Abstract
The coordination compound of the antihypertensive ligand irbesartan (irb) with copper(II) (CuIrb) was synthesized and characterized by FTIR, FT-Raman, UV–visible, reflectance and EPR spectroscopies. Experimental evidence allowed the implementation of structural and vibrational studies by theoretical calculations made in the light of the density functional theory (DFT). This compound was designed to induce structural modifications on the ligand. No antioxidant effects were displayed by both compounds, though CuIrb behaved as a weak 1,1-diphenyl-2-picrylhydrazyl radical (DPPH·) scavenger (IC50 = 425 μM). The measurements of the contractile capacity on human mesangial cell lines showed that CuIrb improved the antihypertensive effects of the parent medication. In vitro cell growth inhibition against prostate cancer cell lines (LNCaP and DU 145) was measured for CuIrb, irbesartan and copper(II). These cell lines have been selected since the angiotensin II type 1 (AT1) receptor (that was blocked by the angiotensin receptor blockers, ARB) has been identified in them. The complex exerted anticancer behavior (at 100 μM) improving the activity of the ligand. Flow cytometry determinations were used to determine late apoptotic mechanisms of cell death.
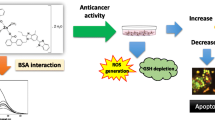
Graphical Abstract
Experimental and DFT characterization of an irbesartan copper(II) complex has been performed. The complex exhibits low scavenging activity against DPPH· and significant growth inhibition of LNCaP and DU 145 prostate cancer cell lines. Flow cytometry determinations were used to determine late apoptotic mechanisms of cell death. This compound improved the antihypertensive effect of irbesartan. This effect was observed earlier for the mononuclear Cu–candesartan complex, but not in structurally modified sartans forming dinuclear or octanuclear Cu–sartan compounds.












Similar content being viewed by others
Abbreviations
- ABTS:
-
2,2′-Azino-bis(3-ethyl-benzothiazoline-6-sulfonic acid diammonium salt)
- Ang II:
-
Angiotensin II
- ARBs:
-
AT1 receptor blockers
- AT1:
-
Ang II type 1 receptor
- CuIrb:
-
[Cu(Irb)2(H2O)], Irb: irbesartan
- DPPH. :
-
1,1-Diphenyl-2-picrylhydrazyl radical
- DTA:
-
Differential thermal analysis
- HMC:
-
Human mesangial cells
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- NBT:
-
Nitroblue tetrazolium
- PBS:
-
Phosphate buffered saline
- PCSA:
-
Planar cell surface area
- PI:
-
Propidium iodide
- RAS:
-
Renin–angiotensin system
- SOD:
-
Superoxide dismutase
- TGA:
-
Thermogravimetric analysis
References
Garrido AM, Griendling KK (2009) Mol Cell Endocrinol 302:148–158
Duncia JV, Chiu AT, Carini DJ, Gregory GB, Johnson AL, Price WA, Wells GJ, Wong PC, Calabrese JC, Timmermans PB (1990) J Med Chem 33:1312–1329
Ram CVS (2008) Am J Med 121:656–663
Fotakis C, Christodouleas D, Zoumpoulakis P, Kritsi E, Benetis N-P, Mavromoustakos T, Reis H, Gili A, Papadopoulos MG, Zervou M (2011) J Phys Chem B 115:6180–6192
Potamitis C, Zervou M, Katsiaras V, Zoumpoulakis P, Kyrikou I, Argyropoulos D, Vatougia G, Mavromoustakos T (2009) J Chem Inform Model 49:726–739
Franca CA, Etcheverry SB, Diez RP, Williams PAM (2009) J Raman Spectrosc 2009:1296–1300
Abali H, Güllü IH, Engin H, Haznedaroğlu IC, Erman M, Tekuzman G (2002) Med Hypotheses 59:344–348
Deshayes F, Nahmias C (2005) Trends Endocrinol Metab 16:293–299
Arafat H, Gong Q, Chipitsyna G, Rizvi A, Saa CT, Yeo CJ (2007) J Am Coll Surg 204:996–1005 (discussion 1005–1006)
Klevay LM, Ann NY (1980) Acad Sci 355:140–151
Suliburska J, Bogdanski P, Jakubowski H (2014) Eur J Pharmacol 738:326–331
Etcheverry SB, Ferrer EG, Naso L, Barrio DA, Lezama L, Rojo T, Williams PAM (2007) Bioorg Med Chem 15:6418–6424
Etcheverry S, Di Virgilio AL, Nascimento OR, Williams PAM (2012) J Inorg Biochem 107:25–33
Islas MS, Lezama L, Griera Merino M, Cortes MA, Rodriguez Puyol M, Ferrer EG, Williams PAM (2013) J Inorg Biochem 123:23–33
Islas MS, Martínez Medina JJ, López Tévez LL, Rojo T, Lezama L, Griera Merino M, Calleros L, Cortes MA, Rodriguez Puyol M, Echeverría GA, Piro OE, Ferrer EG, Williams PAM (2014) Inorg Chem 53:5724–5737
Chow L, Rezmann L, Imamura K, Wang L, Catt K, Tikellis C, Louis WJ, Frauman AG, Louis SNS (2008) Prostate 68:651–660
Analytische Messtechnik-GmbH (1996) WINEPR SimFonia v1.25 software, Bruker
Zhao Y, Truhlar DG (2007) Theor Chem Acc 120:215–241
Dunning TH, Hay PJ (1977) In: Schaefer HF (ed) Methods of electronic structure theory, vol 3. Plenum press, New York
Casida ME, Jamorski C, Casida KC, Salahub DRJ (1998) Chem Phys 108:4439–4449
Keresztury G, Holly S, Besenyei G, Varga J, Wang A, Durig JR (1993) Spectrochim Acta Part A Mol Spectrosc 49:2007–2026
Cariaga-Martinez AE, López-Ruiz P, Nombela-Blanco MP, Motiño O, González-Corpas A, Rodriguez-Ubreva J, Lobo MVT, Cortés MA, Colás B (2013) Cell Signal 25:1586–1597
Luo C, Li Y, Zhou B, Yang L, Li H, Feng Z, Li Y, Long J, Liu J (2014) Apoptosis 19:542–553
Calleros L, Lasa M, Rodríguez-Álvarez F, Toro M, Chiloeches A (2006) Apoptosis 11:1161–1173
Williams PAM, Ferrer EG, Correa MJ, Baran EJ, Castellano EE, Piro OE (2004) J Chem Crystallogr 34:285–290
Dutta RL, Satapathi SK (1981) J Inorg Nucl Chem 43:1533–1539
Lane TJ, Nakagawa I, Walter JL, Kandathil AJ (1962) Inorg Chem 1:267–276
Klapötke TM, Schmid PC, Stierstorfer J, Szimhardt N (2016) Z Anorg Allg Chem 642:383–389
Nakamoto K (2009) Infrared and Raman spectra of inorganic and coordination compounds, Part B, 6th edn. John Wiley and Sons Inc, New Jersey
van Albada GA, Quiroz-Castro ME, Mutikainen I, Turpeinen U, Reedijk J (2000) Inorg Chim Acta 298:221–225
Tabbì G, Giuffrida A, Bonomo RP (2013) J Inorg Biochem 128:137–145
Hathaway BJ, Billing DE (1970) Coord Chem Rev 5:143–207
Yamaguchi T, Takamura H, Matoba T, Terao J (1998) Biosci Biotechnol Biochem 62:1201–1204
Wang M, Zhao X, Gai C, Liu H, Xu Y, Li J (2011) Chem Res Chinese Univ 27:1–5
Ohno K, Amano Y, Kakuta H, Niimi T, Takakura S, Orita M, Miyata K, Sakashita H, Takeuchi M, Komuro I, Higaki J, Horiuchi M, Kim-Mitsuyama S, Mori Y, Morishita R, Yamagishi S (2011) Biochem Biophys Res Commun 404:434–437
Funao K, Matsuyama M, Kawahito Y, Sano H, Chargui J, Touraine JL, Nakatani T, Yoshimura R (2008) Oncol Rep 20:295–300
Acknowledgments
This work was supported by UNLP, CONICET (PIP 0611), CICPBA and by ANPCyT (PICT2013 0569), Argentina and Grant Renal Research Network: FEDER funds ISCIII RETIC REDINREN RD012/20021/0006, by Grant from Fondo de Investigaciones Sanitarias (FIS/ISCIII PI11/01630 and PI14/02075) to DRP and (FIS/ISCIII PI14/01939) to MRP integrated into the National Plan of I + D + i and co-funded by FEDER and the Instituto de Salud Carlos III and by Instituto de Investigaciones Sanitarias Reina Sofía (IRSIN) and Fundación Renal Iñigo Álvarez de Toledo (FRIAT). EGF and PAMW are research fellows of CONICET and CICPBA, Argentina, respectively. MSI is a fellowship holder from CONICET.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Islas, M.S., Luengo, A., Franca, C.A. et al. Experimental and DFT characterization, antioxidant and anticancer activities of a Cu(II)–irbesartan complex: structure–antihypertensive activity relationships in Cu(II)–sartan complexes. J Biol Inorg Chem 21, 851–863 (2016). https://doi.org/10.1007/s00775-016-1384-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-016-1384-5