Abstract
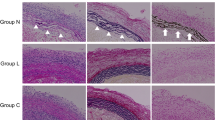
We have succeeded in reducing the calcification of acellular aortas or valves in porcine allogeneic system by removing the DNA and phospholipids, but its further reduction is desirable. Here, the calcification of the acellular tissue was evaluated in rat subcutaneous transplantation model which is known as calcification model. Acellular samples prepared by high-hydrostatic pressure (HHP) protocols with different washing media were implanted and the calcification was monitored under micro-computed tomography for 1 and 3 months. The amount of the calcium deposition was quantitatively evaluated by atomic absorption spectroscopy. A cell culture medium showed very good cell removal ability but led to severe calcification at 1 month, and surprisingly the calcium deposition increased as the washing period increased. This calcification was suppressed by removing the DNA fraction with high DNase concentration. On the other hand, the calcification was greatly reduced when washed with saline even at low DNase concentration after 2 weeks washing. These results suggest that the ion species in the washing medium and the residual DNase cooperatively affect the tendency of in vivo calcification, which led us to the possibility of reduced calcification of acellular cardiac tissues.






Similar content being viewed by others
References
Gilbert TW, Sellaro TL, Badylak SF. Decellularization of tissues and organs. Biomaterials. 2006;27(19):3675–83.
Zeltinger J, et al. Development and characterization of tissue-engineered aortic valves. Tissue Eng. 2001;7(1):9–22.
Erdbrugger W, et al. Decellularized xenogenic heart valves reveal remodeling and growth potential in vivo. Tissue Eng. 2006;12(8):2059–68.
Liu Y, et al. Optimization of a natural collagen scaffold to aid cell-matrix penetration for urologic tissue engineering. Biomaterials. 2009;30(23–24):3865–73.
Schenke-Layland K, et al. Complete dynamic repopulation of decellularized heart valves by application of defined physical signals-an in vitro study. Cardiovasc Res. 2003;60(3):497–509.
Zhou J, et al. Impact of heart valve decellularization on 3-D ultrastructure, immunogenicity and thrombogenicity. Biomaterials. 2010;31(9):2549–54.
Iop L, et al. The influence of heart valve leaflet matrix characteristics on the interaction between human mesenchymal stem cells and decellularized scaffolds. Biomaterials. 2009;30(25):4104–16.
Singelyn JM, et al. Naturally derived myocardial matrix as an injectable scaffold for cardiac tissue engineering. Biomaterials. 2009;30(29):5409–16.
Ingram JH, et al. The use of ultrasonication to aid recellularization of acellular natural tissue scaffolds for use in anterior cruciate ligament reconstruction. Tissue Eng. 2007;13(7):1561–72.
Mirsadraee S, et al. Development and characterization of an acellular human pericardial matrix for tissue engineering. Tissue Eng. 2006;12(4):763–73.
Elkins RC, et al. Decellularized human valve allografts. Ann Thorac Surg. 2001;71(5 Suppl):S428–32.
Schnuriger B, et al. Highly sensitive cardiac troponin in blunt chest trauma: after the gathering comes the scattering? J Trauma. 2011;70(3):766–7.
Lu X, et al. Crosslinking effect of nordihydroguaiaretic acid (NDGA) on decellularized heart valve scaffold for tissue engineering. J Mater Sci Mater Med. 2010;21(2):473–80.
Nam K, et al. Study on the physical properties of tissue-engineered blood vessels made by chemical cross-linking and polymer-tissue cross-linking. J Artif Organs. 2009;12(1):47–54.
Funamoto S, et al. The use of high-hydrostatic pressure treatment to decellularize blood vessels. Biomaterials. 2010;31(13):3590–5.
Stegemann JP, Kaszuba SN, Rowe SL. Review: advances in vascular tissue engineering using protein-based biomaterials. Tissue Eng. 2007;13(11):2601–13.
Fujisato T, et al. Preparation and recellularization of tissue engineered bioscaffold for heart valve replacement. In: Mori H, Matsuda H, editors. Cardiovascular regeneration therapies using tissue engineering approaches. Tokyo: Springer; 2005. p. 83–104.
Simionescu DT, et al. Biocompatibility and remodeling potential of pure arterial elastin and collagen scaffolds. Biomaterials. 2006;27(5):702–13.
Basalyga DM, et al. Elastin degradation and calcification in an abdominal aorta injury model—role of matrix metalloproteinases. Circulation. 2004;110(22):3480–7.
Simionescu A, Simionescu DT, Vyavahare NR. Osteogenic responses in fibroblasts activated by elastin degradation products and transforming growth factor-beta1: role of myofibroblasts in vascular calcification. Am J Pathol. 2007;171(1):116–23.
Lee JS, et al. Elastin calcification in the rat subdermal model is accompanied by up-regulation of degradative and osteogenic cellular responses. Am J Pathol. 2006;168(2):490–8.
Clark JN, et al. Prevention of calcification of bioprosthetic heart valve cusp and aortic wall with ethanol and aluminum chloride. Ann Thorac Surg. 2005;79(3):897–904.
Bailey M, et al. Aluminum chloride pretreatment of elastin inhibits elastolysis by matrix metalloproteinases and leads to inhibition of elastin-oriented calcification. Am J Pathol. 2001;159(6):1981–6.
Hu XJ, et al. Evaluation of a novel tetra-functional branched poly(ethylene glycol) crosslinker for manufacture of crosslinked, decellularized, porcine aortic valve leaflets. J Biomed Mater Res B Appl Biomater. 2014;102(2):322–36.
Lovekamp JJ, et al. Stability and function of glycosaminoglycans in porcine bioprosthetic heart valves. Biomaterials. 2006;27(8):1507–18.
Schoen FJ, et al. Bioprosthetic heart valve calcification: membrane-mediated events and alkaline phosphatase. Bone Miner. 1992;17:129–33.
Schnuriger B, et al. Current practice and the role of the CT in the management of penetrating liver injuries at a level I trauma center. J Emerg Trauma Shock. 2011;4(1):53–7.
Wang JH, et al. The v-3 polyunsaturated fatty acid, eicosapentaenoic acid, attenuates abdominal aortic aneurysm development via suppression of tissue remodeling. PLoS One. 2014;9(5):e96286.
Pyo R, et al. Targeted gene disruption of matrix metalloproteinase-9 (gelatinase B) suppresses development of experimental abdominal aortic aneurysms. J Clin Investig. 2000;105(11):1641–9.
Gertz SD, Kurgan A, Eisenberg D. Aneurysm of the rabbit common carotid artery induced by periarterial application of calcium chloride in vivo. J Clin Investig. 1988;81(3):649–56.
Acknowledgments
A part of this research was supported by the S-innovation Research Program for the “Development of the biofunctional materials for realization of innovative medicine”, Japan Science and Technology Agent (JST).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mahara, A., Sago, M., Yamaguchi, H. et al. Micro-CT evaluation of high pressure-decellularized cardiovascular tissues transplanted in rat subcutaneous accelerated-calcification model. J Artif Organs 18, 143–150 (2015). https://doi.org/10.1007/s10047-014-0808-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10047-014-0808-7