Abstract
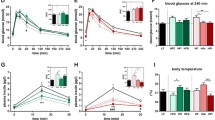
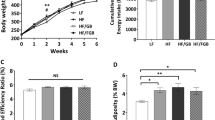
The aim of the present study was to investigate the effects of fermented barley supplementation on blood lipid profiles, carnitine concentrations, and hepatic mRNA levels of enzymes involved in fatty acid metabolism. Thirty-two C57BL/6J male mice were divided into 4 groups; normal diet control group (ND), high fat diet control group (HD), high fat diet plus barley supplemented group (BR), and high fat diet plus fermented barley supplemented group (BR-F). BR-F supplementations decreased total cholesterol (TC) and LDL-cholesterol in serum, triglyceride (TG) in liver. Serum total carnitine (TCNE) concentrations were significantly higher in the BR-F group than HD group. BR-F supplementations significantly increased hepatic lipolysis regulating gene expression such as carnitine palmitoyltransferase-I (CPT-I), peroxisome proliferator-activated receptor-α (PPARα), whereas significantly decreased lipogenic enzyme expression such as acetyl CoA carboxylase (ACC). These results suggest that the fermented barley has anti-obesity properties.
Similar content being viewed by others
References
Brown A, Siahpush M. Risk factors for overweight and obesity: Results from the 2001 National Health Survey. J. Public. Health 121: 603–613 (2007)
Brisbon N, Plumb J, Brawer R, Paxman D. The asthma and obesity epidemics: The role played by the built environment — a public health perspective. J. Allergy Clin. Immun. 115: 1024–1028 (2005)
Jee SH, Kim HJ. Obesity, insulin resistance, and cancer risk. Yonsei Med. J. 46: 449–455 (2005)
Kopelman PG. Obesity as a medical problem. Nature 404 (6778): 635–643 (2000)
Newman CW, Newman RK. A brief history of barley foods. Cereal Foods World. 51: 4–7 (2006)
Madhujith T, Izydorczyk MS, Shahidi F. Antioxidant properties of pearled barley fractions. J. Agr. Food. Chem. 54: 3283–3289 (2006)
Slavin J, Marquart L, Jacobs D. Consumption of whole-grain foods and decreased risk of cancer: Proposed mechanisms. Cereal Foods World. 45: 54–58 (2000)
Martinez VM, Newman RK, Newman CW. Barley diets with different fat sources have hypocholesterolemic effects in chicks. J. Nutr. 122: 1070–1076 (1992)
Kim SY, Song HJ, Lee YY, Cho KH, Roh YK. Biomedical issues of dietary fiber β-glucan. J. Korean Med. Sci. 21: 781–789 (2006)
Wood BPJ, Braaten JT, Scott FW. Effect of dose and modification of viscous properties of oat gum on plasma glucose and insulin following an oral glucose load. Brit. J. Nutr. 72: 731–743 (1994)
Davidson MH, Dugan LD, Burns JH, Bova J, Story K, Drennan KB. The hypocholesterolemic effects of β-glucan in oatmeal and oat bran. A dose controlled study. J. Am. Med. Assoc. 265: 1833–1839 (1991)
Behall KM, Scholfield DJ, Hallfrisch J. Lipids significantly reduced by diets containing barley in moderately hypercholesterolemic men. J. Am. Coll. Nutr. 23: 55–62 (2004)
Friedewald WT, Levy RI, Fredrickson DS. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 18: 499–502 (1972)
Cederblad G, Lindstedt S. A method for the determination of carnitine in the picomole range. Clin. Chim. Acta 37: 235–243 (1972)
Sachan DS, Rhew TH, Ruark RA. Ameliorating effects of carnitine and its precursors on alcohol-induced fatty liver. J. Clin. Nutr. 39: 738–744 (1984)
Lissner L, Lindroos AK, Sjostrom L. Swedish obese subjects (SOS): An obesity intervention study with a nutritional perspective. Eur. J. Clin. Nutr. 52: 316–322 (1998)
Kimm SY. The role of dietary fiber in the development and treatment of childhood obesity. Pediatrics 96: 1010–1014 (1995)
Birketvedt GS, Aaseth J, Florholmen JR, Ryttig K. Long-term effect of fiber supplement and reduced energy intake on body weight and blood lipids in overweight subjects. Acta Med. 43: 129–132 (2000)
Hays NP, Starling RD, Liu X, Sullivan DH, Trappe TA, Fluckey JD, Evans WJ. Effects of an ad libitum low-fat, high-carbohydrate diet on body weight, body composition, and fat distribution in older men and women: A randomized controlled trial. Arch. Intern. Med. 164: 210–217 (2004)
Cave NA, Wood PJ, Burrows VD. Estimation of an acceptable β-glucan level for broiler chick diets. Can. J. Anim. Sci. 72: 691–694 (1992)
Jo J, Gavrilova O, Pack S, Jou W, Mullen S, Sumner AE, Cushman SW, Periwal V. Hypertrophy and/or hyperplasia: Dynamics of adipose tissue growth. PLoS Comput Biol. 5(3): e1000324 (2009)
Petit V, Arnould L, Martin P, Monnot MC, Pineau T, Besnard P, Niot I. Chronic high-fat diet affects intestinal fat absorption and postprandial triglyceride levels in the mouse. J. Lipid Res. 48: 278–287 (2007)
Braaten JT, Wood PJ, Scott FW, Wolynetz MS, Lowe MK, Bradley-White P, Collins MW. Oat β-glucan reduces blood cholesterol concentration in hypercholesterolemic subjects. Eur. J. Clin. Nutr. 48: 465–474 (1994)
Li J, Wang J, Kaneko T, Qin LQ, Sato A. Effect of fiber intake on the blood pressure, lipids, and heart rate in Goto Kakizaki rats. Nutrition 20: 1003–1007 (2004)
Lovegrove JA, Clohessy A, Milon H, Williams CM. Modest doses of β-glucan do not reduce concentrations of potentially atherogenic lipoproteins. Am. J. Clin. Nutr. 72: 49–55 (2000)
McGarry JD, Brown NF. The mitochondrial carnitine palmitoyltransferase system. From concept to molecular analysis. Eur. J. Biochem. 244: 1–14 (1997)
Ramsay RR, Gandou RD, van der Leij FR. Molecular enzymology of carnitine transfer and transport. Biochim. Biophys. Acta 1546: 21–43 (2001)
Jakobs BS, Wanders RJ. Fatty acid β-oxidation in peroxisomes and mitochondria: The ®rst, unequivocal evidence for the involvement of carnitine in shuttling propionyl-CoA from peroxisomes to mitochondria. Biochem. Bioph. Res. Co. 213: 1035–1041 (1995)
Verhoeven NM, Roe DS, Kok RM, Wanders RJ, Jakobs C, Roe CR. Phytanic acid and pristanic acid are oxidized by sequential peroxisomal and mitochondrial reactions in cultured ®broblasts. J. Lipid. Res. 39: 66–74 (1998)
Memon RA, Fulle J, Moser AH, Smith PJ, Grunfeld C, Feingold KR. Regulation of putative fatty acid transporters and acyl-CoA synthetase in liver and adipose tissue in ob/ob mice. Diabetes 48: 121–127 (1999)
McGarry JD, Leatherman GF, Foster DW. Carnitine palmitoyltransferase I. The site of inhibition of hepatic fatty acid oxidation by malonyl-CoA. J. Biol. Chem. 253: 4128–4136 (1978)
Bonnefont JP, Demaugre F, Prip-Buus C, Saudubray JM, Brivet M, Abadi N, Thuillier L. Carnitine palmitoyltransferase deficiencies. Mol. Genet. Metab. 68: 424–440 (1999)
Achouri Y, Hegarty BD, Allanic D, Becard D, Hainault I, Ferre P, Foufelle F. Long chain fatty acyl-CoA synthetase expression is induced by insulin and glucose: Involvement of sterol regulatory element-binding protein-1c. Biochimie 87: 1149–1155 (2005)
Cha YS, Soh JR, Kin JW. Acanthopanax senticosus extract from cultured cells improves lipid parameters in rats fed with a high fat diet. Nutraceut. Food 8: 40–45 (2003)
Patsouris D, Reddy JK, Muller M, Kersten S. Peroxizome proliferator-activated receptor alpha mediates the effects of high-fat diet on hepatic gene expression. Endocrinology 147: 1508–1516 (2007)
Crespillo A, Alonso M, Vida M, Pavón FJ, Serrano A, Rivera P, Romero-Zerbo Y, Fernández-Llebrez P, Martínez A, Pérez-Valero V, Bermúdez-Silva FJ, Suárez J, de Fonseca FR. Reduction of body weight, liver steatosis, and SCD1 expression by pharmacologic administration of the isoflavone daidzein in diet-induced obesity. Brit. J. Pharmacol. 10: 1476-5381331–337
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Choi, JH., Kim, TH., Ko, MS. et al. Effects of fermented barley on lipid and carnitine profiles in C57BL/6J mice. Food Sci Biotechnol 21, 323–329 (2012). https://doi.org/10.1007/s10068-012-0043-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10068-012-0043-z