Abstract
Introduction
The periosteum has a bilayered structure that surrounds cortical bone. The outer layer is rich in connective tissue and fibroblasts, while the inner layer in contact with the cortical surface of the bone predominantly consists of osteoblasts and osteoblast progenitors. The identification of cell-specific surface markers of the bilayered structure of the periosteum is important for the purpose of tissue regeneration.
Materials and methods
We investigated the expression of the discoidin domain tyrosine kinase receptor DDR2, fibroblast specific protein-1 (FSP-1) and alkaline phosphatase (ALP) in the periosteum of cortical bone by immunohistochemistry. Osteogenic differentiation was compared between DDR2- and FSP-1-expressing cells flow-sorted from the periosteum.
Results
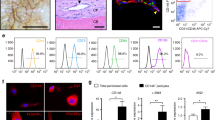
We showed that DDR2 predominantly labeled osteogenic cells residing in the inner layer of the periosteum and that Pearson’s coefficient of colocalization indicated a significant correlation with the expression of ALP. The mineralization of DDR2-expressing osteogenic cells isolated from the periosteum was significantly induced. In contrast, FSP-1 predominantly labeled the outer layer of periosteal fibroblasts, and Pearson’s coefficient of colocalization indicated that FSP-1 was poorly correlated with the expression of DDR2 and ALP. FSP-1-expressing periosteal fibroblasts did not exhibit osteogenic differentiation for the induction of bone mineralization.
Conclusion
DDR2 is a novel potential cell surface marker for identifying and isolating osteoblasts and osteoblast progenitors within the periosteum that can be used for musculoskeletal regenerative therapies.



Similar content being viewed by others
References
Allen MR, Hock JM, Burr DB (2004) Periosteum: biology, regulation, and response to osteoporosis therapies. Bone 35:1003–1012. https://doi.org/10.1016/j.bone.2004.07.014
Colnot C, Zhang X, Knothe Tate ML (2012) Current insights on the regenerative potential of the periosteum: molecular, cellular, and endogenous engineering approaches. J Orthop Res 30:1869–1878. https://doi.org/10.1002/jor.22181
Colnot C (2009) Skeletal cell fate decisions within periosteum and bone marrow during bone regeneration. J Bone Miner Res 24:274–282. https://doi.org/10.1359/jbmr.081003
Zhang X, Xie C, Lin AS, Ito H, Awad H, Lieberman JR, Rubery PT, Schwarz EM, O'Keefe RJ, Guldberg RE (2005) Periosteal progenitor cell fate in segmental cortical bone graft transplantations: implications for functional tissue engineering. J Bone Miner Res 20:2124–2137. https://doi.org/10.1359/JBMR.050806
Ozaki A, Tsunoda M, Kinoshita S, Saura R (2000) Role of fracture hematoma and periosteum during fracture healing in rats: interaction of fracture hematoma and the periosteum in the initial step of the healing process. J Orthop Sci 5:64–70
Ogita M, Rached MT, Dworakowski E, Bilezikian JP, Kousteni S (2008) Differentiation and proliferation of periosteal osteoblast progenitors are differentially regulated by estrogens and intermittent parathyroid hormone administration. Endocrinology 149:5713–5723. https://doi.org/10.1210/en.2008-0369
van Gastel N, Torrekens S, Roberts SJ, Moermans K, Schrooten J, Carmeliet P, Luttun A, Luyten FP, Carmeliet G (2012) Engineering vascularized bone: osteogenic and proangiogenic potential of murine periosteal cells. Stem Cells 30:2460–2471. https://doi.org/10.1002/stem.1210
Ferretti C, Borsari V, Falconi M, Gigante A, Lazzarini R, Fini M, Di Primio R, Mattioli-Belmonte M (2012) Human periosteum-derived stem cells for tissue engineering applications: the role of VEGF. Stem Cell Rev Rep 8:882–890. https://doi.org/10.1007/s12015-012-9374-7
Chang H, Knothe Tate ML (2012) Concise review: the periosteum: tapping into a reservoir of clinically useful progenitor cells. Stem Cells Transl Med 1:480–491. https://doi.org/10.5966/sctm.2011-0056
Arnsdorf EJ, Jones LM, Carter DR, Jacobs CR (2009) The periosteum as a cellular source for functional tissue engineering. Tissue Eng Part A 15:2637–2642. https://doi.org/10.1089/ten.TEA.2008.0244
Ng AM, Saim AB, Tan K, Tan GH, Mokhtar SA, Rose IM, Othman F, Idrus RBH (2005) Comparison of bioengineered human bone construct from four sources of osteogenic cells. J Orthop Sci 10:192–199. https://doi.org/10.1007/s00776-004-0884-2
Park J, Gelse K, Frank S, von der Mark K, Aigner T, Schneider H (2006) Transgene-activated mesenchymal cells for articular cartilage repair: a comparison of primary bone marrow-, perichondrium/periosteum- and fat-derived cells. J Gene Med 8:112–125. https://doi.org/10.1002/jgm.826
Vogel W (1999) Discoidin domain receptors: structural relations and functional implications. FASEB J 13(Suppl):S77–82. https://doi.org/10.1096/fasebj.13.9001.s77
Vogel W, Gish GD, Alves F, Pawson T (1997) The discoidin domain receptor tyrosine kinases are activated by collagen. Mol Cell 1:13–23. https://doi.org/10.1016/s1097-2765(00)80003-9
Ruiz PA, Jarai G (2011) Collagen I induces discoidin domain receptor (DDR) 1 expression through DDR2 and a JAK2-ERK1/2-mediated mechanism in primary human lung fibroblasts. J Biol Chem 286:12912–12923. https://doi.org/10.1074/jbc.M110.143693
Zhang K, Corsa CA, Ponik SM, Prior JL, Piwnica-Worms D, Eliceiri KW, Keely PJ, Longmore GD (2013) The collagen receptor discoidin domain receptor 2 stabilizes SNAIL1 to facilitate breast cancer metastasis. Nat Cell Biol 15:677–687. https://doi.org/10.1038/ncb2743
Bargal R, Cormier-Daire V, Ben-Neriah Z, Le Merrer M, Sosna J, Melki J, Zangen DH, Smithson SF, Borochowitz Z, Belostotsky R, Raas-Rothschild A (2009) Mutations in DDR2 gene cause SMED with short limbs and abnormal calcifications. Am J Hum Genet 84:80–84. https://doi.org/10.1016/j.ajhg.2008.12.004
Labrador JP, Azcoitia V, Tuckermann J, Lin C, Olaso E, Mañes S, Brückner K, Goergen JL, Lemke G, Yancopoulos G, Angel P, Martínez AC, Klein R (2001) The collagen receptor DDR2 regulates proliferation and its elimination leads to dwarfism. EMBO Rep 2:446–452. https://doi.org/10.1093/embo-reports/kve094
Ge C, Wang Z, Zhao G, Li B, Liao J, Sun H, Franceschi RT (2016) Discoidin receptor 2 controls bone formation and marrow adipogenesis. J Bone Miner Res 31:2193–2203. https://doi.org/10.1002/jbmr.2893
An essential role of discoidin domain receptor 2 (DDR2) in osteoblast differentiation and chondrocyte maturation via modulation of Runx2 activation—Zhang—2011—Journal of Bone and Mineral Research—Wiley Online Library. https://asbmr.onlinelibrary.wiley.com/doi/full/10.1002/jbmr.225. Accessed 17 Mar 2020
Zhang Y, Su J, Wu S, Teng Y, Yin Z, Guo Y, Li J, Li K, Yao L, Li X (2015) DDR2 (discoidin domain receptor 2) suppresses osteoclastogenesis and is a potential therapeutic target in osteoporosis. Sci Signal 8:ra31. https://doi.org/10.1126/scisignal.2005835
Bakker A, Klein-Nulend J (2003) Osteoblast isolation from murine calvariae and long bones. Methods Mol Med 80:19–28. https://doi.org/10.1385/1-59259-366-6:19
Jaiswal N, Haynesworth SE, Caplan AI, Bruder SP (1997) Osteogenic differentiation of purified, culture-expanded human mesenchymal stem cells in vitro. J Cell Biochem 64:295–312
Strutz F, Okada H, Lo CW, Danoff T, Carone RL, Tomaszewski JE, Neilson EG (1995) Identification and characterization of a fibroblast marker: FSP1. J Cell Biol 130:393–405. https://doi.org/10.1083/jcb.130.2.393
Beck TJ, Ruff CB, Scott WW, Plato CC, Tobin JD, Quan CA (1992) Sex differences in geometry of the femoral neck with aging: a structural analysis of bone mineral data. Calcif Tissue Int 50:24–29. https://doi.org/10.1007/bf00297293
Baudino TA, Carver W, Giles W, Borg TK (2006) Cardiac fibroblasts: friend or foe? Am J Physiol Heart Circ Physiol 291:H1015–1026. https://doi.org/10.1152/ajpheart.00023.2006
Acknowledgements
This work was supported by National Natural Science Foundation of China (NSFC#81670262), Guangzhou lingnan yingjie project and the start-up funding from Guangzhou Women and Children’s Medical Center and grants HL129178, HL137241, AR075867 from the National Institues of Health, and PR190268 and PR161247 from the Department of Defense, United states of America.
Author information
Authors and Affiliations
Contributions
HY performed most of the experiments and data analysis with LS, JG and DC, WQ, AD and JD designed the study and wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors report no conflict of interest.
Ethical approval
All animal studies were approved by the Institutional Animal Care and Use Committee of the Guangzhou Medical University (the acceptance number:2016-025) and all animal procedures conform to the National Institutes of Health (NIH) guidelines. This article does not contain any studies with human participants performed by any of the authors.
Informed consent
No informed consent needed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Yang, H., Sun, L., Cai, W. et al. DDR2, a discoidin domain receptor, is a marker of periosteal osteoblast and osteoblast progenitors. J Bone Miner Metab 38, 670–677 (2020). https://doi.org/10.1007/s00774-020-01108-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-020-01108-y