Abstract
Carbohydrates have drawn considerable interest from researchers recently due to their affinity for CO2. However, most of the research in this field has focused on peracetylated derivatives. Compared with acetylated carbohydrates, which have already been studied in depth, methyl d-glucopyranoside derivatives are more stable and could have additional applications. Thus, in the present work, ab initio calculations were performed to elucidate the characteristics of the interactions of methylglucoside derivatives with CO2, and to investigate how the binding energy (ΔE) is affected by isomerization or the introduction of various acyl groups. Four methyl d-glucopyranosides (each with two anomers) bearing acetyl, propionyl, butyryl, and isobutyryl moieties, respectively, were designed as substrates, and the 1:1 complexes of a CO2 molecule with each of these sugar substrates were modeled. The results indicate that ΔE is mainly influenced by interaction distance and the number of negatively charged donors or interacting pairs in the complex; the structure of the acyl group present in the substrate is a secondary influence. Except in the case of methyl 2-O-acetyl-d-glucopyranose, the ΔE values of the α- and β-anomers of each methylglucoside were found to be almost the same. Therefore, we would expect the CO2 affinities of the four derivatives studied here to be as strong as or even stronger than that of peracetylated d-glucopyranose.

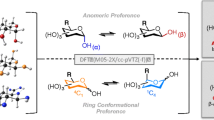
The binding energy between methyl d-glucopyranoside derivatives with various substituted acyl groups and CO2 are evaluated by ab initio calculations. The strong interaction between these methyl dglucopyranoside derivatives and CO2 showed the potential of their application for CO2 capture






















Similar content being viewed by others
Notes
Please note that, in this paper, a “larger” energy is an energy that is larger in magnitude than another energy, and a “smaller” energy is an energy that is smaller in magnitude than another energy. Thus, ΔE of II(a) is larger than ΔE of I(a), even though the ΔE of II(a) is more negative than the ΔE of I(a).
References
Karami D, Mahinpey N (2012) Ind Eng Chem Res 51:4567–4572
Manzanares M, Fabrega C, Osso JO, Vega LF, Andreu T, Morante JR (2014) Appl Catal B Environ 150–151:57–62
Laintz KE, Wai CM, Yonker CR, Smith RD (1991) J Supercrit Fluids 4:194–198
Kazarian SG, Vincent MF, Bright FV, Liotta CL, Eckert CA (1996) J Am Chem Soc 118:1729–1736
Sarbu T, Styranec T, Beckman EJ (2000) Nature 405:165–168
Donat F, Florin NH, Anthony EJ, Fennell PS (2011) Environ Sci Technol 46:1262–1269
Zhao C, Chen X, Zhao C (2010) Int J Greenh Gas Control 4:655–658
Raveendran P, Wallen SL (2000) J Am Chem Soc 124:7274–7275
Potluri VK, Xu J, Enick R, Beckman E, Hamilton AD (2002) Org Lett 4:2333–2335
Ma SL, Wu YT, Hurrey ML, Wallen SL, Grant CS (2010) J Phys Chem B 114:3809–3817
Hurrey ML, Wallen SL (2006) Langmuir 22:7324–7330
Nunesa AVM, Almeidaa APC, Marquesa SR, Sampaio de Sousa AR, Casimiroc T, Duarte CMM (2010) J Supercrit Fluids 54:357–361
Cummings S, Trickett K, Enick R, Eastoe JL (2011) Phys Chem Chem Phys 13:1276–1289
Kim KH, Kim Y (2008) J Phys Chem A 112:1596–1603
Altarsha M, Ingrosso F, Ruiz-Lopez MF (2012) ChemPhysChem 13:3397–3403
Dai HQ, Tri NN, Trang NTT, Trung NT (2014) RSC Adv 4:13901–13908
Zhang PY, Yang HJ, Xu LX (2013) J Chem Thermodyn 67:234–240
Chang HH, Yang CC, Li X, Wei WL, Pang XY, Qiao Y (2014) J Mol Model 20:2259–2268
Chang HH, Yang CC, Li X, Gao WC, Wei WL, Liu G, Pang XY, Qiao Y (2015) CR Chim 18:935–944
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick K, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul G, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian 03, revision B.01. Gaussian, Inc., Pittsburgh
Raveendran P, Wallen SL (2002) J Am Chem Soc 124:12590–12599
Trung NT, Hung NP, Hue TT, Nguyen MT (2011) Phys Chem Chem Phys 13:14033–14042
Wang Y, Hong L, Tapriyal D, Kim IC, Paik IH, Crosthwaite JM, Hamilton AD, Thies MC, Beckman EJ, Enick RM, Johnson JK (2009) J Phys Chem B 113:14971–14980
Acknowledgments
The authors would like to acknowledge the financial support from the Natural Science Foundation of China (no. 21106172), and the Natural Science Foundation for Youths of Shanxi (2013021008-7).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chang, H.H., Cao, R.X., Yang, C.C. et al. Interactions of acylated methylglucoside derivatives with CO2: simulation and calculations. J Mol Model 22, 39 (2016). https://doi.org/10.1007/s00894-015-2903-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00894-015-2903-y