Abstract
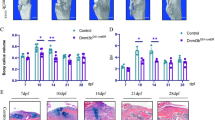
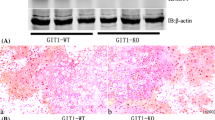
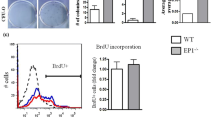
Bone fracture healing is achieved through the proliferation and differentiation of stem cells, while bone marrow stem cells (BMSCs) contribute to endochondral ossification. During fracture healing, mesenchymal progenitor cells first form a cartilaginous blastema that becomes vascularized to recruit precursor cells of osteoblasts through the bone morphogenetic protein 2 (Bmp2)/Smad-dependent Runx2 pathway. Statins deplete geranylgeranyl diphosphate (GGPP), which participates in the regulation of BMSCs differentiation, through the inhibition of 3-hydroxy-3-methylglutaryl (HMG)-CoA reductase, leading to impaired protein geranylgeranylation, which strongly impacts the bone synthesis induced by Bmp2. Accordingly, we would like to investigate the role of geranylgeranyl diphosphate synthase 1 (Ggps1) in bone fracture via endochondral ossification in mice. We used a Cre-loxP system, namely the tamoxifen-inducible Collagen 2-CreERT2 Ggps1 fl/fl, to eliminate specifically the Ggps1 activity in chondrocytes of 8–10-week-old mice. We found that the endochondral bone formation, calcification and vasculogenesis of the bony callus were accelerated in fractures in Ggps1−/−mice. Together, the results of this study confirm that the specific deletion of Ggps1, using the Collagen 2-CreERT2 mice, will accelerate the fracture healing process by activating the Bmp2/Smad-dependent Runx2 pathway. In addition, we managed to improve the fracture healing process by inhibiting the Ggps1 activity and its related products with statin drugs.








Similar content being viewed by others
References
Akiyama H, Chaboissier MC, Martin JF, Schedl A, de Crorugghe B (2002) The transcription factor Sox9 has essential roles in successive steps of the chondrocyte differentiation pathway and is required for expression of Sox5 and Sox6. Genes Dev 16:2813–2828. doi:10.1101/gad.1017802
Bi W, Deng JM, Zhang Z, Behringer RR, de Crombrugghe B (1999) Sox9 is required for cartilage formation. Nat Genet 22:85–89. doi:10.1038/8792
Bonnarens F, Einhorn TA (1984) Production of a standard closed fracture in laboratory animal bone. J Orthop Res 2:97–101. doi:10.1002/jor.1100020115
Bradley EW, McGee-Lawrence ME, Westendorf JJ (2011) Hdac-mediated control of endochondral and intramembranous ossification. Crit Rev Eukaryot Gene Expr 21:101–113
Chen M, Lichtler AC, Sheu TJ, Xie C, Zhang X, O’keefe RJ, Chen D (2007) Generation of a transgenic mouse model with chondrocyte-specific and tamoxifen-inducible expression of Cre recombinase. Genesis 45:44–50. doi:10.1002/dvg.20261
Chen P, Xia C, Mei S, Wang J, Shan Z, Lin X, Fan S (2016) Intra-articular delivery of sinomenium encapsulated by chitosan microspheres and photo-crosslinked GelMA hydrogel ameliorates osteoarthritis by effectively regulating autophagy. Biomaterials 81:1–13. doi:10.1016/j.biomaterials.2015.12.006
Dodge GR, Poole AR (1989) Immunohistochemical detection and immunochemical analysis of type II collagen degradation in human normal, rheumatoid, and osteoarthritic articular cartilages and in explants of bovine articular cartilage cultured with interleukin 1. J Clin Invest 83:647–661. doi:10.1172/jci113929
Einhorn TA (1995) Enhancement of fracture-healing. J Bone Jnt Surg Am 77:940–956
Fromigue O, Hay E, Modrowski D, Bouvet S, Jacquel A, Auberger P, Marie PJ (2006) RhoA GTPase inactivation by statins induces osteosarcoma cell apoptosis by inhibiting p42/p44-MAPKs-Bcl-2 signaling independently of BMP-2 and cell differentiation. Cell Death Differ 13:1845–1856. doi:10.1038/sj.cdd.4401873
Frommer J, Margolies MR (1971) Contribution of Meckel’s cartilage to ossification of the mandible in mice. J Dent Res 50:1260–1267
Fukui T, Ii M, Shoji T, Matsumoto T, Mifune Y, Kawakami Y, Akimaru H, Kawamoto A, Kuroda T, Saito T, Tabata Y, Kuroda R, Kurosaka M, Asahara T (2012) Therapeutic effect of local administration of low-dose simvastatin-conjugated gelatin hydrogel for fracture healing. J Bone Miner Res 27:1118–1131. doi:10.1002/jbmr.1558
Garrett IR, Mundy GR (2002) The role of statins as potential targets for bone formation. Arthritis Res 4:237–240. doi:10.1186/ar413
Hecht J, Seitz V, Urban M, Wagner F, Robinson PN, Stiege A, Dieterich C, Kornak U, Wilkening U, Brieske N, Zwingman C, Kidess A, Stricker S, Mundlos S (2007) Detection of novel skeletogenesis target genes by comprehensive analysis of a Runx2(-/-) mouse model. Gene Expr Patterns 7:102–112. doi:10.1016/j.modgep.2006.05.014
Ito T, Ikeda U, Yamamoto K, Shimada K (2002) Regulation of interleukin-8 expression by HMG-CoA reductase inhibitors in human vascular smooth muscle cells. Atherosclerosis 165:51–55
Jadhav SB, Jain GK (2006) Statins and osteoporosis: new role for old drugs. J Pharm Pharmacol 58:3–18. doi:10.1211/jpp.58.1.0002
Jia Z, Zhang Y, Chen YH, Dusad A, Yuan H, Ren K, Li F, Fehringer EV, Purdue PE, Goldring SR, Daluiski A, Wang D (2015) Simvastatin prodrug micelles target fracture and improve healing. J Control Release 200:23–34. doi:10.1016/j.jconrel.2014.12.028
Komori T, Yagi H, Nomura S, Yamaguchi A, Sasaki K, Deguchi K, Shimizu Y, Bronson RT, Gao YH, Inada M, Sato M, Okamoto R, Kitamura Y, Yoshiki S, Kishimoto T (1997) Targeted disruption of Cbfa1 results in a complete lack of bone formation owing to maturational arrest of osteoblasts. Cell 89:755–764
Kong RY, Kwan KM, Lau ET, Thomas JT, Boot-Handford RP, Grant ME, Cheah KS (1993) Intron-exon structure, alternative use of promoter and expression of the mouse collagen X gene, Col10a-1. Eur J Biochem 213:99–111
Kumar D, Lassar AB (2009) The transcriptional activity of Sox9 in chondrocytes is regulated by RhoA signaling and actin polymerization. Mol Cell Biol 29:4262–4273. doi:10.1128/MCB.01779-08
Li C, Ominsky MS, Tan HL, Barrero M, Niu QT, Asuncion FJ, Lee E, Liu M, Simonet WS, Paszty C, Ke HZ (2011) Increased callus mass and enhanced strength during fracture healing in mice lacking the sclerostin gene. Bone 49:1178–1185. doi:10.1016/j.bone.2011.08.012
Li F, Lu Y, Ding M, Napierala D, Abbassi S, Chen Y, Duan X, Wang S, Lee B, Zheng Q (2011) Runx2 contributes to murine Col10a1 gene regulation through direct interaction with its cis-enhancer. J Bone Miner Res 26:2899–2910. doi:10.1002/jbmr.504
Marsh D (1998) Concepts of fracture union, delayed union, and nonunion. Clin Orthop Relat Res 355:S22–S30
McTaggart SJ (2006) Isoprenylated proteins. Cell Mol Life Sci 63:255–267. doi:10.1007/s00018-005-5298-6
Mundy G, Garrett R, Harris S, Chan J, Chen D, Rossini G, Boyce B, Zhao M, Gutierrez G (1999) Stimulation of bone formation in vitro and in rodents by statins. Science 286:1946–1949
Mungo DV, Zhang X, O’Keefe RJ, Rosier RN, Puzas JE, Schwarz EM (2002) COX-1 and COX-2 expression in osteoid osteomas. J Orthop Res 20:159–162. doi:10.1016/s0736-0266(01)00065-1
Nagaoka Y, Kajiya H, Ozeki S, Ikebe T, Okabe K (2015) Mevalonates restore zoledronic acid-induced osteoclastogenesis inhibition. J Dent Res 94:594–601. doi:10.1177/0022034514564187
Niu YB, Kong XH, Li YH, Fan L, Pan YL, Li CR, Wu XL, Lu TL, Mei QB (2015) Radix Dipsaci total saponins stimulate MC3T3-E1 cell differentiation via the bone morphogenetic protein-2/MAPK/Smad-dependent Runx2 pathway. Mol Med Rep 11:4468–4472. doi:10.3892/mmr.2015.3249
Saklatvala J (1986) Tumour necrosis factor alpha stimulates resorption and inhibits synthesis of proteoglycan in cartilage. Nature 322:547–549. doi:10.1038/322547a0
Staal A, Frith JC, French MH, Swartz J, Gungor T, Harrity TW, Tamasi J, Rogers MJ, Feyen JH (2003) The ability of statins to inhibit bone resorption is directly related to their inhibitory effect on HMG-CoA reductase activity. J Bone Miner Res 18:88–96. doi:10.1359/jbmr.2003.18.1.88
Sverdrup FM, Yates MP, Vickery LE, Klover JA, Song LR, Anglin CP, Misko TP (2010) Protein geranylgeranylation controls collagenase expression in osteoarthritic cartilage. Osteoarthritis Cartilage 18:948–955. doi:10.1016/j.joca.2010.03.015
Tsubaki M, Yamazoe Y, Yanae M, Satou T, Itoh T, Kaneko J, Kidera Y, Moriyama K, Nishida S (2011) Blockade of the Ras/MEK/ERK and Ras/PI3K/Akt pathways by statins reduces the expression of bFGF, HGF, and TGF-beta as angiogenic factors in mouse osteosarcoma. Cytokine 54:100–107. doi:10.1016/j.cyto.2011.01.005
Tsuji K, Bandyopadhyay A, Harfe BD, Cox K, Kakar S, Gerstenfeld L, Einhorn T, Tabin CJ, Rosen V (2006) BMP2 activity, although dispensable for bone formation, is required for the initiation of fracture healing. Nat Genet 38:1424–1429. doi:10.1038/ng1916
Valcourt U, Gouttenoire J, Moustakas A, Herbage D, Mallein-Gerin F (2002) Functions of transforming growth factor-beta family type I receptors and Smad proteins in the hypertrophic maturation and osteoblastic differentiation of chondrocytes. J Biol Chem 277:33545–33558. doi:10.1074/jbc.M202086200
van der Kraan PM, Blaney Davidson EN, Blom A, van den Berg WB (2009) TGF-beta signaling in chondrocyte terminal differentiation and osteoarthritis: modulation and integration of signaling pathways through receptor-Smads. Osteoarthritis Cartilage 17:1539–1545. doi:10.1016/j.joca.2009.06.008
Wang G, Beier F (2005) Rac1/Cdc42 and RhoA GTPases antagonistically regulate chondrocyte proliferation, hypertrophy, and apoptosis. J Bone Miner Res 20:1022–1031. doi:10.1359/JBMR.050113
Wang G, Woods A, Agoston H, Ulici V, Glogauer M, Beier F (2007) Genetic ablation of Rac1 in cartilage results in chondrodysplasia. Dev Biol 306:612–623. doi:10.1016/j.ydbio.2007.03.520
Woods A, Wang G, Beier F (2005) RhoA/ROCK signaling regulates Sox9 expression and actin organization during chondrogenesis. J Biol Chem 280:11626–11634. doi:10.1074/jbc.M409158200
Woods A, Wang G, Dupuis H, Shao Z, Beier F (2007) Rac1 signaling stimulates N-cadherin expression, mesenchymal condensation, and chondrogenesis. J Biol Chem 282:23500–23508. doi:10.1074/jbc.M700680200
Wu D, Peng F, Zhang B, Ingram AJ, Kelly DJ, Gilbert RE, Gao B, Krepinsky JC (2009) PKC-beta1 mediates glucose-induced Akt activation and TGF-beta1 upregulation in mesangial cells. J Am Soc Nephrol 20:554–566. doi:10.1681/asn.2008040445
Yuasa M, Mignemi NA, Nyman JS, Duvall CL, Schwartz HS, Okawa A, Yoshii T, Bhattacharjee G, Zhao C, Bible JE, Obremskey WT, Flick MJ, Degen JL, Barnett JV, Cates JM, Schoenecker JG (2015) Fibrinolysis is essential for fracture repair and prevention of heterotopic ossification. J Clin Invest 125:3723. doi:10.1172/jci84059
Yuasa M, Mignemi NA, Nyman JS, Duvall CL, Schwartz HS, Okawa A, Yoshii T, Bhattacharjee G, Zhao C, Bible JE, Obremskey WT, Flick MJ, Degen JL, Barnett JV, Cates JM, Schoenecker JG (2015) Fibrinolysis is essential for fracture repair and prevention of heterotopic ossification. J Clin Invest 125:3117–3131. doi:10.1172/jci80313
Zehentner BK, Dony C, Burtscher H (1999) The transcription factor Sox9 is involved in BMP-2 signaling. J Bone Miner Res 14:1734–1741. doi:10.1359/jbmr.1999.14.10.1734
Zhang Y, Bradley AD, Wang D, Reinhardt RA (2014) Statins, bone metabolism and treatment of bone catabolic diseases. Pharmacol Res 88:53–61. doi:10.1016/j.phrs.2013.12.009
Acknowledgements
This work was supported by the project of the National Natural Science Foundation of China (81271945) and the National Natural Science Foundation of China (81420108021).
Author contributions
BD, CL, HT, and QJ conceived and designed the study. XS, CW, QL, and YG collected the data. KZ and JW performed the experiments. DC, YZ, and ZX analyzed the data. BD and QL wrote the paper. CL and QJ reviewed and edited the manuscript. None of the authors have conflict of interest.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None of the authors have conflict of interest.
Additional information
B. Dai and Q. Li contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
About this article
Cite this article
Dai, B., Li, Q., Song, X. et al. Knockdown of Ggps1 in chondrocyte expedites fracture healing by accelerating the progression of endochondral ossification in mice. J Bone Miner Metab 36, 133–147 (2018). https://doi.org/10.1007/s00774-017-0824-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00774-017-0824-9