Summary.
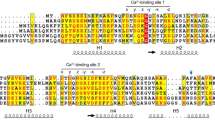
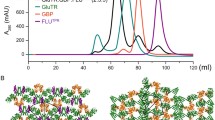
Abundant flavin binding sites have been found in membranes of plants and fungi. With flavin mononucleotide-agarose affinity columns, riboflavin-binding activity from microsomes of Cucurbita pepo L. hypocotyls was purified and identified as a specific PIP1-homologous protein of the aquaporin family. Sequences such as gi|2149955 in Phaseolus vulgaris, PIP1b of Arabidopsis thaliana, and NtAQP1 of tobacco are closely related. The identification as a riboflavin-binding protein was confirmed by binding tests with an extract of Escherichia coli cells expressing the tobacco NtAQP1 as well as leaves of transgenic tobacco plants that overexpress NtAQP1 or were inhibited in PIP1 expression by antisense constructs. When binding was assayed in the presence of dithionite, the reduced flavin formed a relatively stable association with the protein. Upon dilution under oxidizing conditions, the adduct was resolved, and free flavin reappeared with a half time of about 30 min. Such an association can also be induced photochemically, with oxidized flavin by blue light at 450 nm, in the presence of an electron donor. Several criteria, localization in the plasma membrane, high abundance, affinity to roseoflavin, and photochemistry, argue for a role of the riboflavin-binding protein PIP1 as a photoreceptor.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received May 10, 2002; accepted September 20, 2002; published online May 21, 2003
RID="*"
ID="*" Correspondence and reprints: Institut für Biologie III, Universität Freiburg, Schänzlestrasse 1, 79104 Freiburg im Breisgau, Federal Republic of Germany.
Rights and permissions
About this article
Cite this article
Lorenz, A., Kaldenhoff, R. & Hertel, R. A major integral protein of the plant plasma membrane binds flavin. Protoplasma 221, 19–30 (2003). https://doi.org/10.1007/s00709-002-0066-z
Issue Date:
DOI: https://doi.org/10.1007/s00709-002-0066-z