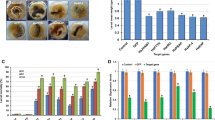
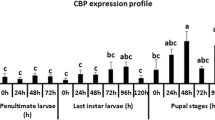
Abstract
GRIM-19 (genes associated with retinoid-IFN-induced mortality-19) is a subunit of mitochondrial respiratory complex I in mammalian systems. However, its function in vivo is not really understood. We cloned GRIM-19 and explored its function and hormonal regulation in insect, the cotton bollworm, Helicoverpa armigera. The results showed that Ha-GRIM-19 was highly expressed during the larval stage. Its transcript levels could be upregulated by juvenile hormone (JH) analog methoprene or by methoprene plus 20E. The methoprene-upregulated transcription enhancement of Ha-GRIM-19 was mediated by the transcription factor Ha-Met1, the putative receptor of JH. Other transcription factors Ha-USP1 and Ha-Br-Z2 suppressed the action of methoprene in inducing Ha-GRIM-19 expression, but Ha-Br-Z2 introduced interaction between 20E and methoprene in upregulation of Ha-GRIM-19. The knockdown of Ha-GRIM-19 by RNA interference in larvae and in insect cell line induced programmed cell death. These data imply that Ha-GRIM-19 plays role in keeping the normal cellular growth and it is able to be upregulated by methoprene through putative JH receptor Met.






Similar content being viewed by others
References
Altincicek B, Vilcinskas A (2008) Identification of a lepidopteran matrix metalloproteinase with dual roles in metamorphosis and innate immunity. Dev Comp Immunol 32(4):400–409
Angell JE, Lindner DJ, Shapiro PS, Hofmann ER, Kalvakolanu DV (2000) Identification of grim-19, a novel cell death-regulatory gene induced by the interferon-beta and retinoic acid combination, using a genetic approach. J Biol Chem 275(43):33416–33426
Bayer CA, Holley B, Fristrom JW (1996) A switch in broad-complex zinc-finger isoform expression is regulated posttranscriptionally during the metamorphosis of Drosophila imaginal discs. Dev Biol 177(1):1–14
Cohen GM (1997) Caspases: the executioners of apoptosis. Biochem J 326(Pt 1):1–16
Deveraux QL, Reed JC (1999) IAP family proteins-suppressors of apoptosis. Genes Dev 13(3):239–252
DiBello PR, Withers DA, Bayer CA, Fristrom JW, Guild GM (1991) The Drosophila broad-complex encodes a family of related proteins containing zinc fingers. Genetics 129(2):385–397
Dubrovsky EB (2005) Hormonal cross talk in insect development. Trends Endocrinol Metab 16(1):6–11
Fearnley IM, Carroll J, Shannon RJ, Runswick MJ, Walker JE, Hirst J (2001) GRIM-19, a cell death regulatory gene product, is a subunit of bovine mitochondrial NADH:ubiquinone oxidoreductase (complex I). J Biol Chem 276(42):38345–38348
Hatefi Y (1985) The mitochondrial electron transport and oxidative phosphorylation system. Annu Rev Biochem 54:1015–1069
Huang G, Lu H, Hao A, Ng DC, Ponniah S, Guo K, Lufei C, Zeng Q, Cao X (2004) GRIM-19, a cell death regulatory protein, is essential for assembly and function of mitochondrial complex I. Mol Cell Biol 24(19):8447–8456
Huang G, Chen Y, Lu H, Cao X (2007) Coupling mitochondrial respiratory chain to cell death: an essential role of mitochondrial complex I in the interferon-beta and retinoic acid-induced cancer cell death. Cell Death Differ 14(2):327–337
Jones G, Sharp PA (1997) Ultraspiracle: an invertebrate nuclear receptor for juvenile hormones. Proc Natl Acad Sci USA 94(25):13499–13503
Jones G, Jones D, Teal P, Sapa A, Wozniak M (2006) The retinoid-X receptor ortholog, ultraspiracle, binds with nanomolar affinity to an endogenous morphogenetic ligand. FEBS J 273(21):4983–4996
Knorr E, Schmidtberg H, Vilcinskas A, Altincicek B (2009) MMPs regulate both development and immunity in the Tribolium model insect. PLoS One 4(3):e4751
Konopova B, Jindra M (2008) Broad-complex acts downstream of Met in juvenile hormone signaling to coordinate primitive holometabolan metamorphosis. Development 135(3):559–568
Lawen A (2003) Apoptosis—an introduction. Bioessays 25(9):888–896
Lindner DJ, Borden EC, Kalvakolanu DV (1997) Synergistic antitumor effects of a combination of interferons and retinoic acid on human tumor cells in vitro and in vivo. Clin Cancer Res 3(6):931–937
Lockshin RA, Zakeri Z (2001) Programmed cell death and apoptosis: origins of the theory. Nat Rev Mol Cell Biol 2(7):545–550
Lu H, Cao X (2008) GRIM-19 is essential for maintenance of mitochondrial membrane potential. Mol Biol Cell 19(5):1893–1902
Maximo V, Lima J, Soares P, Silva A, Bento I, Sobrinho-Simoes M (2008) GRIM-19 in health and disease. Adv Anat Pathol 15(1):46–53
Miura K, Oda M, Makita S, Chinzei Y (2005) Characterization of the drosophila methoprene -tolerant gene product. Juvenile hormone binding and ligand-dependent gene regulation. FEBS J 272(5):1169–1178
Nakamura A, Stiebler R, Fantappie MR, Fialho E, Masuda H, Oliveira MF (2007) Effects of retinoids and juvenoids on moult and on phenoloxidase activity in the blood-sucking insect Rhodnius prolixus. Acta Trop 103(3):222–230
Newmeyer DD, Ferguson-Miller S (2003) Mitochondria: releasing power for life and unleashing the machineries of death. Cell 112(4):481–490
Nishiura JT, Ray K, Murray J (2005) Expression of nuclear receptor-transcription factor genes during Aedes aegypti midgut metamorphosis and the effect of methoprene on expression. Insect Biochem Mol Biol 35(6):561–573
Page-McCaw A, Serano J, Sante JM, Rubin GM (2003) Drosophila matrix metalloproteinases are required for tissue remodeling, but not embryonic development. Dev Cell 4(1):95–106
Parthasarathy R, Palli SR (2007) Developmental and hormonal regulation of midgut remodeling in a lepidopteran insect, Heliothis virescens. Mech Dev 124(1):23–34
Riddiford LM (2008) Juvenile hormone action: a 2007 perspective. J Insect Physiol 54(6):895–901
Riddiford LM, Hiruma K, Zhou X, Nelson CA (2003) Insights into the molecular basis of the hormonal control of molting and metamorphosis from Manduca sexta and Drosophila melanogaster. Insect Biochem Mol Biol 33(12):1327–1338
Shao HL, Zheng WW, Liu PC, Wang Q, Wang JX, Zhao XF (2008) Establishment of a new cell line from lepidopteran epidermis and hormonal regulation on the genes. PLoS One 3(9):e3127
Staal GB (1975) Insect growth regulators with juvenile hormone activity. Annu Rev Entomol 20:417–460
Thummel CS, Chory J (2002) Steroid signaling in plants and insects—common themes, different pathways. Genes Dev 16(24):3113–3129
Tian H, Peng H, Yao Q, Chen H, Xie Q, Tang B, Zhang W (2009) Developmental control of a lepidopteran pest Spodoptera exigua by ingestion of bacteria expressing dsRNA of a non-midgut gene. PLoS One 4(7):e6225
Timmons L, Court DL, Fire A (2001) Ingestion of bacterially expressed dsRNAs can produce specific and potent genetic interference in Caenorhabditis elegans. Gene 263(1–2):103–112
Wheeler DE, Nijhout HF (2003) A perspective for understanding the modes of juvenile hormone action as a lipid signaling system. Bioessays 25(10):994–1001
Wigglesworth V (1972) The principles of insect physiology. Chapman Hall, London, pp 476–552
Wilson TG (1996) Genetic evidence that mutants of the methoprene-tolerant gene of Drosophila melanogaster are null mutants. Arch Insect Biochem Physiol 32(3–4):641–649
Yang D, Chai L, Wang J, Zhao X (2008) Molecular cloning and characterization of Hearm caspase-1 from Helicoverpa armigera. Mol Biol Rep 35(3):405–412
Zamzami N, Marchetti P, Castedo M, Zanin C, Vayssiere JL, Petit PX, Kroemer G (1995) Reduction in mitochondrial potential constitutes an early irreversible step of programmed lymphocyte death in vivo. J Exp Med 181(5):1661–1672
Zhao XF, Wang JX, Xu XL, Schmid R, Wieczorek H (2002) Molecular cloning and characterization of the cathepsin B-like proteinase from the cotton boll worm, Helicoverpa armigera. Insect Mol Biol 11(6):567–575
Zhao XF, An XM, Wang JX, Dong DJ, Du XJ, Sueda S, Kondo H (2005) Expression of the Helicoverpa cathepsin B-like proteinase during embryonic development. Arch Insect Biochem Physiol 58(1):39–46
Zheng WW, Yang DT, Wang JX, Song QS, Gilbert LI, Zhao XF (2009) Hsc70 binds to ultraspiracle resulting in the upregulation of 20-hydroxyecdsone-responsive genes in Helicoverpa armigera. Mol Cell Endocrinol 315(1–2):282–291
Zhou X, Riddiford LM (2002) Broad specifies pupal development and mediates the ‘status quo’ action of juvenile hormone on the pupal-adult transformation in Drosophila and Manduca. Development 129(9):2259–2269
Acknowledgments
This work was supported by grants from the National Natural Science Foundation of China (Nos. 30900148 and 30710103901), The National Science Foundation for Post-doctoral Scientists of China (No. 20100471533) and The Shandong Province Postdoctoral Special Funds for Innovative Projects (No. 200802019). We thank Drs. Marek Jindra and Masako Asahina of the Biology Center, Czech Academy of Sciences and Department of Molecular Biology, University of South Bohemia, Czech Republic, for providing plasmid L4440 and HT115 (DE3).
Author information
Authors and Affiliations
Corresponding author
Additional information
The nucleotide sequence reported in this paper has been submitted to GenBank with accession number: HM369463.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dong, DJ., Liu, PC., Wang, JX. et al. The knockdown of Ha-GRIM-19 by RNA interference induced programmed cell death. Amino Acids 42, 1297–1307 (2012). https://doi.org/10.1007/s00726-010-0824-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-010-0824-8