ, Lin MT, Murong SX, Wang N (2012) Modification of phenotype by SMN2 copy numbers in two Chinese families with SMN1 deletion in two continuous generations. Clin Chim Acta 413(23–24):1855–1860. doi:10.1016/j.cca.2012.07.020
Jedrzejowska M, Milewski M, Zimowski J, Borkowska J, Kostera-Pruszczyk A, Sielska D, Jurek M, Hausmanowa-Petrusewicz I (2009) Phenotype modifiers of spinal muscular atrophy: the number of SMN2 gene copies, deletion in the NAIP gene and probably gender influence the course of the disease. Acta Biochim Pol 56(1):103–108
Vezain M, Saugier-Veber P, Goina E, Touraine R, Manel V, Toutain A, Fehrenbach S, Frebourg T, Pagani F, Tosi M, Martins A (2010) A rare SMN2 variant in a previously unrecognized composite splicing regulatory element induces exon 7 inclusion and reduces the clinical severity of spinal muscular atrophy. Hum Mutat 31(1):E1110–E1125. doi:10.1002/humu.21173
Hauke J, Riessland M, Lunke S, Eyupoglu IY, Blumcke I, El-Osta A, Wirth B, Hahnen E (2009) Survival motor neuron gene 2 silencing by DNA methylation correlates with spinal muscular atrophy disease severity and can be bypassed by histone deacetylase inhibition. Hum Mol Genet 18(2):304–317. doi:10.1093/hmg/ddn357
Acknowledgments
We would like to thank the patients and their families for their contribution to this study. This manuscript is dedicated to the memory of Dr. Goula Stylianidou.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Theodorou, L., Nicolaou, P., Koutsou, P. et al. Genetic findings of Cypriot spinal muscular atrophy patients. Neurol Sci 36, 1829–1834 (2015). https://doi.org/10.1007/s10072-015-2263-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10072-015-2263-5