Abstract
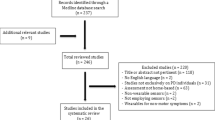
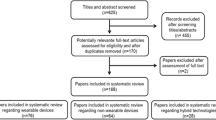
The potential of using wearable technologies for the objective assessment of motor symptoms in Parkinson’s disease (PD) has gained prominence recently. Nonetheless, compared to tremor and gait impairment, less emphasis has been placed on the quantification of bradykinesia and rigidity. This review aimed to consolidate the existing research on objective measurement of bradykinesia and rigidity in PD through the use of wearables, focusing on the continuous monitoring of these two symptoms in free-living environments. A search of PubMed was conducted through a combination of keyword and MeSH searches. We also searched the IEEE, Google Scholar, Embase, and Scopus databases to ensure thorough results and to minimize the chances of missing relevant studies. Papers published after the year 2000 with sample sizes greater than five were included. Studies were assessed for quality and information was extracted regarding the devices used and their location on the body, the setting and duration of the study, the “gold standard” used as a reference for validation, the metrics used, and the results of each paper. Thirty-one and eight studies met the search criteria and evaluated bradykinesia and rigidity, respectively. Several studies reported strong associations between wearable-based measures and the gold-standard references for bradykinesia, and, to a lesser extent, rigidity. Only a few, pilot studies investigated the measurement of bradykinesia and rigidity in the home and free-living settings. While the current results are promising for the future of wearables, additional work is needed on their validation and adaptation in ecological, free-living settings. Doing so has the potential to improve the assessment and treatment of motor fluctuations and symptoms of PD more generally through real-time objective monitoring of bradykinesia and rigidity.




Similar content being viewed by others
References
Ansari NN, Naghdi S, Younesian P, Shayeghan M (2008) Inter- and intrarater reliability of the Modified Modified Ashworth Scale in patients with knee extensor poststroke spasticity. Physiother Theory Pract 24:205–213. https://doi.org/10.1080/09593980701523802
Appleman ER, Stavitsky K, Cronin-Golomb A (2011) Relation of subjective quality of life to motor symptom profile in Parkinson’s disease. Parkinsons Dis 2011:472830. https://doi.org/10.4061/2011/472830
Assis S, Costa P, Rosas MJ, et al (2016) An adaptive model approach for quantitative wrist rigidity evaluation during deep brain stimulation surgery. In: 2016 38th annual international conference of the ieee engineering in medicine and biology society (EMBC). IEEE, pp 5809–5812
Berardelli A, Rothwell JC, Thompson PD, Hallett M (2001) Pathophysiology of bradykinesia in Parkinson’s disease. Brain 124:2131–2146
Bhidayasiri R, Tarsy D (2012) Parkinson’s disease: “on-off” phenomenon. Movement disorders: a video Atlas. Humana, Totowa, pp 14–15
Bhidayasiri R, Sringean J, Taechalertpaisarn P, Thanawattano C (2016a) Capturing nighttime symptoms in Parkinson disease: technical development and experimental verification of inertial sensors for nocturnal hypokinesia. J Rehabil Res Dev 53:487–498. https://doi.org/10.1682/JRRD.2015.04.0062
Bhidayasiri R, Sringean J, Thanawattano C (2016b) Sensor-based evaluation and treatment of nocturnal hypokinesia in Parkinson’s disease: an evidence-based review. Parkinsonism Relat Disord 22:S127–S133. https://doi.org/10.1016/J.PARKRELDIS.2015.09.049
Bhidayasiri R, Sringean J, Chaiwong S et al (2017a) Rotigotine for nocturnal hypokinesia in Parkinson’s disease: quantitative analysis of efficacy from a randomized, placebo-controlled trial using an axial inertial sensor. Parkinsonism Relat Disord 44:124–128. https://doi.org/10.1016/J.PARKRELDIS.2017.08.010
Bhidayasiri R, Sringean J, Thanawattano C (2017b) Impaired bed mobility: quantitative torque analysis with axial inertial sensors. Neurodegener Dis Manag 7:235–243. https://doi.org/10.2217/nmt-2017-0016
Bower K, Thilarajah S, Pua Y-H et al (2019) Dynamic balance and instrumented gait variables are independent predictors of falls following stroke. J Neuroeng Rehabil 16:3. https://doi.org/10.1186/s12984-018-0478-4
Cancela J, Pansera M, Arredondo MT et al (2010) A comprehensive motor symptom monitoring and management system: the bradykinesia case. Conf Proc Annu Int Conf IEEE Eng Med Biol Soc IEEE Eng Med Biol Soc Annu Conf 2010:1008–1011. https://doi.org/10.1109/IEMBS.2010.5627775
Costa P, Rosas MJ, Vaz R, Cunha JP (2015) Wrist rigidity assessment during Deep Brain Stimulation surgery. In: 2015 37th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC). IEEE, pp 3423–3426
Dai H, Lin H, Lueth TC (2015) Quantitative assessment of parkinsonian bradykinesia based on an inertial measurement unit. Biomed Eng Online 14:68. https://doi.org/10.1186/s12938-015-0067-8
Daneault J-F, Lee SI, Golabchi FN, et al (2017) Estimating Bradykinesia in Parkinson’s disease with a minimum number of wearable sensors. In: 2017 IEEE/ACM international conference on connected health: applications, systems and engineering technologies (CHASE). IEEE, pp 264–265
Delrobaei M, Tran S, Gilmore G et al (2016) Characterization of multi-joint upper limb movements in a single task to assess bradykinesia. J Neurol Sci 368:337–342. https://doi.org/10.1016/j.jns.2016.07.056
di Biase L, Summa S, Tosi J et al (2018) Quantitative analysis of bradykinesia and rigidity in Parkinson’s disease. Front Neurol 9:121. https://doi.org/10.3389/fneur.2018.00121
Dorsey ER, Glidden AM, Holloway MR et al (2018) Teleneurology and mobile technologies: the future of neurological care. Nat Rev Neurol 14:285–297. https://doi.org/10.1038/nrneurol.2018.31
Ellis T, Cavanaugh JT, Earhart GM et al (2011) Which measures of physical function and motor impairment best predict quality of life in Parkinson’s disease? Parkinsonism Relat Disord 17:693–697. https://doi.org/10.1016/j.parkreldis.2011.07.004
Endo T, Okuno R, Yokoe M et al (2009) A novel method for systematic analysis of rigidity in Parkinson’s disease. Mov Disord 24:2218–2224. https://doi.org/10.1002/mds.22752
Endo T, Yokoe M, Fujimura H, Sakoda S (2012) Novel methods to evaluate symptoms in Parkinson’s disease – rigidity and finger tapping. In: Dushanova J (ed) Diagnostics and rehabilitation of Parkinson’s disease. InTech, pp 191–206
Eskofier BM, Lee SI, Daneault J-F et al (2016) Recent machine learning advancements in sensor-based mobility analysis: deep learning for Parkinson’s disease assessment. Conf Proc Annu Int Conf IEEE Eng Med Biol Soc IEEE Eng Med Biol Soc Annu Conf 2016:655–658. https://doi.org/10.1109/EMBC.2016.7590787
Gao C, Smith S, Lones M et al (2018) Objective assessment of bradykinesia in Parkinson’s disease using evolutionary algorithms: clinical validation. Transl Neurodegener 7:18. https://doi.org/10.1186/s40035-018-0124-x
Griffiths RI, Kotschet K, Arfon S et al (2012) Automated assessment of bradykinesia and dyskinesia in Parkinson’s disease. J Parkinsons Dis 2:47–55. https://doi.org/10.3233/JPD-2012-11071
Hafsteinsdóttir TB, Rensink M, Schuurmans M (2014) Clinimetric properties of the timed up and go test for patients with stroke: a systematic review. Top Stroke Rehabil 21:197–210. https://doi.org/10.1310/tsr2103-197
Heldman DA, Giuffrida JP, Chen R et al (2011) The modified bradykinesia rating scale for Parkinson’s disease: reliability and comparison with kinematic measures. Mov Disord 26:1859–1863. https://doi.org/10.1002/mds.23740
Heldman DA, Filipkowski DE, Riley DE et al (2012) Automated motion sensor quantification of gait and lower extremity bradykinesia. Conf Proc Annu Int Conf IEEE Eng Med Biol Soc IEEE Eng Med Biol Soc Annu Conf 2012:1956–1959. https://doi.org/10.1109/EMBC.2012.6346338
Hillel I, Gazit E, Nieuwboer A et al (2019) Is every-day walking in older adults more analogous to dual-task walking or to usual walking? Elucidating the gaps between gait performance in the lab and during 24/7 monitoring. Eur Rev Aging Phys Act 16:6. https://doi.org/10.1186/s11556-019-0214-5
Hoff JI, van der Meer V, van Hilten JJ (2004) Accuracy of objective ambulatory accelerometry in detecting motor complications in patients with Parkinson disease. Clin Neuropharmacol 27:53–57
Iluz T, Weiss A, Gazit E et al (2016) Can a body-fixed sensor reduce heisenberg’s uncertainty when it comes to the evaluation of mobility? Effects of aging and fall risk on transitions in daily living. J Gerontol Ser A Biol Sci Med Sci 71:1459–1465. https://doi.org/10.1093/gerona/glv049
Johansson D, Malmgren K, Alt Murphy M (2018) Wearable sensors for clinical applications in epilepsy, Parkinson’s disease, and stroke: a mixed-methods systematic review. J Neurol 265:1740–1752. https://doi.org/10.1007/s00415-018-8786-y
Kassavetis P, Saifee TA, Roussos G et al (2016) Developing a tool for remote digital assessment of Parkinson’s disease. Mov Disord Clin Pract 3:59–64. https://doi.org/10.1002/mdc3.12239
Keijsers NLW, Horstink MWIM, Gielen SCAM (2006) Ambulatory motor assessment in Parkinson’s disease. Mov Disord 21:34–44. https://doi.org/10.1002/mds.20633
Kim J-W, Lee J-H, Kwon Y et al (2011) Quantification of bradykinesia during clinical finger taps using a gyrosensor in patients with Parkinson’s disease. Med Biol Eng Comput 49:365–371. https://doi.org/10.1007/s11517-010-0697-8
Kim J-W, Kwon Y, Kim Y-M et al (2012) Analysis of lower limb bradykinesia in Parkinson’s disease patients. Geriatr Gerontol Int 12:257–264. https://doi.org/10.1111/j.1447-0594.2011.00761.x
Kim J-W, Kwon Y, Yun J-S et al (2015) Regression models for the quantification of Parkinsonian bradykinesia. Biomed Mater Eng 26(Suppl 1):S2249–S2258. https://doi.org/10.3233/BME-151531
Koop MM, Shivitz N, Brontë-Stewart H (2008) Quantitative measures of fine motor, limb, and postural bradykinesia in very early stage, untreated Parkinson’s disease. Mov Disord 23:1262–1268. https://doi.org/10.1002/mds.22077
Lee CY, Kang SJ, Hong S-K et al (2016) A validation study of a Smartphone-based finger tapping application for quantitative assessment of bradykinesia in Parkinson’s disease. PLoS One 11:e0158852. https://doi.org/10.1371/journal.pone.0158852
Lin Z, Dai H, Xiong Y, et al (2017) Quantification assessment of bradykinesia in Parkinson’s disease based on a wearable device. In: 2017 39th annual international conference of the IEEE engineering in medicine and biology society (EMBC). IEEE, pp 803–806
Martinez-Manzanera O, Roosma E, Beudel M et al (2016) A method for automatic and objective scoring of bradykinesia using orientation sensors and classification algorithms. IEEE Trans Biomed Eng 63:1016–1024. https://doi.org/10.1109/TBME.2015.2480242
Memar S, Delrobaei M, Pieterman M et al (2018) Quantification of whole-body bradykinesia in Parkinson’s disease participants using multiple inertial sensors. J Neurol Sci 387:157–165. https://doi.org/10.1016/j.jns.2018.02.001
Mirelman A, Bernad-Elazari H, Thaler A et al (2016) Arm swing as a potential new prodromal marker of Parkinson’s disease. Mov Disord 31:1527–1534. https://doi.org/10.1002/mds.26720
Moreira RC, Zonta MB, de Araújo APS et al (2017) Quality of life in Parkinson’s disease patients: progression markers of mild to moderate stages. Arq Neuropsiquiatr 75:497–502. https://doi.org/10.1590/0004-282X20170091
Muhamed SA, Newby R, Smith SL et al (2018) Objective evaluation of bradykinesia in Parkinson’s disease using evolutionary algorithms. In: Proceedings of the 11th international joint conference on biomedical engineering systems and technologies (BIOSTEC 2018). SCITEPRESS – Science and Technology Publications, pp 63–69. https://doi.org/10.5220/0006601700630069
Ossig C, Antonini A, Buhmann C et al (2016) Wearable sensor-based objective assessment of motor symptoms in Parkinson’s disease. J Neural Transm 123:57–64. https://doi.org/10.1007/s00702-015-1439-8
Pastorino M, Cancela J, Arredondo MT et al (2011) Assessment of Bradykinesia in Parkinson’s disease patients through a multi-parametric system. Conf Proc Annu Int Conf IEEE Eng Med Biol Soc IEEE Eng Med Biol Soc Annu Conf 2011:1810–1813. https://doi.org/10.1109/IEMBS.2011.6090516
Patel S, Lorincz K, Hughes R et al (2009) Monitoring motor fluctuations in patients with Parkinson’s disease using wearable sensors. IEEE Trans Inf Technol Biomed 13:864–873. https://doi.org/10.1109/TITB.2009.2033471
Patel S, Chen B-R, Buckley T et al (2010) Home monitoring of patients with Parkinson’s disease via wearable technology and a web-based application. Conf Proc Annu Int Conf IEEE Eng Med Biol Soc IEEE Eng Med Biol Soc Annu Conf 2010:4411–4414. https://doi.org/10.1109/IEMBS.2010.5627124
Perlmutter JS (2009) Assessment of Parkinson disease manifestations. Curr Protoc Neurosci Chapter 10(Unit10):1. https://doi.org/10.1002/0471142301.ns1001s49
Phan D, Horne M, Pathirana PN, Farzanehfar P (2018) Measurement of axial rigidity and postural instability using wearable sensors. Sensors (Basel). https://doi.org/10.3390/s18020495
Politis M, Wu K, Molloy S et al (2010) Parkinson’s disease symptoms: the patient’s perspective. Mov Disord 25:1646–1651. https://doi.org/10.1002/mds.23135
Rabelo AG, Neves LP, Paixão APS et al (2017) Objective assessment of bradykinesia estimated from the wrist extension in older adults and patients with Parkinson’s disease. Ann Biomed Eng 45:2614–2625. https://doi.org/10.1007/s10439-017-1908-3
Richards M, Marder K, Cote L, Mayeux R (1994) Interrater reliability of the unified Parkinson’s disease rating scale motor examination. Mov Disord 9:89–91
Riva F, Toebes MJP, Pijnappels M et al (2013) Estimating fall risk with inertial sensors using gait stability measures that do not require step detection. Gait Posture 38:170–174. https://doi.org/10.1016/j.gaitpost.2013.05.002
Rodríguez-Molinero A, Samà A, Pérez-López C et al (2017) Analysis of Correlation between an accelerometer-based algorithm for detecting Parkinsonian gait and UPDRS subscales. Front Neurol 8:431. https://doi.org/10.3389/fneur.2017.00431
Rodríguez-Molinero A, Pérez-López C, Samà A et al (2018) A kinematic sensor and algorithm to detect motor fluctuations in Parkinson disease: validation study under real conditions of use. JMIR Rehabil Assist Technol 5:e8. https://doi.org/10.2196/rehab.8335
Rovini E, Maremmani C, Cavallo F (2017) How wearable sensors can support Parkinson’s disease diagnosis and treatment: a systematic review. Front Neurosci 11:555. https://doi.org/10.3389/fnins.2017.00555
Ruonala V, Pekkonen E, Airaksinen O et al (2018) Levodopa-induced changes in electromyographic patterns in patients with advanced Parkinson’s disease. Front Neurol 9:35. https://doi.org/10.3389/fneur.2018.00035
Salarian A, Russmann H, Wider C et al (2007) Quantification of tremor and bradykinesia in Parkinson’s disease using a novel ambulatory monitoring system. IEEE Trans Biomed Eng 54:313–322. https://doi.org/10.1109/TBME.2006.886670
Samà A, Pérez-López C, Rodríguez-Martín D et al (2017) Estimating bradykinesia severity in Parkinson’s disease by analysing gait through a waist-worn sensor. Comput Biol Med 84:114–123. https://doi.org/10.1016/j.compbiomed.2017.03.020
Schaafsma JD, Giladi N, Balash Y et al (2003) Gait dynamics in Parkinson’s disease: relationship to Parkinsonian features, falls and response to levodopa. J Neurol Sci 212:47–53. https://doi.org/10.1016/S0022-510X(03)00104-7
Schilder JCM, Overmars SS, Marinus J et al (2017) The terminology of akinesia, bradykinesia and hypokinesia: past, present and future. Parkinsonism Relat Disord 37:27–35. https://doi.org/10.1016/j.parkreldis.2017.01.010
Shah A, Coste J, Lemaire J-J et al (2017) A novel assistive method for rigidity evaluation during deep brain stimulation surgery using acceleration sensors. J Neurosurg 127:602–612. https://doi.org/10.3171/2016.8.JNS152770
Souissi H, Zory R, Bredin J et al (2018) Co-contraction around the knee and the ankle joints during post-stroke gait. Eur J Phys Rehabil Med 54:380–387. https://doi.org/10.23736/S1973-9087.17.04722-0
Sringean J, Taechalertpaisarn P, Thanawattano C, Bhidayasiri R (2016) How well do Parkinson’s disease patients turn in bed? Quantitative analysis of nocturnal hypokinesia using multisite wearable inertial sensors. Parkinsonism Relat Disord 23:10–16. https://doi.org/10.1016/j.parkreldis.2015.11.003
Sringean J, Anan C, Thanawattano C, Bhidayasiri R (2017) Time for a strategy in night-time dopaminergic therapy? An objective sensor-based analysis of nocturnal hypokinesia and sleeping positions in Parkinson’s disease. J Neurol Sci 373:244–248. https://doi.org/10.1016/j.jns.2016.12.045
Stamatakis J, Ambroise J, Crémers J et al (2013) Finger tapping clinimetric score prediction in Parkinson’s disease using low-cost accelerometers. Comput Intell Neurosci 2013:717853. https://doi.org/10.1155/2013/717853
Steins D, Dawes H, Esser P, Collett J (2014) Wearable accelerometry-based technology capable of assessing functional activities in neurological populations in community settings: a systematic review. J Neuroeng Rehabil 11:36. https://doi.org/10.1186/1743-0003-11-36
Summa S, Tosi J, Taffoni F et al (2017) Assessing bradykinesia in Parkinson’s disease using gyroscope signals. IEEE Int Conf Rehabil Robot 2017:1556–1561. https://doi.org/10.1109/ICORR.2017.8009469
van Hilten JJ, van Eerd AA, Wagemans EAH et al (1998) Bradykinesia and hypokinesia in Parkinson’s disease: what’s in a name? J Neural Transm 105:229–237. https://doi.org/10.1007/s007020050051
van Schooten KS, Pijnappels M, Rispens SM et al (2016) Daily-life gait quality as predictor of falls in older people: a 1-year prospective cohort study. PLoS One 11:e0158623. https://doi.org/10.1371/journal.pone.0158623
Weiss A, Shimkin I, Giladi N, Hausdorff JM (2010) Automated detection of near falls: algorithm development and preliminary results. BMC Res Notes 3:62. https://doi.org/10.1186/1756-0500-3-62
Xue F, Wang F-Y, Mao C-J et al (2018) Analysis of nocturnal hypokinesia and sleep quality in Parkinson’s disease. J Clin Neurosci 54:96–101. https://doi.org/10.1016/j.jocn.2018.06.016
Zwartjes DGM, Heida T, van Vugt JPP et al (2010) Ambulatory monitoring of activities and motor symptoms in Parkinson’s disease. IEEE Trans Biomed Eng 57:2778–2786. https://doi.org/10.1109/TBME.2010.2049573
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
IT, EG, and IH report no conflicts of interests. NG reports that he is a consultant for: Neuroderm, Intec Pharma, Teva, Genzyme-Sanofi, Biogen, Lysosomal Therapeutics, Denali, Cellanis, GaitBetter, Vibrant and Sionara; that he holds shares or options in Lysosomal Therapeutics, Cellanis, GaitBetter and Vibrant; that he has received royalties from Lysosomal Therapeutics; that he received honorarium from UCB, Teva, Novartis, Abbvie, Genzyme-Sanofie, Neuroderm, Bial, Shire, MDS; that he have chaired the DSMBs for Teva and Pharma2B; that he is a PI on a Center Grant given by Biogen to TLVMC; that he has submitted a patent application on the use of body-fixed sensors for assessing PD symptoms, the intellectual property rights for which are held by the Tel Aviv Medical Center, and that he received grants from Teva, Biogen, LTI, ISF, EU, NIH, MJFF, Parkinson Foundation, and Pfizer. AM serves as chair of the Michael J Fox Foundation task force on gait. She has submitted a patent application on the use of body-fixed sensors for assessing PD symptoms, the intellectual property rights for which are held by the Tel Aviv Medical Center. JH has or currently serves on the Movement Disorders Society Technology Task Force, on the Michael J Fox Foundation task force on gait, on the board of the International Society for the Measurement of Physical Behavior, and on advisory boards for Sanofi and Biogen. He has submitted a patent application on the use of body-fixed sensors for assessing PD symptoms, the intellectual property rights for which are held by the Tel Aviv Medical Center. He has received grant support from the NIH, the Michael J Fox Foundation for Parkinson’s Research, the EU (H2020), the BSF, the Israeli Science Foundation and the National Multiple Sclerosis Society.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Teshuva, I., Hillel, I., Gazit, E. et al. Using wearables to assess bradykinesia and rigidity in patients with Parkinson’s disease: a focused, narrative review of the literature. J Neural Transm 126, 699–710 (2019). https://doi.org/10.1007/s00702-019-02017-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00702-019-02017-9