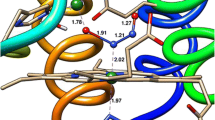
Abstract
The mechanism for the reduction of nitric oxide to nitrous oxide and water in an A-type flavoprotein (FprA) in Moorella thermoacetica, which has been proposed to be a scavenging type of nitric oxide reductase, has been investigated using density functional theory (B3LYP). A dinitrosyl complex, [{FeNO}7]2, has previously been proposed to be a key intermediate in the NO reduction catalyzed by FprA. The electrons and protons involved in the reduction were suggested to “super-reduce” the dinitrosyl intermediate to [{FeNO}8]2 or the corresponding diprotonated form, [{FeNO(H)}8]2. In this type of mechanism the electron and/or proton transfers will be a part of the rate-determining step. In the present study, on the other hand, a reaction mechanism is suggested in which N2O can be formed before the protons and electrons enter the catalytic cycle. One of the irons in the diiron center is used to stabilize the formation of a hyponitrite dianion, instead of binding a second NO. Cleaving the N–O bond in the hyponitrite dianion intermediate is the rate-determining step in the proposed reaction mechanism. The barrier of 16.5 kcal mol−1 is in good agreement with the barrier height of the experimental rate-determining step of 14.8 kcal mol−1. The energetics of some intermediates in the “super-reduction” mechanism and the mechanism proceeding via a hyponitrite dianion are compared, favoring the latter. It is also discussed how to experimentally discriminate between the two mechanisms.








Similar content being viewed by others
References
Zumft WG (1997) Microbiol Mol Biol Rev 61:533–616
Watmough NJ, Butland G, Cheesman MR, Moir JWB, Richardson DJ, Spiro S (1999) Biochim Biophys Acta 1411:456–474
Ischiropoulos H (1998) Arch Biochem Biophys 356:1–11
Radi R, Beckman JS, Bush KM, Freeman BA (1991) J Biol Chem 266:4244–4250
King PA, Anderson VE, Edwards JO, Gustafson G, Plumb RC, Suggs JW (1992) J Am Chem Soc 114:5430–5432
Bredt DS, Snyder SH (1994) Annu Rev Biochem 63:175–195
Silaghi-Dumitrescu R, Kurtz DM Jr, Ljungdahl LG, Lanzilotta WN (2005) Biochemistry 44:6492–6501
Silaghi-Dumitrescu R, Ng KY, Viswanathan R, Kurtz DM Jr (2005) Biochemistry 44:3572–3579
Gomes CM, Giuffré A, Forte E, Vicente JB, Saraiva LM, Brunori M, Teixeira M (2002) J Biol Chem 277:25273–25276
Gardner AM, Helmick RA, Gardner PR (2002) J Biol Chem 277:8172–8177
Rodrigues R, Vicente JB, Félix R, Oliveira S, Teixeira M, Rodrigues-Pousada C (2006) J Bacteriol 188:2745–2751
Silaghi-Dumitrescu R, Coulter ED, Das A, Ljungdahl LG, Jameson GNL, Huynh BH, Kurtz DM Jr (2003) Biochemistry 42:2806–2815
Haskin CJ, Ravi N, Lynch JB, Münck E, Que L Jr (1995) Biochemistry 34:11090–11098
Coufal DE, Tavares P, Pereira AS, Hyunh BH, Lippard SJ (1999) Biochemistry 38:4504–4513
Daiber A, Shoun H, Ullrich V (2005) J Inorg Biochem 99:185–193
Vincent MA, Hillier IH, Ge J (2005) Chem Phys Lett 407:333–336
Silaghi-Dumitrescu R (2003) Eur J Inorg Chem 2003:1048–1052
Becke AD (1993) J Chem Phys 98:5648–5652
Lee C, Yang W, Parr RG (1988) Phys Rev B 37:785–789
Frisch MJ et al (2003) Gaussian 03, revision B.03. Gaussian, Pittsburgh
Schrodinger (2003) Jaguar 5.5. Schrodinger, Portland
Blomberg LM, Blomberg MRA, Siegbahn PEM (2004) J Biol Inorg Chem 9:923–935
Blomberg LM, Blomberg MRA, Siegbahn PEM, van der Donk WA, Tsai A-L (2003) J Phys Chem B 107:3297–3308
Tannor DJ, Marten B, Murphy R, Friesner RA, Sitkoff D, Nicholls A, Ringnalda M, Goddard WA III, Honig B (1994) J Am Chem Soc 116:11875–11882
Blomberg MRA, Siegbahn PEM, Babcock GT (1998) J Am Chem Soc 120:8812–8824
Curtiss LA, Raghavachari K, Redfern RC, Pople JA (2000) J Chem Phys 112:7374–7383
Siegbahn PEM, Blomberg MRA (1999) Annu Rev Phys Chem 50:221–249
Siegbahn PEM, Blomberg MRA (2000) Chem Rev 100:421–437
Blomberg MRA, Siegbahn PEM (2001) J Phys Chem B 105:9375–9386
Silaghi-Dumitrescu R, Silaghi-Dumitrescu I (2006) J Inorg Biochem 100:161–166
Blomberg LM, Blomberg MRA, Siegbahn PEM (2005) J Inorg Biochem 99:949–958
Schenk G, Pau MYM, Solomon EI (2004) J Am Chem Soc 126:505–515
Kurtz DM Jr (1990) Chem Rev 90:585–606
Gomes CM, Vicente JB, Wasserfallen A, Teixeira M (2000) Biochemistry 39:16230–16237
Zhang Y, Pavlosky MA, Brown CA, Westre TE, Hedman B, Hodgson KO, Solomon EI (1992) J Am Chem Soc 114:9189–9191
Brown CA, Pavlosky MA, Westre TE, Zhang Y, Hedman B, Hodgson KO, Solomon EI (1995) J Am Chem Soc 117:715–732
Galpin JR, Veldink GA, Vliegenthart JFG, Boldingh J (1978) Biochim Biophys Acta 536:356–362
Feig AL, Bautista MT, Lippard SL (1996) Inorg Chem 35:6892–6898
Nocek JM, Kurtz DM Jr, Sage JT, Xia Y-M, Debrunner P, Shiemke AK, Sanders-Loehr J, Loehr TM (1988) Biochemistry 27:1014–1024
Nelson MJ (1987) J Biol Chem 262:12137–12412
Shafirovich V, Lymar SV (2002) Proc Natl Acad Sci USA 99:7340–7345
Bassan A, Blomberg MRA, Siegbahn PEM (2003) Chem Eur J 9:4055–4067
Bassan A, Blomberg MRA, Siegbahn PEM (2003) Chem Eur J 7:106–115
Cheesman MR, Zumft WG, Thomson A (1998) J Biochem 37:3994–4000
Hendriks J, Warne A, Gohlke U, Halita T, Ludovici C, Lübben M, Saraste M (1998) Biochemistry 37:13102–13109
Blomberg LM, Blomberg MRA, Siegbahn PEM (2006) Biochim Biophys Acta 1757:240–252
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Blomberg, L.M., Blomberg, M.R.A. & Siegbahn, P.E.M. Theoretical study of the reduction of nitric oxide in an A-type flavoprotein. J Biol Inorg Chem 12, 79–89 (2007). https://doi.org/10.1007/s00775-006-0166-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00775-006-0166-x