Abstract
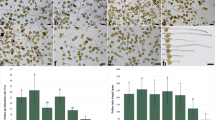
The aim of the current study was to examine the effect of different exogenous putrescine concentrations (200, 400, 600, and 800 μM) on the tea pollen performance. It was shown that putrescine has a dose-dependent effect on pollen performance. Results exhibited that pollen germination and tube elongation were induced by 200 and 400 μM putrescine treatment, especially, 400 μM putrescine–enhanced pollen performance. However, pollen performance was inhibited by higher concentrations of putrescine. Putrescine concentrations above 400 μM changed the actin filament distribution in pollen tubes by affecting the distribution of sucrose synthase enzyme. Alterations of the distribution on sucrose synthase enzyme also caused the alterations in the dispersion of cellulose and callose in the cell wall, and morphological alterations such as balloon-shaped and snake-shaped pollen tube tip accompanied them. Moreover, putrescine concentrations above 400 μM caused a decrease of ROS level in apex and led to chromatin condensation of the generative nucleus. In conclusion, exogenous putrescine application can be used as a pollen performance enhancer at low concentrations while the high concentrations cause adverse effects reducing fertilization success.







Similar content being viewed by others

References
Aloisi I, Cai G, Tumiatti V, Minarini A, Del Duca S (2015) Natural polyamines and synthetic analogs modify the growth and the morphology of Pyrus communis pollen tubes affecting ROS levels and causing cell death. Plant Sci 239:92–105. https://doi.org/10.1016/j.plantsci.2015.07.008
Aloisi I, Cai G, Serafini-Fracassini D, Del Duca S (2016) Polyamines in pollen: from microsporogenesis to fertilization. Front Plant Sci 7:155. https://doi.org/10.3389/fpls.2016.00155
Aloisi I, Cai G, Faleri C, Navazio L, Serafini-Fracassini D, Del Duca S (2017) Spermine regulates pollen tube growth by modulating Ca2+-dependent actin organization and cell wall structure. Front Plant Sci 8:1701. https://doi.org/10.3389/fpls.2017.01701
Anderhag P, Hepler PK, Lazzaro MD (2000) Microtubules and microfilaments are both responsible for pollen tube elongation in the conifer Picea abies (Norway spruce). Protoplasma 214(3–4):141–157. https://doi.org/10.1007/BF01279059
Bashline L, Lei L, Li S, Gu Y (2014) Cell wall, cytoskeleton, and cell expansion in higher plants. Mol Plant 7(4):586–600. https://doi.org/10.1093/mp/ssu018
Bhattacharjee S (2012) An inductive pulse of hydrogen peroxide pretreatment restores redox- homeostasis and mitigates oxidative membrane damage under extremes of temperature in two rice cultivars (Oryza sativa L., cultivars Ratna and SR 26B). Plant Growth Regul 68:395–410. https://doi.org/10.1007/s10725-012-9728-9
Biagini G, Faleri C, Cresti M, Cai G (2014) Sucrose concentration in the growth medium affects the cell wall composition of tobacco pollen tubes. Plant Reprod 27(3):129–144. https://doi.org/10.1007/s00497-014-0246-y
Boudaoud A, Burian A, Borowska-Wykręt D, Uyttewaal M, Wrzalik R, Kwiatkowska D, Hamant O (2014) FibrilTool, an ImageJ plug-in to quantify fibrillar structures in raw microscopy images. Nat Protoc 9(2):457–463. https://doi.org/10.1038/nprot.2014.024
Brewbaker JL, Kwack BH (1963) The essential role of calcium ion in pollen germination and pollen tube growth. Am J Bot 50(9):859–865. https://doi.org/10.1002/j.1537-2197.1963.tb06564.x
Cai G, Cresti M (2010) Microtubule motors and pollen tube growth—still an open question. Protoplasma 247(3–4):131–143. https://doi.org/10.1007/s00709-010-0214-9
Cai G, Faleri C, Del Casino C, Emons AMC, Cresti M (2011) Distribution of callose synthase, cellulose synthase, and sucrose synthase in tobacco pollen tube is controlled in dissimilar ways by actin filaments and microtubules. Plant Physiol 155(3):1169–1190. https://doi.org/10.1104/pp.110.171371
Cai G, Della Mea M, Faleri C, Fattorini L, Aloisi I, Serafini-Fracassini D, Del Duca S (2015a) Spermine either delays or promotes cell death in Nicotiana tabacum L. corolla depending on the floral developmental stage and affects the distribution of transglutaminase. Plant Sci 241:11–22. https://doi.org/10.1016/j.plantsci.2015.09.023
Cai G, Parrotta L, Cresti M (2015b) Organelle trafficking, the cytoskeleton, and pollen tube growth. J Integr Plant Biol 57(1):63–78. https://doi.org/10.1111/jipb.12289
Cameron C, Geitmann A (2018) Cell mechanics of pollen tube growth. Curr Opin Genet Dev 51:11–17. https://doi.org/10.1016/j.gde.2018.03.008
Cardenas L, Lovy-Wheeler A, Kunkel JG, Hepler PK (2008) Pollen tube growth oscillations and intracellular calcium levels are reversibly modulated by actin polymerization. Plant Physiol 146(4):1611–1621. https://doi.org/10.1104/pp.107.113035
Çetin E, Yildirim C, Palavan-Ünsal N, Ünal M (2000) Effect of spermine and cyclohexylamine on in vitro pollen germination and tube growth in Helianthus annuus. Can J Plant Sci 80(2):241–245. https://doi.org/10.4141/P99-097
Chakraborty K, Bhattacharjee S, Pal TK, Bhattacharyya S (2015) Evaluation of in vitro antioxidant potential of tea (Camelia sinensis) leaves obtained from different heights of Darjeeling Hill, West Bengal. J Appl Pharm Sci 5(1):63–68. https://doi.org/10.7324/JAPS.2015.50112
Chang T, Fan C, Man Y, Zhou J, Jing Y (2015) Bisphenol A affects germination and tube growth in Picea meyeri pollen through modulating Ca2+ flux and disturbing actin-dependent vesicular trafficking during cell wall construction. Plant Physiol Biochem 94:216–224. https://doi.org/10.1016/j.plaphy.2015.06.010
Chen T, Teng N, Wu X, Wang Y, Tang W, Samaj J, Baluska F, Lin J (2007) Disruption of actin filaments by latrunculin B affects cell wall construction in Picea meyeri pollen tube by disturbing vesicle trafficking. Plant Cell Physiol 48(1):19–30. https://doi.org/10.1093/pcp/pcl036
Chen J, Wang P, de Graaf BH, Zhang H, Jiao H, Tang C, Zhang S, Wu J (2018) Phosphatidic acid counteracts S-RNase signaling in pollen by stabilizing the actin cytoskeleton. Plant Cell 30(5):1023–1039. https://doi.org/10.1105/tpc.18.00021
Cui Y, Ling Y, Zhou J, Li X (2015) Interference of the histone deacetylase inhibits pollen germination and pollen tube growth in Picea wilsonii mast. PLoS One 10(12):e0145661. https://doi.org/10.1371/journal.pone.0145661
Deveci A, Aksoy O, Al G (2017) Investigation of the effects of quizalofop-P-ethyl on pollen germination of Hyacinthus orientalis L. Caryologia 70(1):77–81. https://doi.org/10.1080/00087114.2016.1275931
Dixin C, Shaoling Z (2002) Effects of polyamines and polyamine synthesis inhibitor on in vitro pollen germination and tube growth in pears. Journal of Fruit Science 19(6):377–380
Fang K, Zhang W, Xing Y, Zhang Q, Yang L, Cao Q, Qin L (2016) Boron toxicity causes multiple effects on Malus domestica pollen tube growth. Front Plant Sci 7:208. https://doi.org/10.3389/fpls.2016.00208
Fang KF, Zhang Q, Yang R, Cao QQ, Qin L (2018) Cytochalasin B treatment of apple (Malus pumila Mill.) pollen tubes alters the cytoplasmic calcium gradient and causes major changes in the cell wall components. Russ J Plant Physiol 65(3):384–393. https://doi.org/10.1134/S1021443718030111
Franklin-Tong VE, Drobak BK, Allan AC, Watkins PA, Trewavas AJ (1996) Growth of pollen tubes of Papaver rhoeas is regulated by a slow-moving calcium wave propagated by inositol 1, 4, 5-trisphosphate. Plant Cell 8(8):1305–1321. https://doi.org/10.1105/tpc.8.8.1305
Fu Y, Wu G, Yang Z (2001) Rop GTPase–dependent dynamics of tip-localized F-actin controls tip growth in pollen tubes. J Cell Biol 152(5):1019–1032. https://doi.org/10.1083/jcb.152.5.1019
Gao Y, Zhou H, Chen J, Jiang X, Tao S, Wu J, Zhang S (2015) Mitochondrial dysfunction mediated by cytoplasmic acidification results in pollen tube growth cessation in Pyrus pyrifolia. Physiol Plant 153(4):603–615. https://doi.org/10.1111/ppl.12260
Hao H, Chen T, Fan L, Li R, Wang X (2013) 2, 6-dichlorobenzonitrile causes multiple effects on pollen tube growth beyond altering cellulose synthesis in Pinus bungeana Zucc. PLoS One 8(10):e76660. https://doi.org/10.1371/journal.pone.0076660
Hepler PK, Rounds CM, Winship LJ (2013) Control of cell wall extensibility during pollen tube growth. Mol Plant 6(4):998–1017. https://doi.org/10.1093/mp/sst103
Jiang X, Gao Y, Zhou H, Chen J, Wu J, Zhang S (2014) Apoplastic calmodulin promotes self-incompatibility pollen tube growth by enhancing calcium influx and reactive oxygen species concentration in Pyrus pyrifolia. Plant Cell Rep 33(2):255–263. https://doi.org/10.1007/s00299-013-1526-y
Kang S, Chen S, Dai S (2010) Proteomics characteristics of rice leaves in response to environmental factors. Front Biol 5(3):246–254. https://doi.org/10.1007/s11515-010-0027-4
Ketelaar T, Meijer HJ, Spiekerman M, Weide R, Govers F (2012) Effects of latrunculin B on the actin cytoskeleton and hyphal growth in Phytophthora infestans. Fungal Genet Biol 49(12):1014–1022. https://doi.org/10.1016/j.fgb.2012.09.008
Kusano T, Berberich T, Tateda C, Takahashi Y (2008) Polyamines: essential factors for growth and survival. Planta 228(3):367–381. https://doi.org/10.1007/s00425-008-0772-7
Latrasse D, Benhamed M, Bergounioux C, Raynaud C, Delarue M (2016) Plant programmed cell death from a chromatin point of view. J Exp Bot 67(20):5887–5900. https://doi.org/10.1093/jxb/erw329
Lazzaro MD, Donohue JM, Soodavar FM (2003) Disruption of cellulose synthesis by isoxaben causes tip swelling and disorganizes cortical microtubules in elongating conifer pollen tubes. Protoplasma 220(3–4):201–207. https://doi.org/10.1007/s00709-002-0042-7
Lee YJ, Szumlanski A, Nielsen E, Yang Z (2008) Rho-GTPase–dependent filamentous actin dynamics coordinate vesicle targeting and exocytosis during tip growth. J Cell Biol 181(7):1155–1168. https://doi.org/10.1083/jcb.200801086
Liu Z, Yuan YL, Liu SQ, Yu XN, Rao LQ (2006) Screening for high-temperature tolerant cotton genotypes by testing in vitro pollen germination, pollen tube growth and boll retention. J Integr Plant Biol 48(6):706–714. https://doi.org/10.1111/j.1744-7909.2006.00276.x
Lovy-Wheeler A, Wilsen KL, Baskin TI, Hepler PK (2005) Enhanced fixation reveals the apical cortical fringe of actin filaments as a consistent feature of the pollen tube. Planta 221(1):95–104. https://doi.org/10.1007/s00425-004-1423-2
Masson PH, Takahashi T, Angelini R (2017) Molecular mechanisms underlying polyamine functions in plants. Front Plant Sci 8:14. https://doi.org/10.3389/fpls.2017.00014
McKenna ST, Kunkel JG, Bosch M, Rounds CM, Vidali L, Winship LJ, Hepler PK (2009) Exocytosis precedes and predicts the increase in growth in oscillating pollen tubes. Plant Cell 21(10):3026–3040. https://doi.org/10.1105/tpc.109.069260
Pal M, Ivanovska B, Olah T, Tajti J, Hamow KA, Szalai G, Khalil R, Vankova R, Dobrev P, Misheva SP, Janda T (2019) Role of polyamines in plant growth regulation of Rht wheat mutants. Plant Physiol Biochem 137:189–202. https://doi.org/10.1016/j.plaphy.2019.02.013
Pan J, Wang W, Li D, Shu Z, Ye X, Chang P, Wang Y (2016) Gene expression profile indicates involvement of NO in Camellia sinensis pollen tube growth at low temperature. BMC Genomics 17(1):809. https://doi.org/10.1186/s12864-016-3158-4
Papini A, Mosti S, Milocani E, Tani G, Di Falco P, Brighigna L (2011) Megasporogenesis and programmed cell death in Tillandsia (Bromeliaceae). Protoplasma 248(4):651–662. https://doi.org/10.1007/s00709-010-0221-x
Parrotta L, Faleri C, Cresti M, Cai G (2016) Heat stress affects the cytoskeleton and the delivery of sucrose synthase in tobacco pollen tubes. Planta. 243(1):43–63. https://doi.org/10.1007/s00425-015-2394-1
Parrotta L, Faleri C, Guerriero G, Cai G (2019) Cold stress affects cell wall deposition and growth pattern in tobacco pollen tubes. Plant Sci 283:329–342. https://doi.org/10.1016/j.plantsci.2019.03.010
Pasqualini S, Cresti M, Del Casino C, Faleri C, Frenguelli G, Tedeschini E, Ederli L (2015) Roles for NO and ROS signalling in pollen germination and pollen-tube elongation in Cupressus arizonica. Biol Plant 59(4):735–744. https://doi.org/10.1007/s10535-015-0538-6
Persia D, Cai G, Del Casino C, Faleri C, Willemse MT, Cresti M (2008) Sucrose synthase is associated with the cell wall of tobacco pollen tubes. Plant Physiol 147(4):1603–1618. https://doi.org/10.1104/pp.108.115956
Potocky M, Jones MA, Bezvoda R, Smirnoff N, Zarsky V (2007) Reactive oxygen species produced by NADPH oxidase are involved in pollen tube growth. New Phytol 174(4):742–751. https://doi.org/10.1111/j.1469-8137.2007.02042.x
Qu X, Zhang H, Xie Y, Wang J, Chen N, Huang S (2013) Arabidopsis villins promote actin turnover at pollen tube tips and facilitate the construction of actin collars. Plant Cell 25(5):1803–1817. https://doi.org/10.1105/tpc.113.110940
Qu X, Jiang Y, Chang M, Liu X, Zhang R, Huang S (2015) Organization and regulation of the actin cytoskeleton in the pollen tube. Front Plant Sci 5:786. https://doi.org/10.3389/fpls.2014.00786
Rojas ER, Hotton S, Dumais J (2011) Chemically mediated mechanical expansion of the pollen tube cell wall. Biophys J 101(8):1844–1853. https://doi.org/10.1016/j.bpj.2011.08.016
Salnikov VV, Grimson MJ, Seagull RW, Haigler CH (2003) Localization of sucrose synthase and callose in freeze-substituted secondary-wall-stage cotton fibers. Protoplasma 221:175–184. https://doi.org/10.1007/s00709-002-0079-7
Sato S, Kamiyama M, Iwata T, Makita N, Furukawa H, Ikeda H (2006) Moderate increase of mean daily temperature adversely affects fruit set of Lycopersicon esculentum by disrupting specific physiological processes in male reproductive development. Ann Bot 97(5):731–738. https://doi.org/10.1093/aob/mcl037
Seifi HS, Shelp BJ (2019) Spermine differentially refines plant defense responses against biotic and abiotic stresses. Front Plant Sci 10:117. https://doi.org/10.3389/fpls.2019.00117
Sequera-Mutiozabal M, Antoniou C, Tiburcio AF, Alcázar R, Fotopoulos V (2017) Polyamines: emerging hubs promoting drought and salt stress tolerance in plants. Curr Mol Biol Rep 3(1):28–36. https://doi.org/10.1007/s40610-017-0052-z
Shi XG, Fu QY, Jin JH, Quan C (2017) Mummified Oligocene fruits of Schima (Theaceae) and their systematic and biogeographic implications. Sci Rep 7(1):4009. https://doi.org/10.1038/s41598-017-04349-6
Sivakumar S, Siva G, Sathish S, Kumar GP, Vigneswaran M, Vinoth S, Kumar T, Sathishkumar R, Jayabalan N (2019) Influence of exogenous polyamines and plant growth regulators on high frequency in vitro mass propagation of Gloriosa superba L. and its colchicine content. Biocatal Agric Biotechnol 18:101030. https://doi.org/10.1016/j.bcab.2019.101030
Song J, Nada K, Tachibana S (1999) Ameliorative effect of polyamines on the high temperature inhibition of in vitro pollen germination in tomato (Lycopersicon esculentum Mill.). Sci Hortic 80(3–4):203–212. https://doi.org/10.1016/S0304-4238(98)00254-4
Sorkheh K, Shiran B, Rouhi V, Khodambashi M, Wolukau JN, Ercisli S (2011) Response of in vitro pollen germination and pollen tube growth of almond (Prunus dulcis Mill.) to temperature, polyamines and polyamine synthesis inhibitor. Biochem Syst Ecol 39(4–6):749–757. https://doi.org/10.1016/j.bse.2011.06.015
Srinivasan A, Saxena N, Johansen C (1999) Cold tolerance during early reproductive growth of chickpea (Cicer arietinum L.): genetic variation in gamete development and function. Field Crop Res 60(3):209–222. https://doi.org/10.1016/S0378-4290(98)00126-9
Stein O, Granot D (2019) An overview of sucrose synthases in plants. Front Plant Sci 10:95. https://doi.org/10.3389/fpls.2019.00095
Swanson S, Gilroy S (2010) ROS in plant development. Physiol Plant 138(4):384–392. https://doi.org/10.1111/j.1399-3054.2009.01313.x
Ünal M, Vardar F, Aytürk Ö (2013) Callose in plant sexual reproduction. .In Current progress in biological research. IntechOpen. https://doi.org/10.5772/53001
Van Hemelryck M, Bernal R, Ispolatov Y, Dumais J (2018) Lily pollen tubes pulse according to a simple spatial oscillator. Sci Rep 8(1):12135. https://doi.org/10.1038/s41598-018-30635-y
Vidali L, McKenna ST, Hepler PK (2001) Actin polymerization is essential for pollen tube growth. Mol Biol Cell 12(8):2534–2545. https://doi.org/10.1091/mbc.12.8.2534
Wang CL, Wu J, Xu GH, Gao YB, Chen G, Wu JY, Wu HQ, Zhang SL (2010) S-RNase disrupts tip-localized reactive oxygen species and induces nuclear DNA degradation in incompatible pollen tubes of Pyrus pyrifolia. J Cell Sci 123(24):4301–4309. https://doi.org/10.1242/jcs.075077
Wang L, Lv X, Li H, Zhang M, Wang H, Jin B, Chen T (2013) Inhibition of apoplastic calmodulin impairs calcium homeostasis and cell wall modeling during Cedrus deodara pollen tube growth. PLoS One 8(2):e55411. https://doi.org/10.1371/journal.pone.0055411
Wang W, Sheng X, Shu Z, Li D, Pan J, Ye X, Chang P, Li X, Wang Y (2016) Combined cytological and transcriptomic analysis reveals a nitric oxide signaling pathway involved in cold-inhibited Camellia sinensis pollen tube growth. Front Plant Sci 7:456. https://doi.org/10.3389/fpls.2016.00456
Wolukau JN, Zhang S, Xu G, Chen D (2004) The effect of temperature, polyamines and polyamine synthesis inhibitor on in vitro pollen germination and pollen tube growth of Prunus mume. Sci Hortic 99(3–4):289–299. https://doi.org/10.1016/S0304-4238(03)00112-2
Wu J, Shang Z, Wu J, Jiang X, Moschou PN, Sun W, Roubelakis-Angelakis KA, Zhang S (2010) Spermidine oxidase-derived H2O2 regulates pollen plasma membrane hyperpolarization-activated Ca2+-permeable channels and pollen tube growth. Plant J 63(6):1042–1053. https://doi.org/10.1111/j.1365-313X.2010.04301.x
Yazici K, Goksu B (2017) Effects of kaolin (M-99-099) application on antioxidant and phenolic compounds in tea leaves (Camellia sinensis LO Kuntze). Biochem Genet 55(5–6):367–377. https://doi.org/10.1007/s10528-017-9805-x
Yu Z, Yang Z (2019) Understanding different regulatory mechanisms of proteinaceous and non-proteinaceous amino acid formation in tea (Camellia sinensis) provides new insights into the safe and effective alteration of tea flavor and function. Crit Rev Food Sci Nutr:1–15. https://doi.org/10.1080/10408398.2018.1552245
Zhan H, Nie X, Zhang T, Li S, Wang X, Du X, Tong W, Song W (2019) Melatonin: a small molecule but important for salt stress tolerance in plants. Int J Mol Sci 20(3):709. https://doi.org/10.3390/ijms20030709
Acknowledgement
I thank Rukiye Aydın for pollen material support. Also, I thank Prof Meral Ünal, Prof Filiz Vardar and Prof Giampiero Cai for the fruitful discussion.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The author declares that there is no conflict of interest.
Additional information
Handling Editor: Benedikt Kost
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Çetinbaş-Genç, A. Putrescine modifies the pollen tube growth of tea (Camellia sinensis) by affecting actin organization and cell wall structure. Protoplasma 257, 89–101 (2020). https://doi.org/10.1007/s00709-019-01422-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-019-01422-x