Abstract
Objectives
Root resorptions are common undesirable side effects of orthodontic treatment. In most patients, these defects are repaired by cementoblasts. However, in 1–5 % of patients, the repair fails. The repair mechanism is not well understood. Apoptosis of cementoblasts might contribute to an impaired repair of root resorptions induced by orthodontic forces.
Materials and methods
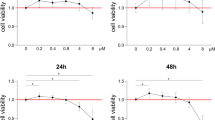
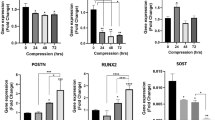
To gain insight into putative molecular pathways leading to compression-induced apoptosis of human primary cementoblasts (HPCBs), three independent cell populations were subjected to compressive loading at 5, 20, and 30 g/cm2 for 1, 6, and 10 h. The mRNA expression of AXUD1, a novel pro-apoptotic gene, was monitored by quantitative reverse transcription PCR (qRT-PCR). To identify a possible function in compression-dependent apoptosis, AXUD1 was silenced in cementoblasts using an siRNA approach. Apoptosis of cementoblasts was measured by annexin V staining and flow cytometry. The phosphorylation of c-Jun-N-terminal kinases (JNKs) was investigated by Western blotting.
Results
AXUD1 was significantly induced in a time- and force-dependent manner. The rate of apoptotic HPCBs increased by 20–40 % after 10 h of compression (30 g/cm2). Phosphorylation of JNKs was detected after 10 h at 30 g/cm2. SiRNA-mediated knockdown of AXUD1 led to decreased phosphorylation of JNKs and reduced apoptosis rates in compressed HPCBs.
Conclusions
Compression-induced apoptosis of HPCBs is mediated by AXUD1 via a JNK-dependent pathway.
Clinical relevance
AXUD1-dependent apoptosis of human cementoblasts might contribute to an impaired repair of root resorptions during orthodontic tooth movement. Further studies are needed to develop treatment strategies aiming to minimize root resorption during orthodontic tooth movement.



Similar content being viewed by others
References
Owman-Moll P, Kurol J, Lundgren D (1995) Repair of orthodontically induced root resorption in adolescents. Angle Orthod 65(6):403–408 discussion 409-410
Weltman B, Vig KW, Fields HW, Shanker S, Kaizar EE (2010) Root resorption associated with orthodontic tooth movement: a systematic review. Am J Orthod Dentofac Orthop 137(4):462–476
Levander E, Malmgren O, Eliasson S (1994) Evaluation of root resorption in relation to two orthodontic treatment regimes. A clinical experimental study. Eur J Orthod 16(3):223–228
Jäger A, Kunert D, Friesen T, Zhang D, Lossdörfer S, Gotz W (2008) Cellular and extracellular factors in early root resorption repair in the rat. Eur J Orthod 30(4):336–345
Owman-Moll P, Kurol J (1998) The early reparative process of orthodontically induced root resorption in adolescents–location and type of tissue. Eur J Orthod 20(6):727–732
Diercke K, Kohl A, Lux CJ, Erber R (2012) IL-1beta and compressive forces lead to a significant induction of RANKL-expression in primary human cementoblasts. J Orofac Orthop 73(5):397–412
Diercke K, Zingler S, Kohl A, Lux CJ, Erber R (2014) Gene expression profile of compressed primary human cementoblasts before and after IL-1beta stimulation. Clin Oral Investig 18(8):1925–1939
Feijoo CG, Sarrazin AF, Allende ML, Glavic A (2009) Cystein-serine-rich nuclear protein 1, Axud1/Csrnp1, is essential for cephalic neural progenitor proliferation and survival in zebrafish. Dev Dyn 238(8):2034–2043
Glavic A, Molnar C, Cotoras D, de Celis JF (2009) Drosophila Axud1 is involved in the control of proliferation and displays pro-apoptotic activity. Mech Dev 126(3–4):184–197
Ishiguro H, Tsunoda T, Tanaka T, Fujii Y, Nakamura Y, Furukawa Y (2001) Identification of AXUD1, a novel human gene induced by AXIN1 and its reduced expression in human carcinomas of the lung, liver, colon and kidney. Oncogene 20(36):5062–5066
Kong D, Zheng T, Zhang M, Wang D, Du S, Li X, Fang J, Cao X (2013) Static mechanical stress induces apoptosis in rat endplate chondrocytes through MAPK and mitochondria-dependent caspase activation signaling pathways. PLoS One 8(7):e69403
Matsui H, Fukuno N, Kanda Y, Kantoh Y, Chida T, Nagaura Y, Suzuki O, Nishitoh H, Takeda K, Ichijo H, Sawada Y, Sasaki K, Kobayashi T, Tamura S (2014) The expression of Fn14 via mechanical stress-activated JNK contributes to apoptosis induction in osteoblasts. J Biol Chem 289(10):6438–6450
Johnson GL, Nakamura K (2007) The c-jun kinase/stress-activated pathway: regulation, function and role in human disease. Biochim Biophys Acta 1773(8):1341–1348
Donovan N, Becker EB, Konishi Y, Bonni A (2002) JNK phosphorylation and activation of BAD couples the stress-activated signaling pathway to the cell death machinery. J Biol Chem 277(43):40944–40949
Diercke K, Konig A, Kohl A, Lux CJ, Erber R (2012) Human primary cementoblasts respond to combined IL-1beta stimulation and compression with an impaired BSP and CEMP-1 expression. Eur J Cell Biol 91(5):402–412
Kitagawa M, Tahara H, Kitagawa S, Oka H, Kudo Y, Sato S, Ogawa I, Miyaichi M, Takata T (2006) Characterization of established cementoblast-like cell lines from human cementum-lining cells in vitro and in vivo. Bone 39(5):1035–1042
Davidovitch Z (1991) Tooth movement. Crit Rev Oral Biol Med 2(4):411–450
Redlich M, Roos H, Reichenberg E, Zaks B, Grosskop A, Bar Kana I, Pitaru S, Palmon A (2004) The effect of centrifugal force on mRNA levels of collagenase, collagen type-I, tissue inhibitors of metalloproteinases and beta-actin in cultured human periodontal ligament fibroblasts. J Periodontal Res 39(1):27–32
Kook SH, Hwang JM, Park JS, Kim EM, Heo JS, Jeon YM, Lee JC (2009) Mechanical force induces type I collagen expression in human periodontal ligament fibroblasts through activation of ERK/JNK and AP-1. J Cell Biochem 106(6):1060–1067
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(−delta delta C(T)) method. Methods 25(4):402–408
Aras B, Cheng LL, Turk T, Elekdag-Turk S, Jones AS, Darendeliler MA (2012) Physical properties of root cementum: part 23. Effects of 2 or 3 weekly reactivated continuous or intermittent orthodontic forces on root resorption and tooth movement: a microcomputed tomography study. Am J Orthod Dentofac Orthop 141(2):e29–e37
Darendeliler MA, Kharbanda OP, Chan EK, Srivicharnkul P, Rex T, Swain MV, Jones AS, Petocz P (2004) Root resorption and its association with alterations in physical properties, mineral contents and resorption craters in human premolars following application of light and heavy controlled orthodontic forces. Orthod Craniofac Res 7(2):79–97
Diercke K, Kohl A, Lux CJ, Erber R (2014) Compression of human primary cementoblasts leads to apoptosis: a possible cause of dental root resorption? J Orofac Orthop 75(6):430–445
Diercke K, Sen S, Kohl A, Lux CJ, Erber R (2011) Compression-dependent up-regulation of ephrin-A2 in PDL fibroblasts attenuates osteogenesis. J Dent Res 90(9):1108–1115
Sen S, Diercke K, Zingler S, Lux CJ, Erber R (2015) Compression induces ephrin-A2 in PDL fibroblasts via c-fos. J Dent Res 94(3):464–472
Wang YL, He H, Liu ZJ, Cao ZG, Wang XY, Yang K, Fang Y, Han M, Zhang C, Huo FY (2015) Effects of TNF-alpha on cementoblast differentiation, mineralization, and apoptosis. J Dent Res 94(9):1225–1232
Garlet TP, Coelho U, Silva JS, Garlet GP (2007) Cytokine expression pattern in compression and tension sides of the periodontal ligament during orthodontic tooth movement in humans. Eur J Oral Sci 115(5):355–362
Huang S, Jiang Y, Li Z, Nishida E, Mathias P, Lin S, Ulevitch RJ, Nemerow GR, Han J (1997) Apoptosis signaling pathway in T cells is composed of ICE/Ced-3 family proteases and MAP kinase kinase 6b. Immunity 6(6):739–749
Cardone MH, Salvesen GS, Widmann C, Johnson G, Frisch SM (1997) The regulation of anoikis: MEKK-1 activation requires cleavage by caspases. Cell 90(2):315–323
Lee JH, Lee DS, Nam H, Lee G, Seo BM, Cho YS, Bae HS, Park JC (2012) Dental follicle cells and cementoblasts induce apoptosis of ameloblast-lineage and Hertwig's epithelial root sheath/epithelial rests of Malassez cells through the Fas-Fas ligand pathway. Eur J Oral Sci 120(1):29–37
Rodrigues LV, Del Puerto HL, Brant JM, Leite RC, Vasconcelos AC (2012) Caspase-3/caspase-8, bax and bcl2 in pulps of human primary teeth with physiological root resorption. Int J Paediatr Dent 22(1):52–59
Gonzalez YR, Zhang Y, Behzadpoor D, Cregan S, Bamforth S, Slack RS, Park DS (2008) CITED2 signals through peroxisome proliferator-activated receptor-gamma to regulate death of cortical neurons after DNA damage. J Neurosci 28(21):5559–5569. doi:10.1523/JNEUROSCI.1014-08.2008
Hao Y, Xu C, Sun SY, Zhang FQ (2009) Cyclic stretching force induces apoptosis in human periodontal ligament cells via caspase-9. Arch Oral Biol 54(9):864–870
Sakai Y, Balam TA, Kuroda S, Tamamura N, Fukunaga T, Takigawa M, Takano-Yamamoto T (2009) CTGF and apoptosis in mouse osteocytes induced by tooth movement. J Dent Res 88(4):345–350
Mabuchi R, Matsuzaka K, Shimono M (2002) Cell proliferation and cell death in periodontal ligaments during orthodontic tooth movement. J Periodontal Res 37(2):118–124
Rana MW, Pothisiri V, Killiany DM, Xu XM (2001) Detection of apoptosis during orthodontic tooth movement in rats. Am J Orthod Dentofac Orthop 119(5):516–521
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Funding
The study was supported by the University of Heidelberg, Germany.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
For this type of study, formal consent is not required.
Electronic Supplementary Material
ESM 1
(PDF 280 kb)
Rights and permissions
About this article
Cite this article
Korb, K., Katsikogianni, E., Zingler, S. et al. Inhibition of AXUD1 attenuates compression-dependent apoptosis of cementoblasts. Clin Oral Invest 20, 2333–2341 (2016). https://doi.org/10.1007/s00784-016-1740-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00784-016-1740-4