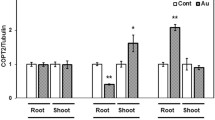
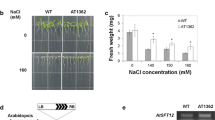
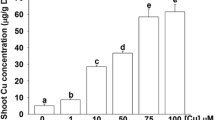
Abstract
Iron (Fe) is an essential micronutrient for humans. Fe deficiency disease is widespread and has led to extensive studies on the mechanisms of Fe uptake and storage, especially in staple food crops such as rice. However, studies of functionally related genes in rice and other crops are often time and space demanding. Here, we demonstrate that transgenic Arabidopsis suspension culture cells and Arabidopsis plants can be used as an efficient expression system for gain-of-function study of selected transporters, using Fe transporters as a proof-of-principle. The vacuolar membrane transporters OsVIT1 and OsVIT2 have been described to be important for iron sequestration, and disruption of these two genes leads to Fe accumulation in rice seeds. In this study, we have taken advantage of the fluorescent-tagged protein GFP-OsVIT1, which functionally complements the Fe hypersensitivity of ccc1 yeast mutant, to generate transgenic Arabidopsis suspension cell lines and plants. GFP-OsVIT1 was shown to localize on the vacuolar membrane using confocal microscopy and immunogold EM. More importantly, the Fe concentration, as well as the concentration of Zn, in the transgenic cell lines and plants were significantly increased compared to that in the WT. Taken together, our study shows that the heterologous expression of rice vacuolar membrane transporter OsVIT1 in Arabidopsis system is functional and effectively enhances iron accumulation, indicating an useful approach for studying other putative transporters of crop plants in this system.








Similar content being viewed by others
Reference
Bashir K, Ishimaru Y, Nishizawa N (2010) Iron Uptake and Loading into Rice Grains Rice 3:122-130 doi:10.1007/s12284-010-9042-y
Bashir K, Ishimaru Y, Nishizawa NK (2011a) Identification and characterization of the major mitochondrial Fe transporter in rice. Plant Signal Behav 6:1591–1593. doi:10.4161/psb.6.10.17132
Bashir K et al (2011b) The rice mitochondrial iron transporter is essential for plant growth. Nat Commun 2:322. doi:10.1038/ncomms1326
Bashir K, Nozoye T, Ishimaru Y, Nakanishi H, Nishizawa NK (2013a) Exploiting new tools for iron bio-fortification of rice. Biotechnol Adv 31:1624–1633. doi:10.1016/j.biotechadv.2013.08.012
Bashir K, Takahashi R, Akhtar S, Ishimaru Y, Nakanishi H, Nishizawa NK (2013b) The knockdown of OsVIT2 and MIT affects iron localization in rice seed. Rice 6:31. doi:10.1186/1939-8433-6-31
Briat JF (1999) Plant ferritin and human iron deficiency. Nat Biotechnol 17:621. doi:10.1038/10797
Clough SJ, Bent AF (1998) Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 16:735–743
Cui Y, Shen J, Gao C, Zhuang X, Wang J, Jiang L (2016) Biogenesis of Plant Prevacuolar Multivesicular Bodies Molecular plant doi:10.1016/j.molp.2016.01.011
Curie C, Briat JF (2003) Iron transport and signaling in plants. Annu Rev Plant Biol 54:183–206. doi:10.1146/annurev.arplant.54.031902.135018
Curie C, Panaviene Z, Loulergue C, Dellaporta SL, Briat JF, Walker EL (2001) Maize yellow stripe1 encodes a membrane protein directly involved in Fe(III) uptake. Nature 409:346–349. doi:10.1038/35053080
Duy D, Wanner G, Meda AR, von Wiren N, Soll J, Philippar K (2007) PIC1, an ancient permease in Arabidopsis chloroplasts, mediates iron transport. Plant Cell 19:986–1006. doi:10.1105/tpc.106.047407
Duy D, Stube R, Wanner G, Philippar K (2011) The chloroplast permease PIC1 regulates plant growth and development by directing homeostasis and transport of iron. Plant Physiol 155:1709–1722. doi:10.1104/pp.110.170233
Elble R (1992) A simple and efficient procedure for transformation of yeasts. BioTechniques 13:18–20
Garcia CB, Grusak MA (2015) Mineral accumulation in vegetative and reproductive tissues during seed development in Medicago truncatula. Front Plant Sci 6:622. doi:10.3389/fpls.2015.00622
Goto F, Yoshihara T, Shigemoto N, Toki S, Takaiwa F (1999) Iron fortification of rice seed by the soybean ferritin gene. Nat Biotechnol 17:282–286. doi:10.1038/7029
Harrison PM, Arosio P (1996) The ferritins: molecular properties, iron storage function and cellular regulation. Biochim Biophys Acta 1275:161–203
Ishimaru Y et al (2010) Rice metal-nicotianamine transporter, OsYSL2, is required for the long-distance transport of iron and manganese. Plant J 62:379–390. doi:10.1111/j.1365-313X.2010.04158.x
Ishimaru Y, Bashir K, Nakanishi H, Nishizawa NK (2012) OsNRAMP5, a major player for constitutive iron and manganese uptake in rice. Plant Signal Behav 7:763–766. doi:10.4161/psb.20510
Jiang L, Rogers JC (1998) Integral membrane protein sorting to vacuoles in plant cells: evidence for two pathways. J Cell Biol 143:1183–1199
Kim SA, Guerinot ML (2007) Mining iron: iron uptake and transport in plants. FEBS Lett 581:2273–2280. doi:10.1016/j.febslet.2007.04.043
Kim SA et al (2006) Localization of iron in Arabidopsis seed requires the vacuolar membrane transporter VIT1. Science 314:1295–1298. doi:10.1126/science.1132563
Koike S, Inoue H, Mizuno D, Takahashi M, Nakanishi H, Mori S, Nishizawa NK (2004) OsYSL2 is a rice metal-nicotianamine transporter that is regulated by iron and expressed in the phloem. Plant J 39:415–424. doi:10.1111/j.1365-313X.2004.02146.x
Lam SK, Siu CL, Hillmer S, Jang S, An G, Robinson DG, Jiang L (2007) Rice SCAMP1 defines clathrin-coated, trans-Golgi-located tubular-vesicular structures as an early Endosome in Tobacco BY-2 cells. Plant Cell 19:296–319. doi:10.1105/tpc.106.045708
Lam SK, Cai Y, Hillmer S, Robinson DG, Jiang L (2008) SCAMPs highlight the developing cell plate during cytokinesis in tobacco BY-2 cells. Plant Physiol 147:1637–1645. doi:10.1104/pp.108.119925
Lanquar V et al (2005) Mobilization of vacuolar iron by AtNRAMP3 and AtNRAMP4 is essential for seed germination on low iron. EMBO J 24:4041–4051. doi:10.1038/sj.emboj.7600864
Lee S, Chiecko JC, Kim SA, Walker EL, Lee Y, Guerinot ML, An G (2009a) Disruption of OsYSL15 leads to iron inefficiency in rice plants. Plant Physiol 150:786–800. doi:10.1104/pp.109.135418
Lee S et al (2009b) Iron fortification of rice seeds through activation of the nicotianamine synthase gene. Proc Natl Acad Sci U S A 106:22014–22019. doi:10.1073/pnas.0910950106
Li L, Chen OS, McVey Ward D, Kaplan J (2001) CCC1 is a transporter that mediates vacuolar iron storage in yeast. J Biol Chem 276:29515–29519. doi:10.1074/jbc.M103944200
Mhatre M, Srinivas L, Ganapathi TR (2011) Enhanced iron and zinc accumulation in genetically engineered pineapple plants using soybean ferritin gene. Biol Trace Elem Res 144:1219–1228. doi:10.1007/s12011-011-9092-z
Miao Y, Jiang L (2007a) Transient expression of fluorescent fusion proteins in protoplasts of suspension cultured cells. Nat Protoc 2:2348–2353. doi:10.1038/nprot.2007.360
Miao Y, Jiang L (2007b) Transient expression of fluorescent fusion proteins in protoplasts of suspension cultured cells. Nat Protocols 2:2348–2353
Momonoi K et al (2009) A vacuolar iron transporter in tulip, TgVit1, is responsible for blue coloration in petal cells through iron accumulation. Plant J 59:437–447. doi:10.1111/j.1365-313X.2009.03879.x
Narayanan N, Beyene G, Chauhan RD, Gaitan-Solis E, Grusak MA, Taylor N, Anderson P (2015) Overexpression of Arabidopsis VIT1 increases accumulation of iron in cassava roots and stems. Plant Sci 240:170–181. doi:10.1016/j.plantsci.2015.09.007
Roberts LA, Pierson AJ, Panaviene Z, Walker EL (2004) Yellow stripe1. Expanded roles for the maize iron-phytosiderophore transporter. Plant Physiol 135:112–120. doi:10.1104/pp.103.037572
Robinson NJ, Procter CM, Connolly EL, Guerinot ML (1999) A ferric-chelate reductase for iron uptake from soils. Nature 397:694–697. doi:10.1038/17800
Schroeder JI et al (2013) Using membrane transporters to improve crops for sustainable food production. Nature 497:60–66. doi:10.1038/nature11909
Shen J et al. (2014) Isolation, culture, and transient transformation of plant protoplasts Current protocols in cell biology / editorial board, Juan S Bonifacino [et al] 63:2 8 1-2 8 17 doi:10.1002/0471143030.cb0208s63
Tse YC, Mo B, Hillmer S, Zhao M, Lo SW, Robinson DG, Jiang L (2004) Identification of multivesicular bodies as prevacuolar compartments in Nicotiana tabacum BY-2 cells. Plant Cell 16:672–693. doi:10.1105/tpc.019703
Vert G, Grotz N, Dedaldechamp F, Gaymard F, Guerinot ML, Briat JF, Curie C (2002) IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell 14:1223–1233
Wang J et al (2010) EXPO, an exocyst-positive organelle distinct from multivesicular endosomes and autophagosomes, mediates cytosol to cell wall exocytosis in Arabidopsis and tobacco cells. Plant Cell 22:4009–4030. doi:10.1105/tpc.110.080697
Wang X, Cai Y, Wang H, Zeng Y, Zhuang X, Li B, Jiang L (2014) Trans-Golgi network-located AP1 gamma adaptins mediate dileucine motif-directed vacuolar targeting in Arabidopsis. Plant Cell 26:4102–4118. doi:10.1105/tpc.114.129759
Zeng Y et al (2015) Unique COPII component AtSar1a/AtSec23a pair is required for the distinct function of protein ER export in Arabidopsis thaliana. Proc Natl Acad Sci U S A 112:14360–14365. doi:10.1073/pnas.1519333112
Zhang Y, Xu YH, Yi HY, Gong JM (2012) Vacuolar membrane transporters OsVIT1 and OsVIT2 modulate iron translocation between flag leaves and seeds in rice. Plant J 72:400–410. doi:10.1111/j.1365-313X.2012.05088.x
Acknowledgments
This work was supported by grants from the Research Grants Council of Hong Kong (CUHK4666011, 46112, 466613, CUHK2/CRF/11G, C4011-14R, and AoE/M-05/12), NSFC/RGC (N_CUHK406/12), NSFC (31270226 and 31470294), and Shenzhen Peacock Project (KQTD201101) to L. Jiang. This work was also supported in part by funding from the US Department of Agriculture, Agricultural Research Service through Cooperative Agreement Number 58-6250-0-008 to M.A. Grusak. The contents of this publication do not necessarily reflect the views or policies of the US Department of Agriculture, nor does mention of trade names, commercial products, or organizations imply endorsement by the US Government.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Handling Editor: Peter Nick
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplement Figure S1
The concentration of macronutrients Ca, Mg and P in Arabidopsis cell lines (PDF 532 kb)
Supplement Figure S2
The concentration of Fe (A), Mn (B), Zn (C) and Cu (D) in Arabidopsis stem tissues (PDF 1529 kb)
Supplement Figure S3
The concentration of macronutrients Ca, Mg and P in Arabidopsis stem (A) and silique (B) tissues (PDF 1123 kb)
Supplement Figure S4
. The concentration of Fe (A), Mn (B) and Zn (C) in Arabidopsis stem tissue when supplemented with 50 μM Fe-EDTA (PDF 736 kb)
Supplement Figure S5
The concentration of Ca, Mg, P and Cu in Arabidopsis stem (A) and silique (B) tissues when supplemented with 50 μM Fe-EDTA (PDF 1.39 MB)
Rights and permissions
About this article
Cite this article
Wang, X., Zhong, F., Woo, C.H. et al. A rapid and efficient method to study the function of crop plant transporters in Arabidopsis . Protoplasma 254, 737–747 (2017). https://doi.org/10.1007/s00709-016-0987-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00709-016-0987-6