Abstract
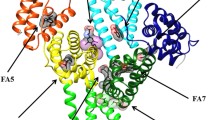
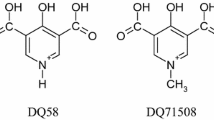
Human serum heme–albumin (HSA-heme) displays globin-like properties. Here, the allosteric inhibition of ferric heme [heme-Fe(III)] binding to human serum albumin (HSA) and of ferric HSA–heme [HSA-heme-Fe(III)]-mediated peroxynitrite isomerization by isoniazid and rifampicin is reported. Moreover, the allosteric inhibition of isoniazid and rifampicin binding to HSA by heme-Fe(III) has been investigated. Data were obtained at pH 7.2 and 20.0 °C. The affinity of isoniazid and rifampicin for HSA [K 0 = (3.9 ± 0.4) × 10−4 and (1.3 ± 0.1) × 10−5 M, respectively] decreases by about 1 order of magnitude upon heme-Fe(III) binding to HSA [K h = (4.3 ± 0.4) × 10−3 and (1.2 ± 0.1) × 10−4 M, respectively]. As expected, the heme-Fe(III) affinity for HSA [H 0 = (1.9 ± 0.2) × 10−8 M] decreases by about 1 order of magnitude in the presence of saturating amounts of isoniazid and rifampicin [H d = (2.1 ± 0.2) × 10−7 M]. In the absence and presence of CO2, the values of the second-order rate constant (l on) for peroxynitrite isomerization by HSA-heme-Fe(III) are 4.1 × 105 and 4.3 × 105 M−1 s−1, respectively. Moreover, isoniazid and rifampicin inhibit dose-dependently peroxynitrite isomerization by HSA-heme-Fe(III) in the absence and presence of CO2. Accordingly, isoniazid and rifampicin impair in a dose-dependent fashion the HSA-heme-Fe(III)-based protection of free l-tyrosine against peroxynitrite-mediated nitration. This behavior has been ascribed to the pivotal role of Tyr150, a residue that either provides a polar environment in Sudlow’s site I (i.e., the binding pocket of isoniazid and rifampicin) or protrudes into the heme-Fe(III) cleft, depending on ligand binding to Sudlow’s site I or to the FA1 pocket, respectively. These results highlight the role of drugs in modulating heme-Fe(III) binding to HSA and HSA-heme-Fe(III) reactivity.






Similar content being viewed by others
Abbreviations
- FA:
-
Fatty acid
- Heme–Fe(III):
-
Ferric heme
- HSA:
-
Human serum albumin
- HSA-heme:
-
Human serum heme–albumin
- HSA-heme-Fe(II)-NO:
-
Ferrous nitrosylated human serum heme–albumin
- HSA-heme-Fe(III):
-
Ferric human serum heme–albumin
References
Sudlow G, Birkett DJ, Wade DN (1975) Mol Pharmacol 11:824–832
Peters T Jr (ed) (1996) All about albumin: biochemistry, genetics and medical applications. Academic Press, San Diego
Curry S (2002) Vox Sang 83(Suppl 1):315–319
Kragh-Hansen U, Chuang VT, Otagiri M (2002) Biol Pharm Bull 25:695–704
Sakurai Y, Ma SF, Watanabe H, Yamaotsu N, Hirono S, Kurono Y, Kragh-Hansen U, Otagiri M (2004) Pharm Res 21:285–292
Sułkowska A, Bojko B, Równicka J, Sułkowski W (2004) Biopolymers 74:256–262
Ghuman J, Zunszain PA, Petitpas I, Bhattacharya AA, Otagiri M, Curry S (2005) J Mol Biol 353:38–52
Ascenzi P, Bocedi A, Notari S, Fanali G, Fesce R, Fasano M (2006) Mini Rev Med Chem 6:483–489
Ascenzi P, Fasano M (2010) Biophys Chem 148:16–22
Curry S, Mandelkov H, Brick P, Franks N (1998) Nat Struct Biol 5:827–835
Sugio S, Kashima A, Mochizuki S, Noda M, Kobayashi K (1999) Protein Eng 12:439–446
Yamasaki K, Maruyama T, Yoshimoto K, Tsutsumi Y, Narazaki R, Fukuhara A, Kragh-Hansen U, Otagiri M (1999) Biochim Biophys Acta 1432:313–323
Bhattacharya AA, Curry S, Franks NP (2000) J Biol Chem 275:38731–38738
Bhattacharya AA, Grüne T, Curry S (2000) J Mol Biol 303:721–732
Petitpas I, Bhattacharya AA, Twine S, East M, Curry S (2001) J Biol Chem 276:22804–22809
Chuang VTG, Otagiri M (2002) Pharm Res 19:1458–1464
Hamilton JA (2004) Prog Lipid Res 43:177–199
Lejon S, Frick IM, Björck L, Wikström M, Svensson S (2004) J Biol Chem 279:42924–42928
Curry S (2009) Drug Metab Pharmacokinet 24:342–357
Miller YI, Shaklai N (1999) Biochim Biophys Acta 1454:153–164
Ascenzi P, Bocedi A, Notari S, Menegatti E, Fasano M (2005) Biochem Biophys Res Commun 334:481–486
Bocedi A, Notari S, Menegatti E, Fanali G, Fasano M, Ascenzi P (2005) FEBS J 272:6287–6296
Fasano M, Fanali G, Leboffe L, Ascenzi P (2007) IUBMB Life 59:436–440
Fasano M, Baroni S, Vannini A, Ascenzi P, Aime S (2001) J Biol Inorg Chem 6:650–658
Wardell M, Wang Z, Ho JX, Robert J, Rüker F, Ruble J, Carter DC (2002) Biochem Biophys Res Commun 291:813–819
Nicoletti FP, Howes BD, Fittipaldi M, Fanali G, Fasano M, Ascenzi P, Smulevich G (2008) J Am Chem Soc 130:11677–11688
Komatsu T, Matsukawa Y, Tsuchida E (2000) Bioconjug Chem 11:772–776
Monzani E, Bonafé B, Fallarini A, Redaelli C, Casella L, Minchiotti L, Galliano M (2001) Biochim Biophys Acta 1547:302–312
Kamal JK, Behere DV (2002) J Biol Inorg Chem 7:273–283
Komatsu T, Ohmichi N, Nakagawa A, Zunszain PA, Curry S, Tsuchida E (2005) J Am Chem Soc 127:15933–15942
Ascenzi P, Imperi F, Coletta M, Fasano M (2008) Biochem Biophys Res Commun 369:686–691
Fasano M, Fanali G, Fesce R, Ascenzi P (2008) In: Bolognesi M, di Prisco G, Verde C (eds) Dioxygen binding and sensing proteins. Springer, Heidelberg, pp 121–131
Ascenzi P, di Masi A, Coletta M, Ciaccio C, Fanali G, Nicoletti FP, Smulevich G, Fasano M (2009) J Biol Chem 284:31006–31017
Baroni S, Mattu M, Vannini A, Cipollone R, Aime S, Ascenzi P, Fasano M (2001) Eur J Biochem 268:6214–6220
Mattu M, Vannini A, Coletta M, Fasano M, Ascenzi P (2001) J Inorg Biochem 84:293–296
Fasano M, Mattu M, Coletta M, Ascenzi P (2002) J Inorg Biochem 91:487–490
Monzani E, Curto M, Galliano M, Minchiotti L, Aime S, Baroni S, Fasano M, Amoresano A, Salzano AM, Pucci P, Casella L (2002) Biophys J 83:2248–2258
Fanali G, Fesce R, Agrati C, Ascenzi P, Fasano M (2005) FEBS J 272:4672–4683
Fanali G, Bocedi A, Ascenzi P, Fasano M (2007) FEBS J 274:4491–4502
Fanali G, De Sanctis G, Gioia M, Coletta M, Ascenzi P, Fasano M (2009) J Biol Inorg Chem 14:209–217
Fanali G, Pariani G, Ascenzi P, Fasano M (2009) FEBS J 276:2241–2250
Kragh-Hansen U, Watanabe H, Nakajou K, Iwao Y, Otagiri M (2006) J Mol Biol 363:702–712
Simard JR, Zunszain PA, Hamilton JA, Curry S (2006) J Mol Biol 361:336–351
du Toit LC, Pillay V, Danckwerts MP (2006) Respir Res 7:118
Wyman J Jr (1964) Adv Protein Chem 19:223–286
Kharitonov VG, Sharma VS, Magde D, Koesling D (1997) Biochemistry 36:6814–6818
Boffi A, Das TK, Della Longa S, Spagnolo C, Rousseau DL (1999) Biophys J 77:1143–1149
Ascenzi P, Fasano M (2007) Biochem Biophys Res Commun 353:469–474
Notari S, Mancone C, Sergi M, Gullotta F, Bevilacqua N, Tempestilli M, Urso R, Lauria FN, Pucillo LP, Tripodi M, Ascenzi P (2010) IUBMB Life 62:387–393
Bohle DS, Glassbrenner PA, Hansert B (1996) Methods Enzymol 269:302–311
Koppenol WH, Kissner R, Beckman JS (1996) Methods Enzymol 269:296–302
Herold S, Exner M, Boccini F (2003) Chem Res Toxicol 16:390–402
Herold S, Kalinga S, Matsui T, Watanabe Y (2004) J Am Chem Soc 126:6945–6955
Ascenzi P, Visca P (2008) Methods Enzymol 436:317–337
Goldstein S, Merényi G (2008) Methods Enzymol 436:49–61
Goldstein S, Lind J, Merényi G (2005) Chem Rev 105:2457–2470
Bocedi A, Notari S, Narciso P, Bolli A, Fasano M, Ascenzi P (2004) IUBMB Life 56:609–614
Yang JD, Deng SX, Liu ZF, Kong L, Liu SP (2007) Luminescence 22:559–566
Herold S, Kalinga S (2003) Biochemistry 42:14036–14046
Herold S, Matsui T, Watanabe Y (2001) J Am Chem Soc 123:4085–4086
Miranda KM, Espey MG, Wink DA (2001) Nitric Oxide 5:62–71
Ascenzi P, Bocedi A, Bolognesi M, Fabozzi G, Milani M, Visca P (2006) Biochem Biophys Res Commun 339:450–456
Goodsell DS, Olson AJ (1990) Proteins 8:195–202
Goodsell DS, Morris GM, Olson AJ (1998) J Mol Recogn 9:1–5
Morris GM, Goodsell DS, Halliday RS, Huey R, Hart WE, Belew RK, Olson AJ (1998) J Comput Chem 19:1639–1662
Metcalfe C, Macdonald IK, Murphy EJ, Brown KA, Raven EL, Moody PC (2008) J Biol Chem 283:6193–6200
Campbell EA, Korzheva N, Mustaev A, Murakami K, Nair S, Goldfarb A, Darst SA (2001) Cell 104:901–912
Maes V, Engelborghs Y, Hoebeke J, Maras Y, Vercruysse A (1982) Mol Pharmacol 21:100–107
Fanali G, Rampoldi V, di Masi A, Bolli A, Lopiano L, Ascenzi P, Fasano M (2010) IUBMB Life 62:371–376
Herold S, Fago A (2005) Comp Biochem Physiol A Mol Integr Physiol 142:124–129
Ascenzi P, di Masi A, Sciorati C, Clementi E (2010) Biofactors 36:264–273
Fasano M, Curry S, Terreno E, Galliano M, Fanali G, Narciso P, Notari S, Ascenzi P (2005) IUBMB Life 57:787–796
Muller-Eberhard U, Javid J, Liem HH, Hanstein A, Hanna M (1968) Blood 32:811–815
Houin G, Beucler A, Richelet S, Brioude R, Lafaix C, Tillement JP (1983) Ther Drug Monit 5:67–72
Delahunty T, Lee B, Conte JE (1998) J Chromatogr B Biomed Sci Appl 705:323–329
Alvarez B, Radi R (2003) Amino Acids 25:295–311
Pfeiffer S, Gorren AC, Schmidt K, Werner ER, Hansert B, Bohle DS, Mayer B (1997) J Biol Chem 272:3465–3470
Zunszain PA, Ghuman J, Komatsu T, Tsuchida E, Curry S (2003) BMC Struct Biol 3:6
Acknowledgments
This work was partially supported by grants from the Ministero dell’Istruzione, dell’Università e della Ricerca of Italy (PRIN 2007ECX29E_002 and University Roma Tre, CLAR 2009, to P.A.) and from the Ministero della Salute of Italy (Istituto Nazionale per le Malattie Infettive I.R.C.C.S. ‘Lazzaro Spallanzani’, Ricerca corrente 2009 to P.A.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ascenzi, P., Bolli, A., di Masi, A. et al. Isoniazid and rifampicin inhibit allosterically heme binding to albumin and peroxynitrite isomerization by heme–albumin. J Biol Inorg Chem 16, 97–108 (2011). https://doi.org/10.1007/s00775-010-0706-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-010-0706-2