Abstract
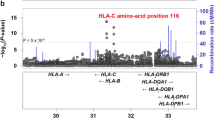
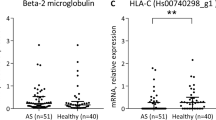
The aim of this study is to investigate the association of HLA-A, B and HLA-DR gene expression and to assess an association of additional HLA antigens besides HLA-B27 in south Tunisian patients with spondyloarthritis (SpA). Eighty-five patients diagnosed with ankylosing spondylitis (AS, n = 68) and reactive arthrithis (ReA, n = 17) were selected and compared with 100 healthy controls (HC). HLA class I antigens were typed serologically using microlymphocytotoxicity technique. HLA-DRB1* alleles were studied by polymerase chain reaction amplification with sequence-specific primers. The significance of differences between patients and controls was tested by chi-square analysis. We found significantly increased frequencies of HLA-A3 (30.6%; pC = 0.04; OR = 2.95), HLA-B27 (62.35%; pC = 4.10−17, OR = 53.55), and HLA-DRB1*15 (17.2%; pC = 0.026; RR = 2.58) alleles in SpA patients compared to HC. The most frequent and strongest association was observed for HLA-B27 in AS (pC = 6.6 × 10−16, OR = 52.23). When AS and ReA patients were analysed separately, HLA-DRB1*15 and HLA-A3 were increased only in AS (pC = 0.01, OR = 2.99 and pC = 0.03, OR = 3.14, respectively). In ReA patients, HLA-DRB1*04 (p = 0.033, pC = NS, OR = 2.89) was found to be the most common allele. By analysing the HLA-B27-negative subgroup, HLA-A3 and HLA-DRB1*15 expression was found to be dependent on the presence of HLA-B27. HLA-B27 expression was higher in male (45/53; 85%) as compared to female (8/53; 15%) patients (p = 0.03). Apart from HLA-B27, HLA-A3 and HLA-DRB1*15 are the MHC class I and II alleles found most frequent in Tunisian patients with AS, whereas HLA-DRB1*04 was found most frequent in ReA patients. HLA-B27 is more frequent in male than in female patients.
Similar content being viewed by others
References
Lopez de Castro JA (2006) HLA-B27 and the pathogenesis of spondyloarthropathies. Immunol Lett 15:27–33
Reveille JD, Arnett FC (2005) Spondyloarthritis: update on pathogenesis and management. Am J Med 118:592–603
Hannu T, Inman R, Granfors K, Leirisalo-Repo M (2006) Reactive arthritis or post-infectious arthritis? Best Pract Res Clin Rheumatol 20:419–433
Toussirot E, Wendling D (2006) Immunogenetic of ankylosing spondylitis. Rev Méd Interne 27:762–771
Khan MA (1997) Prevalence of HLA B27 in world populations. In: Lopez-Larrea C (ed) HLA B27 in the development of spondyloarthropathies. Landes Springer, Austin, p 1e16
Gonzalez-Roces S, Alvarez MW, Gonzalez S et al (1997) HLA B27 polymorphism and worldwide susceptibility to ankylosing spondylitis. Tissue Antigens 49:116–123
Tuokko J, Reijonen H, Ilonen J et al (1997) Increase of HLA-DRB1*0408 and –DQB1*0301 in HLA-B27 positive reactive arthritis. Ann Rheum Dis 56:37–40
Breban WTN, Miceli-Richard C, Zineviova E (2006) The genetics of spondylarthropathies. Joint Bone Spine 73:355–362
Brown MA, Kennedy GL, Darke C et al (1998) The effect of HLA-DR genes on susceptibility to and severity of ankylosing spondylitis. Arthritis Rheum 41:460–465
Vargas-Alarcón, Londoño JD, Hernández-Pacheco G, Pacheco-Tena C, Castillo E, Cardiel MH, Granados J, Burgos-Vargas R (2002) Effect of HLA-B and HLA-DR genes on susceptibility to and severity of spondyloarthropathies in Mexican patients. Ann Rheum Dis 61:714–717
Fiorillo MT, Cauli A, Carcassi C, Bitti PP, Vacca A, Passiu G, Bettosini F, Mathieu A, Sorrentino R (2003) Two distinctive hla haplotypes harbor the b27 alleles negatively or positively associated with ankylosing spondylitis in Sardinia. Arthritis Rheum 48:1385–1389
Hajjej-Hajjej A, Kâabi H, Sellami MH et al (2006) The contribution of HLA class I and II alleles and haplotypes to the investigation of the evolutionary history of Tunisians. Tissue Antigens 68:153–162
Ayed K, Ayed-Jendoubi S, Sfar I, Labonne MP, Gebuhrer L (2004) HLA class-I and HLA class-II phenotypic, gene and haplotypic frequencies in Tunisians by using molecular typing data. Tissue Antigens 64:520–532
Kchir MM, Hamdi W, Laadhar L et al (2010) HLA-B, DR and DQ antigens polymorphism in Tunisian patients with ankylosing spondylitis (a case–control study). Rheumatol Int 30:933–939
Van der Linden S, Valkenburg HA, Cats A (1984) Evaluation of diagnostic criteria for ankylosing spondylitis. A proposal for modification of the New York criteria. Arthritis Rheum 27:361–368
Dougados M, van der Linden S, Juhlin R et al (1991) The European Spondylarthropathy Study Group preliminary criteria for the classification of spondylarthropathy. Arthritis Rheum 34:1218–1227
Sieper J, Rudwaleit M, Braun J, van der Heijde D (2002) Diagnosing reactive arthritis: role of clinical setting in the value of serologic and microbiologic assays. Arthritis Rheum 46:319–327
Terasaki PI, Mcclemmand JD (1964) Microdropletassay of human serum cytotoxins. Nature 204:998–1000
Degos L (1985) La répartition anthropologique des gènes HLA et dynamique des populations. In: Dausset J, Pla M (eds) HLA, Complexe majeur d'histocompatibilité de l'homme. Médecine-Sciences, Flammarion, pp 101–119
Colombani J. (1993) Évolution du CPMH, des gènes et des allèles. La superfamille des immunoglobulines. Génétique des populations. In: HLA, fonctions immunitaires et applications médicales. J. Libbey: Eurotext Médecine sciences. p. 91–110
Jaakkola E, Herzberg I, Laiho K, Barnardo MCN M, Pointon JJ, Kauppi M, Kaarela K, Tuomilehto-Wolf E, Tuomilehto J, Wordsworth BP, Brown MA (2006) Finnish HLA studies confirm the increased risk conferred by HLA-B27 homozygosity in ankylosing spondylitis. Ann Rheum Dis 65:775–780
Tamouza R, Awada H, Marzais F, Teisserenc H, Poirier JC, Toubert A, Raffoux C, Charron D (1996) HLA B27 in the Lebanese population: subtype analysis and association with ankylosing spondylitis (SA). Hum Immunol 47:74–76
Younsi R, Azrib S, Aitouazar M et al. (2007) Evaluation de l'incidence du gene HLAB-27 chez le patient atteint de spondylarthrite ankylosante au Maroc. 20 éme congrés de la sociéte francaise de rhumatologie (SFR)
Vargas-Alarcon G, Garcia A, Bahena S, Melin-Aldana H, Andrade F, Ibafiez-de-Kasep G, Alcocer-Varela J, Alarcon-Segovia D, Granados J (1994) HLA-B alleles and complotypes in Mexican patients with seronegative spondyloarthropathies. Ann Rheum Dis 53:755–758
Rudwaleit M, Haibel H, Baraliakos X, Listing J, Märker-Hermann E, Zeidler H, Braun J, Sieper J (2009) The early disease stage in axial spondylarthritis: results from the German spondyloarthritis inception cohort. Arthritis Rheum 60:717–727
Acknowledgements
We thank Mouna Sellami (EPS Hédi chaker, Sfax) for helping in the conception, the design and coordination of the study. We thank Ilhem Cheour (Tunis), Nihel Meddeb (Tunis), Mohamed Moalla (Tunis) and Imed kolsi (Sfax) for providing patients. This project was supported by grants from the Ministry of Research and Development of Tunisia with participation of funds from the Ministry of Health of Tunisia.
Disclosures
None
Author information
Authors and Affiliations
Corresponding author
Additional information
The first two authors (Dr. Nadia Mahfoudh and Dr. Mariam Siala) are sharing their first authorship; NM and MS have equally contributed to this work.
Rights and permissions
About this article
Cite this article
Mahfoudh, N., Siala, M., Rihl, M. et al. Association and frequency of HLA-A, B and HLA-DR genes in south Tunisian patients with spondyloarthritis (SpA). Clin Rheumatol 30, 1069–1073 (2011). https://doi.org/10.1007/s10067-011-1705-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10067-011-1705-6