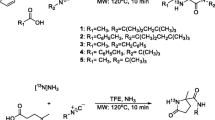
Abstract
Carbon-11 (β+ emitter, t 1/2 = 20.4 min) radiolabeled l-glutamine is a potentially useful molecular imaging agent that can be utilized with positron emission tomography for both human oncological diagnosis and plant imaging research. Based upon a previously reported [11C]cyanide end-capping labeling method, a systematic investigation of nucleophilic cyanation reactions and acidic hydrolysis reaction parameters, including base, metal ion source, phase transfer catalyst, solvent, reaction temperature and reaction time, was conducted. The result was a milder, more reliable, two-step method which provides l-[5-11C]-glutamine with a radiochemical yield of 63.8 ± 8.7 % (range from 51 to 74 %, n = 10) with >90 % radiochemical purity and >90 % enantiomeric purity. The total synthesis time was 40–50 min from the end of bombardment. In addition, an Fmoc derivatization method was developed to measure the specific activity of this radiotracer.





Similar content being viewed by others
References
Ametamey SM, Honer M, Schubiger PA (2008) Molecular imaging with PET. Chem Rev (Washington, DC, United States) 108:1501–1516. doi:10.1021/cr0782426
Bourdier T et al (2011) Radiosynthesis and biological evaluation of l- and d-S-(3-[18F]Fluoropropyl)homocysteine for tumor imaging using positron emission tomography. J Med Chem 54:1860–1870. doi:10.1021/jm101513q
Buehler J, Huber G, Schmid F, Bluemler P (2011) Analytical model for long-distance tracer-transport in plants. J Theor Biol 270:70–79. doi:10.1016/j.jtbi.2010.11.005
Charrier J-D, Hadfield DS, Hitchcock PB, Young DW (2004) Synthesis of (2S,4S)- and (2S,4R)-5-fluoroleucine and (2S,4S)-[5,5-2H2]-5-fluoroleucine. Org Biomol Chem 2:474–482. doi:10.1039/b314933a
Ding YS, Antoni G, Fowler JS, Wolf AP, Langstrom B (1989) Synthesis of l-[5-11C]ornithine. J Labelled Compd Radiopharm 27:1079–1090. doi:10.1002/jlcr.2580270913
Ermert J, Coenen HH (2013) Methods for 11C- and 18F-labeling of amino acids and derivatives for positron emission tomography imaging. J Labelled Compd Radiopharm 56:225–236. doi:10.1002/jlcr.2996
Fowler JS, Wolf AP (1997) Working against time: rapid radiotracer synthesis and imaging the human brain. Acc Chem Res 30:181–188. doi:10.1021/ar960068c
Gelbard AS, Christie TR, Clarke LP, Laughlin JS (1977) Imaging of spontaneous canine tumours with ammonia and l-glutamine labeled with N-13. J Nucl Med Off Publ Soc Nucl Med 18:718–723
Gillings NM, Gee AD (2001) Synthesis of [4-11C]amino acids via ring-opening of aziridine-2-carboxylates. J Labelled Compd Radiopharm 44:909–920. doi:10.1002/jlcr.517
Kim DW et al (2006) A new class of SN2 reactions catalyzed by protic solvents: facile fluorination for isotopic labeling of diagnostic molecules. J Am Chem Soc 128:16394–16397. doi:10.1021/ja0646895
Kim D, Alexoff D, Kim SW, Hooker J, Ferrieri RA (2013) 11C-Labeled cyanide production system. Application: US. US patent 2012-13584033. 20130045151
Kiser MR, Reid CD, Crowell AS, Phillips RP, Howell CR (2008) Exploring the transport of plant metabolites using positron emitting radiotracers. HFSP J 2:189–204. doi:10.2976/1.2921207
Lapi SE, Welch MJ (2013) A historical perspective on the specific activity of radiopharmaceuticals: what have we learned in the 35 years of the ISRC. Nucl Med Biol 40:314–320. doi:10.1016/j.nucmedbio.2012.12.010
Lea PJ, Sodek L, Parry MAJ, Shewry PR, Halford NG (2007) Asparagine in plants. Annals Applied Biol 150:1–26. doi:10.1111/j.1744-7348.2006.00104.x
Lee JW et al (2009) Bis-terminal hydroxy polyethers as all-purpose, multifunctional organic promoters: a mechanistic investigation and applications. Angew Chem Int Ed Engl 48:7683–7686. doi:10.1002/anie.200903903
Miller PW, Long NJ, Vilar R, Gee AD (2008) Synthesis of 11C, 18F, 15O, and 13N radiolabels for positron emission tomography. Angew Chem Int Ed 47:8998–9033. doi:10.1002/anie.200800222
Moorthy JN, Singhal N (2005) Facile and highly selective conversion of nitriles to amides via indirect acid-catalyzed hydration using TFA or AcOH-H2SO4. J Org Chem 70:1926–1929. doi:10.1021/jo048240a
Qu W et al (2011) Synthesis of optically pure 4-Fluoro-Glutamines as potential metabolic imaging agents for tumors. J Am Chem Soc 133:1122–1133. doi:10.1021/ja109203d
Qu W et al (2012) Preparation and characterization of l-[5-11C]-glutamine for metabolic imaging of tumors. J Nucl Med 53:98–105. doi:10.2967/jnumed.111.093831
Sieciechowicz KA, Joy KW, Ireland RJ (1988) The metabolism of asparagine in plants. Phytochemistry 27:663–671. doi:10.1016/0031-9422(88)84071-8
Stitt M et al (2002) Steps towards an integrated view of nitrogen metabolism. J Exp Bot 53:959–970. doi:10.1093/jexbot/53.370.959
Vallabhajosula S (2009) Molecular imaging: radiopharmaceuticals for PET and SPECT. Springer, Heidelberg
Zhou W, Zhang X-Y, Duan G-L (2011) Liquid-chromatography quantitative analysis of 20 amino acids after derivatization with FMOC-Cl and its application to different origin Radix isatidis. J Chin Chem Soc (Taipei, Taiwan) 58:509–515. doi:10.1002/jccs.201190014
Acknowledgments
This manuscript has been co-authored by employees of Brookhaven Science Associates, LLC under Contract No. DE-AC02-98CH10886 with the U.S. Department of Energy, Office of Biological and Environmental Research within the Office of Science. Additional support was provided by the German Academic Exchange Service (Deutscher Akademischer Austauschdienst, DAAD), Bonn, which supported Tassilo Gleede, Barbara Riehl, Lena Kersting, and Aylin Sibel Cankaya. The US Government retains and the publisher, by accepting the article for publication, acknowledges that the USA retains a non-exclusive, paid-up irrevocable, world-wide license to publish or reproduce the published form of this manuscript, or allow others to do so, for US Government purpose.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gleede, T., Riehl, B., Shea, C. et al. Investigation of SN2 [11C]cyanation for base-sensitive substrates: an improved radiosynthesis of l-[5-11C]-glutamine. Amino Acids 47, 525–533 (2015). https://doi.org/10.1007/s00726-014-1883-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-014-1883-z