Abstract
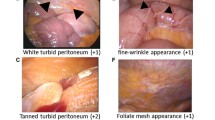
Peritoneal dialysis solution (PDS) plays a role in functional and morphological damage to the peritoneum. This study aimed to clarify the effect of neutral PDS in preventing morphological changes by assessing peritoneal damage and comparing morphological alterations between PD patients treated with neutral PDS and acidic PDS. Sixty-one patients participated from seven hospitals. All patients were treated with neutral PDS excluding icodextrin, during their entire PD treatment, and experienced no episode of peritonitis. The thickness of submesothelial compact (SMC) zone and the presence of vasculopathy in the anterior parietal abdominal peritoneum were assessed. The impact of icodextrin, hybrid therapy, and peritoneal rest and lavage in morphological alterations were determined. There was no significant difference in the average SMC thickness between neutral and acidic PDS. The vessel patency in patients using neutral PDS was significantly higher compared to that in acidic PDS at any time during PD. There were no significant suppressive effects from interventions or use of icodextrin with respect to peritoneal morphological injury. A monolayer of mesothelial cell was observed in approximately half the patients, especially in their receiving lavage patients. Neutral PDS, accompanied by other preventive approaches against peritoneal injury, might suppress the development of peritoneal morphological alterations.



Similar content being viewed by others
References
Liberek T, Topley N, Jörres A, Petersen MM, Coles GA, Gahl GM, Williams JD. Peritoneal dialysis fluid inhibition of polymorphonuclear leukocyte respiratory burst activation is related to the lowering of intracellular pH. Nephron. 1993;65:260–5.
Williams JD, Craig KJ, Topley N, Von Ruhland C, Fallon M, Newman GR, Williams RK, Mackenzie GT, Peritoneal Biopsy Study Group. Morphologic changes in the peritoneal membrane of patients with renal disease. J Am Soc Nephrol. 2002;13:470–9.
Linden T, Forsbäck G, Deppisch R, Henle T, Wieslander A. 3-Deoxyglucosone, a promoter of advanced glycation end products in fluids for peritoneal dialysis. Perit Dial Int. 1998;18:290–3.
Jörres A, Topley N, Witowski J, Liberek T, Gahl GM. Impact of peritoneal dialysis solutions on peritoneal immune defense. Perit Dial Int. 1993;13:S291–4.
Douvdevani A, Rapoport J, Konforty A, Yulzari R, Moran A, Chaimovitz C. Intracellular acidification mediates the inhibitory effect of peritoneal dialysate on peritoneal macrophages. J Am Soc Nephrol. 1995;6:207–13.
Topley N, Coles GA, Williams JD. Biocompatibility studies on peritoneal cells. Perit Dial Int. 1994;14:S21–8.
Honda K, Hamada C, Nakayama M, Miyazaki M, Sherif AM, Harada T, Hirano H, Peritoneal Biopsy Study Group of the Japanese Society for Peritoneal Dialysis. Impact of uremia, diabetes, and peritoneal dialysis itself on the pathogenesis of peritoneal sclerosis: a quantitative study of peritoneal membrane morphology. Clin J Am Soc Nephrol. 2008;3:720–8.
Krishnan M, Tam P, Wu G, Breborowicz A, Oreopoulos DG. Glucose degradation products (GDP’s) and peritoneal changes in patients on chronic peritoneal dialysis: will new dialysis solutions prevent these changes? Int Urol Nephrol. 2005;37:409–18.
Grzegorzewska AE. Biocompatible peritoneal dialysis solutions: do they indeed affect the outcome? Pol Arch Med Wewn. 2009;119:242–7.
Jones S, Holmes CJ, Krediet RT, Mackenzie R, Faict D, Tranaeus A, Williams JD, Coles GA, Topley N, Bicarbonate/Lactate Study Group. Bicarbonate/lactate-based peritoneal dialysis solution increases cancer antigen 125 and decreases hyaluronic acid levels. Kidney Int. 2001;59:1529–38.
Shimaoka T, Hamada C, Kaneko K, Io H, Sekiguchi Y, Aruga S, Inuma J, Inami Y, Hotta Y, Horikoshi S, Kumasaka T, Tomino Y. Quantitative evaluation and assessment of peritoneal morphologic changes in peritoneal dialysis patients. Quantitative evaluation and assessment of peritoneal morphologic changes in peritoneal dialysis patients. Nephrol Dial Transplant. 2010;25:3379–85.
Nakamura S, Niwa T. Advanced glycation end-products and peritoneal sclerosis. Semin Nephrol. 2004;24:502–5.
Miyata T, Horie K, Ueda Y, Fujita Y, Izuhara Y, Hirano H, Uchida K, Saito A, van Ypersele de Strihou C, Kurokawa K. Advanced glycation and lipid oxidation of the peritoneal membrane: respective roles of serum and peritoneal fluid reactive carbonyl compounds. Kidney Int. 2000;58:425–35.
Ayuzawa N, Ishibashi Y, Takazawa Y, Kume H, Fujita T. Peritoneal morphology after long-term peritoneal dialysis with biocompatible fluid: recent clinical practice in Japan. Perit Dial Int. 2012;32:159–67.
Kawanishi K, Honda K, Tsukada M, Oda H, Nitta K. Neutral solution low in glucose degradation products is associated with less peritoneal fibrosis and vascular sclerosis in patients receiving peritoneal dialysis. Perit Dial Int. 2013;33:242–51.
Mateijsen MA, van der Wal AC, Hendriks PM, Zweers MM, Mulder J, Struijk DG, Krediet RT. Vascular and interstitial changes in the peritoneum of CAPD patients with peritoneal sclerosis. Perit Dial Int. 1999;19:517–25.
Working Group Committee for Preparation of Guidelines for Peritoneal Dialysis, Japanese Society for Dialysis Therapy, Japanese Society for Dialysis Therapy. 2009 Japanese Society for Dialysis Therapy guidelines for peritoneal dialysis. Ther Apher Dial. 2010;14:489–504.
Yamamoto T, Nagasue K, Okuno S, Yamakawa T. The role of peritoneal lavage and the prognostic significance of mesothelial cell area in preventing encapsulating peritoneal sclerosis. Perit Dial Int. 2010;30:343–52.
Conflict of interest
The authors have no conflicts of interest to declare expect Dr. Yasuhiko Ito. He is a professor in endowed chair supported by Baxter Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
On behalf of the Peritoneal Biopsy Study Group of the Japanese Society for Peritoneal Dialysis.
Rights and permissions
About this article
Cite this article
Hamada, C., Honda, K., Kawanishi, K. et al. Morphological characteristics in peritoneum in patients with neutral peritoneal dialysis solution. J Artif Organs 18, 243–250 (2015). https://doi.org/10.1007/s10047-015-0822-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-015-0822-4