Abstract
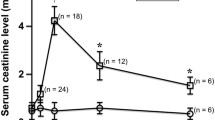
The purpose is to evaluate quantified kidney echogenicity as a biomarker for the early diagnosis of acute kidney injury (AKI) and predicting progression to chronic kidney disease (CKD) in a mouse model of ischemia–reperfusion injury (IRI). Two separate protocols of murine models of IRI were used: (1) 10, 30, and 40 min of bilateral ischemia duration and (2) 45 and 60 min of unilateral ischemia duration. Renal echogenicity was measured with ultrasound and compared with serum creatinine or urine neutrophil gelatinase-associated lipocalin (NGAL) at various timepoints after IRI. In mice subjected to 10, 30, and 40 min of bilateral ischemia, renal echogenicity increased about 2 h after IRI for all ischemia times, earlier than serum creatinine or urine NGAL. In those subjected to 45 and 60 min of unilateral ischemia, 60 min of unilateral ischemia, which represents atrophic changes 28 days after IRI, resulted in a sustained high level of echogenicity and was significantly different 24 h after IRI, while 45 min of unilateral ischemia resulted in trivial levels of histological damage 28 days after IRI. Renal echogenicity might have the potential to be a biomarker for the early diagnosis of AKI and the prognosis of CKD.






Similar content being viewed by others
References
Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group (2013) KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int 3(supple):1–150
Case J, Khan S, Khalid R, Khan A. (2013) Epidemiology of acute kidney injury in the intensive care unit. Crit Care Res Pract. doi:10.1155/2013/479730.
Prasad D (2008) Emerging urinary biomarkers in the diagnosis of acute kidney injury. Expert Opin Med Diagn 2:387–398
Mishra J, Ma Q, Prada A, Mitsnefes M, Zahedi K, Yang J, Barasch J, Devarajan P (2003) Identification of neutrophil gelatinase-associated lipocalin as a novel early urinary biomarker for ischemic renal injury. J Am Soc Nephrol 14:2534–2543
Mishra J, Dent C, Tarabishi R, Mitsnefes MM, Ma Q, Kelly C, Ruff SM, Zahedi K, Shao M, Bean J, Mori K, Barasch J, Devarajan P (2005) Neutrophil gelatinase-associated lipocalin (NGAL) as a biomarker for acute renal injury after cardiac surgery. Lancet 365:1231–1238
Michael RC, John JF, Joseph AE. (2008) Acute kidney injury. In: Brenner BM (eds) Brenner and rector’s the kidney. 8th edn. Saunders, Boston, pp 943–987.
Faubel S, Patel NU, Lockhart ME, Cadnapaphornchai MA (2014) Renal relevant radiology: use of ultrasonography in patients with AKI. Clin J Am Soc Nephrol 9:382–394
Nomura G, Kinoshita E, Yamagata Y, Koga N (1984) Usefulness of renal ultrasonography for assessment of severity and course of acute tubular necrosis. J Clin Ultrasound 12:135–139
Rosenfield AT, Zeman RK, Cicchetti DV, Siegel NJ (1985) Experimental acute tubular necrosis: US appearance. Radiology 157:771–774
Perico N, Cattaneo D, Sayegh MH, Remuzzi G (2004) Delayed graft function in kidney transplantation. Lancet 364:1814–1827
Adachi T, Sugiyama N, Yagita H, Yokoyama T (2014) Renal atrophy after ischemia-reperfusion injury depends on massive tubular apoptosis induced by TNFα in the later phase. Med Mol Morphol 47:213–223
Solez K, Morel-Maroger L, Sraer JD (1979) The morphology of “acute tubular necrosis” in man: analysis of 57 renal biopsies and a comparison with the glycerol model. Medicine (Baltimore) 58:362–376
Adachi T, Sugiyama N, Gondai T, Yagita H, Yokoyama T. (2013) Blockade of death ligand TRAIL inhibits renal ischemia reperfusion injury. Acta Histochem Cytochem 46:161–170
Kelly KJ, Williams WW, Colvin RB, Meehan SM, Springer TA, Gutierrez-Ramos JC, Bonventre JV (1996) Intercellular adhesion molecule-1-deficient mice are protected against ischemic renal injury. J Clin Invest 97(4):1056–1063
Hueper K, Gutberlet M, Rong S, Hartung D, Mengel M, Lu X, Haller H, Wacker F, Meier M, Gueler F (2014) Acute kidney injury: arterial spin labeling to monitor renal perfusion impairment in mice-comparison with histopathologic results and renal function. Radiology 270:117–124
Bonventre JV, Yang L (2011) Cellular pathophysiology of ischemic acute kidney injury. J Clin Invest 121:4210–4221
Nickolas TL, O’Rourke MJ, Yang J, Sise ME, Canetta PA, Barasch N, Buchen C, Khan F, Mori K, Giglio J, Devarajan P, Barasch J (2008) Sensitivity and specificity of a single emergency department measurement of urinary neutrophil gelatinase-associated lipocalin for diagnosing acute kidney injury. Ann Intern Med 148:810–819
Yamamoto T, Noiri E, Ono Y, Doi K, Negishi K, Kamijo A, Kimura K, Fujita T, Kinukawa T, Taniguchi H, Nakamura K, Goto M, Shinozaki N, Ohshima S, Sugaya T (2007) Renal L-type fatty acid-binding protein in acute ischemic injury. J Am Soc Nephrol 18:2894–2902
Negishi K, Noiri E, Maeda R, Portilla D, Sugaya T, Fujita T (2008) Renal L-type fatty acid-binding protein mediates the bezafibrate reduction of cisplatin-induced acute kidney injury. Kidney Int 73:1374–1384
Ferguson MA, Vaidya VS, Waikar SS, Collings FB, Sunderland KE, Gioules CJ, Bonventre JV (2010) Urinary liver-type fatty acid-binding protein predicts adverse outcomes in acute kidney injury. Kidney Int 77:708–714
Liangos O, Perianayagam MC, Vaidya VS, Han WK, Wald R, Tighiouart H, MacKinnon RW, Li L, Balakrishnan VS, Pereira BJ, Bonventre JV, Jaber BL (2007) Urinary N-acetyl-beta-(d)-glucosaminidase activity and kidney injury molecule-1 level are associated with adverse outcomes in acute renal failure. J Am Soc Nephrol 18:904–912
Han WK, Waikar SS, Johnson A, Betensky RA, Dent CL, Devarajan P, Bonventre JV (2008) Urinary biomarkers in the early diagnosis of acute kidney injury. Kidney Int 73:863–869
Parikh CR, Mishra J, Thiessen-Philbrook H, Dursun B, Ma Q, Kelly C, Dent C, Devarajan P, Edelstein CL (2006) Urinary IL-18 is an early predictive biomarker of acute kidney injury after cardiac surgery. Kidney Int 70:199–203
Ikee R, Kobayashi S, Hemmi N, Imakiire T, Kikuchi Y, Moriya H, Suzuki S, Miura S (2005) Correlation between the resistive index by Doppler ultrasound and kidney function and histology. Am J Kidney Dis 46:603–609
Keogan MT, Kliewer MA, Hertzberg BS, DeLong DM, Tupler RH, Carroll BA (1996) Renal resistive indexes: variability in Doppler US measurement in a healthy population. Radiology 199:165–169
Bui TT, Billing H, Alrajab A, Wühl E, Schenk JP (2014) Long-term investigation of kidney ultrasound in cases of hemolytic uremic syndrome in children. J Med. Ultrasonics 41:187–196
Tsau YK, Lee PI, Chang LY, Chen CH. (1997) Correlation of quantitative renal cortical echogenicity with renal function in pediatric renal diseases. Zhonghua Min Guo Xiao Er Ke Yi Xue Hui Za Zhi 38:276–281.
Kasap B, Soylu A, Tu¨rkmen M, Kavukcu S (2006) Relationship of increased renal cortical echogenicity with clinical and laboratory findings in pediatric renal disease. J Clin Ultrasound 34:339–342
Hanamura K, Toji A, Kinugasa S, Asaba K, Fujita T (2012) The resistive index is a marker of renal function, pathology, prognosis, and responsiveness to steroid therapy in chronic kidney disease patients. Int J Nephrol 2012:139565
Siegel MJ. (2011) Spleen and peritoneal cavity. In: Siegel MJ (eds) Pediatric Sonography, 3rd edn. Lippincott Williams & Wilkins, Philadelphia, pp 305–338.
Benter T, Klühs L, Teichgräber U (2011) Sonography of the spleen. J Ultrasound Med 30:1281–1293
Yabuki A, Endo Y, Sakamoto H, Nagayoshi T, Matsumoto M, Suzuki S (2008) Quantitative assessment of renal cortical echogenicity in clinically normal cats. Anat Histol Embryol 37:383–386
Darmon M, Schortgen F, Vargas F, Liazydi A, Schlemmer B, Brun-Buisson C, Brochard L (2011) Diagnostic accuracy of Doppler renal resistive index for reversibility of acute kidney injury in critically ill patients. Intensive Care Med 37:68–76
Lameire N, Van Biesen W, Vanholder R (2005) Acute renal failure. Lancet 365:417–430
Langenberg C, Bagshaw SM, May CN, Bellomo R (2008) The histopathology of septic acute kidney injury: a systematic review. Crit Care 12:R38. doi:10.1186/cc6823
Acknowledgements
The scientific guarantor of this publication is Noriyuki Sugiyama, PhD. The authors of this manuscript declare no relationships with any companies, whose products or services may be related to the subject matter of the article. This work was supported by a Grant-in-Aid for Scientific Research (C, 26461245) from Ministry of Education, Culture, Sports, Science and Technology of Japan. No complex statistical methods were necessary for this paper. Institutional Review Board approval was not required, because this study was only on animal. Approval from the institutional animal care committee was obtained. We are grateful to Dr. Atsushi Yoden, board certified fellow of the Japan Society of Ultrasonics in Medicine (Osaka medical College, Department of Pediatrics), for the valuable suggestions and excellent technical advice regarding ultrasound.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Rights and permissions
About this article
Cite this article
Murata, S., Sugiyama, N., Maemura, K. et al. Quantified kidney echogenicity in mice with renal ischemia reperfusion injury: evaluation as a noninvasive biomarker of acute kidney injury. Med Mol Morphol 50, 161–169 (2017). https://doi.org/10.1007/s00795-017-0157-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00795-017-0157-8