Abstract
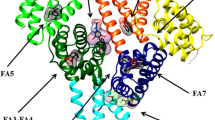
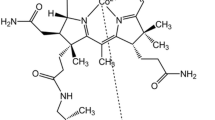
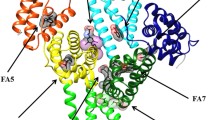
Human serum heme–albumin (HSA–heme–Fe) displays reactivity and spectroscopic properties similar to those of heme proteins. Here, the nitrite reductase activity of ferrous HSA–heme–Fe [HSA–heme–Fe(II)] is reported. The value of the second-order rate constant for the reduction of \( {\text{NO}}_{2}^{ - } \) to NO and the concomitant formation of nitrosylated HSA–heme–Fe(II) (i.e., k on) is 1.3 M−1 s−1 at pH 7.4 and 20 °C. Values of k on increase by about one order of magnitude for each pH unit decrease between pH 6.5 to 8.2, indicating that the reaction requires one proton. Warfarin inhibits the HSA–heme–Fe(II) reductase activity, highlighting the allosteric linkage between the heme binding site [also named the fatty acid (FA) binding site 1; FA1] and the drug-binding cleft FA2. The dissociation equilibrium constant for warfarin binding to HSA–heme–Fe(II) is (3.1 ± 0.4) × 10−4 M at pH 7.4 and 20 °C. These results: (1) represent the first evidence for the \( {\text{NO}}_{2}^{ - } \) reductase activity of HSA–heme–Fe(II), (2) highlight the role of drugs (e.g., warfarin) in modulating HSA(–heme–Fe) functions, and (3) strongly support the view that HSA acts not only as a heme carrier but also displays transient heme-based reactivity.





Similar content being viewed by others
Abbreviations
- FA:
-
Fatty acid
- HSA:
-
Human serum albumin
- HSA–heme–Fe:
-
Human serum heme-albumin
- HSA–heme–Fe(II):
-
Ferrous HSA–heme–Fe
- HSA–heme–Fe(II)–NO:
-
Ferrous nitrosylated HSA–heme–Fe
- HSA–heme–Fe(III):
-
Ferric HSA–heme–Fe
References
Ascenzi P, Fasano M (2009) IUBMB Life 61:1118–1122
Ascenzi P, Fasano M (2010) Biophys Chem 148:16–22
Fanali G, di Masi A, Trezza V, Marino M, Fasano M, Ascenzi P (2012) Mol Aspects Med 33:209–290
Komatsu T, Nakagawa A, Qu X (2009) Drug Metab Pharmacokinet 24:287–299
Tsuchida E, Sou K, Nakagawa A, Sakai H, Komatsu T, Kobayashi K (2009) Bioconjugate Chem 20:1419–1440
Ascenzi P, Imperi F, Coletta M, Fasano M (2008) Biochem Biophys Res Commun 369:686–691
Ascenzi P, di Masi A, De Sanctis G, Coletta M, Fasano M (2009) Biochem Biophys Res Comm 387:83–86
Ascenzi P, Cao Y, Tundo GR, Coletta M, Fanali G, Fasano M (2011) Biochem Biophys Res Commun 411:185–189
Ascenzi P, di Masi A, Leboffe L, Alberio T, Fanali G, Fasano M (2013) IUBMB Life. doi:10.1002/iub.1164
Bocedi A, De Sanctis G, Ciaccio C, Tundo GR, di Masi A, Fanali G, Nicoletti FP, Fasano M, Smulevich G, Ascenzi P, Coletta M (2013) PLoS One 8:e58842
Fasano M, Baroni S, Vannini A, Ascenzi P, Aime S (2001) J Biol Inorg Chem 6:650–658
Monzani E, Bonafè B, Fallarini A, Redaelli C, Casella L, Minchiotti L, Galliano M (2001) Biochim Biophys Acta 1547:302–312
Ascenzi P, Cao Y, di Masi A, Gullotta F, De Sanctis G, Fanali G, Fasano M, Coletta M (2010) FEBS J 277:2474–2485
Ascenzi P, Gullotta F, Gioia M, Coletta M, Fasano M (2011) Biochem Biophys Res Commun 406:112–116
Ascenzi P, Fasano M (2007) Biochem Biophys Res Commun 353:469–474
Ascenzi P, di Masi A, Coletta M, Ciaccio C, Fanali G, Nicoletti FP, Smulevich G, Fasano M (2009) J Biol Chem 284:31006–31017
Ascenzi P, Bolli A, Gullotta F, Fanali G, Fasano M (2010) IUBMB Life 62:776–780
Ascenzi P, Bolli A, di Masi A, Tundo GR, Fanali G, Coletta M, Fasano M (2011) J Biol Inorg Chem 16:97–108
Cha MK, Kim IH (1996) Biochem Biophys Res Commun 222:619–625
Kamal JKA, Behere DV (2002) J Biol Inorg Chem 7:273–283
Doyle MP, Pickering RA, DeWeert TM, Hoekstra JW, Pater D (1981) J Biol Chem 256:12393–12398
Huang Z, Shiva S, Kim-Shapiro DB, Patel RP, Ringwood LA, Irby CE, Huang KT, Ho C, Hogg N, Schechter AN, Gladwin MT (2005) J Clin Invest 115:2099–2107
Huang KT, Keszler A, Patel N, Patel RP, Gladwin MT, Kim-Shapiro DB, Hogg N (2005) J Biol Chem 280:31126–31131
Shiva S, Huang Z, Grubina R, Sun J, Ringwood LA, MacArthur PH, Xu X, Murphy E, Darley-Usmar VM, Gladwin MT (2007) Circ Res 100:654–661
Grubina R, Basu S, Tiso M, Kim-Shapiro DB, Gladwin MT (2008) J Biol Chem 283:3628–3638
Petersen MG, Dewilde S, Fago A (2008) J Inorg Biochem 102:1777–1782
Salhany JM (2008) Biochemistry 47:6059–6072
Sturms R, DiSpirito AA, Hargrove MS (2011) Biochemistry 50:3873–3878
Li H, Hemann C, Abdelghany TM, El-Mahdy MA, Zweier JL (2012) J Biol Chem 287:36623–36633
Tiso M, Tejero J, Basu S, Azarov I, Wang X, Simplaceanu V, Frizzell S, Jayaraman T, Geary L, Shapiro C, Ho C, Shiva S, Kim-Shapiro DB, Gladwin MT (2011) J Biol Chem 286:18277–18289
Tiso M, Tejero J, Kenney C, Frizzell S, Gladwin MT (2012) Biochemistry 51:5285–5292
Baroni S, Mattu M, Vannini A, Cipollone R, Aime S, Ascenzi P, Fasano M (2001) Eur J Biochem 268:6214–6220
Fasano M, Mattu M, Coletta M, Ascenzi P (2002) J Inorg Biochem 91:487–490
Mattu M, Vannini A, Coletta M, Fasano M, Ascenzi P (2001) J Inorg Biochem 84:293–296
Nicoletti FP, Howes BD, Fittipaldi M, Fanali G, Fasano M, Ascenzi P, Smulevich G (2008) J Am Chem Soc 130:11677–11688
Sugio S, Kashima A, Mochizuki S, Noda M, Kobayashi K (1999) Protein Eng 12:439–446
Bhattacharya AA, Grüne T, Curry S (2000) J Mol Biol 303:721–732
Zunszain PA, Ghuman J, Komatsu T, Tsuchida E, Curry S (2003) BMC Struct Biol 3:6
Radwan MA, Bawazeer GA, Aloudah NM, AlQuadeib BT, Aboul-Enein HY (2012) Biomed Chromatogr 26:6–11
Acknowledgments
This work was partially supported by grants from the Ministero dell’Istruzione, dell’Università e della Ricerca of Italy (PRIN 20109MXHMR_001 and Università Roma Tre, CLAR 2013) to P.A.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ascenzi, P., Tundo, G.R., Fanali, G. et al. Warfarin modulates the nitrite reductase activity of ferrous human serum heme–albumin. J Biol Inorg Chem 18, 939–946 (2013). https://doi.org/10.1007/s00775-013-1040-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-013-1040-2