Abstract
A colorimetric assay is described for determination of cytosine-rich ssDNA at physiological pH values. The working principle is based on (a) Ag(I) ion-induced formation of an i-motif structure, and (b) glucose oxidase-controlled growth of gold nanoparticles (AuNPs). The combination between Ag+ and cytosine-rich DNA can modulate the generation of H2O2 resulting from enzyme catalyzed glucose oxidation. Depending on the amount of H2O2 formed, the solution containing the AuNPs will turn red in the presence of cytosine-rich ssDNA but blue in the absence of such DNA if Ag+ is added before the formation of the red AuNPs. Upon addition of C-DNA at different concentrations, the peak shift (Δλ) of the AuNP solution relative to the SPR peak position (560 nm) in the absence of C-DNA is taken as the signal readout. The method shows a good linear response toward C-DNA over the range 10–200 nM with a detection limit of 2.7 nM. It may also be performed visually. The photometric assay is highly sensitive, specific, and rapid. The method is particularly attractive in terms of applications such as in human serum analysis, a colorimetric logic gate, and the calculation of binding constants for the interaction between Ag+ and glucose oxidase (GOx), and between Ag+ and cytosine-rich ssDNAs.

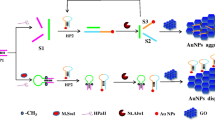
Schematic presentation of colorimetric detection of cytosine (C)-rich ssDNA (C-DNA) based on the modulation of the glucose oxidase (GOx)-catalyzed growth of gold nanoparticles (AuNPs) with Ag+ as the enzyme inhibitor.





Similar content being viewed by others
References
Saxena S, Joshi S, Shankaraswamy J, Tyagi S, Kukreti S (2017) Magnesium and molecular crowding of the cosolutes stabilize the i-motif structure at physiological pH. Biopolymers 107:e23018. https://doi.org/10.1002/bip.23018
Brazier JA, Fisher J, Cosstick R (2006) Stabilization of the DNA I-motif structure by incorporation of 3′-s-phosphorothiolate linkages. Angew Chem Int Ed 45:114–117. https://doi.org/10.1002/anie.200503110
Phan AT, Gueron M, Leroy JL (2000) The solution structure and internal motions of a fragment of the cytidine-rich strand of the human telomere. J Mol Biol 299:123–144. https://doi.org/10.1006/jmbi.2000.3613
Day HA, Pavlou P, Waller ZAE (2014) I-motif DNA: structure, stability and targeting with ligands. Bioorg Med Chem 22(16):4407–4418. https://doi.org/10.1016/j.bmc.2014.05.047
Maurice G, Leroy J-L (2000) The i-motif in nucleic acids. Curr Opin Struct Biol 10(3):326–331. https://doi.org/10.1016/S0959-440X(00)00091-9
Lacroix L, Lienard H, Labourier E, Djavaheri M-M, Lacoste J, Leffers H, Tazi J, Helene C, Mergny JL (2000) Identification of two human nuclear proteins that recognize the cytosine-rich strand of human telomeres in vitro. Nucleic Acids Res 28(7):1564–1575. https://doi.org/10.1093/nar/28.7.1564
Kang HJ, Kendrick S, Hecht SM, Hurley LH (2014) The transcriptional complex between the BCL2 i-motif and hnRNP LL is a molecular switch for control of gene expression that can be modulated by small molecules. J Am Chem Soc 136:4172–4185. https://doi.org/10.1021/ja4109352
Guo KX, Alan P, Kara B-R, Vijay G, Daekyu S, Laurence H (2007) Formation of pseudosymmetrical G-quadruplex and i-motif structures in the proximal promoter region of the RET oncogene. J Am Chem Soc 129(33):10220–10228. https://doi.org/10.1021/ja072185g
Dai JX, Hatzakis E, Hurley LH, Yang DZ (2010) I-motif structures formed in the human c-MYC promoter are highly dynamic-insights into sequence redundancy and i-motif stability. PLoS One 5(7):e11647. https://doi.org/10.1371/journal.pone.0011647
Sun D, Hurley LH (2009) The importance of negative superhelicity in inducing the formation of G-Quadruplex and i-motif structures in the c-myc promoter: implications for drug targeting and control of gene expression. J Med Chem 52(9):2863–2874. https://doi.org/10.1021/jm900055s
Hurley LH (2001) Secondary DNA structures as molecular targets for cancer therapeutics. Biochem Soc Trans 29(6):692–696. https://doi.org/10.1042/bst0290692
Joo H, Seo YJ (2015) A novel gold-based molecular beacon for probing CNG DNA repeat sequences. Tetrahedron Lett 56:542–545. https://doi.org/10.1016/j.tetlet.2014.12.004
Kim KT, Veedu RN, Seo YJ, Kim BH (2014) Quencher-free molecular beacons as probes for oligonucleotides containing CAG repeat sequences. Chem Commun 50(13):1561–1563. https://doi.org/10.1039/C3CC48853E
Kim KT, Kim HW, Moon D, Rhee YM, Kim BH (2013) DNSC: a fluorescent, environmentally sensitive cytidine derivative for the direct detection of GGG triad sequences. Org Biomol Chem 11(34):5605–5614. https://doi.org/10.1039/C3OB41222A
Shibata T, Nakatani K (2016) Fluorescence probe for detecting CCG trinucleotide repeat DNA expansion and slip-out. ChemBioChem 17(18):1685–1688. https://doi.org/10.1002/cbic.201600200
Park Y, Kim KT, Kim BH (2016) G-Quadruplex formation using fluorescent oligonucleotides as a detection method for discriminating AGG trinucleotide repeats. Chem Commun 52(86):12757–12760. https://doi.org/10.1039/C6CC06566J
Chen C, Li M, Xing Y, Li Y, Joedecke CC, Jin J, Yang Z, Liu D (2012) Study of pH-induced folding and unfolding kinetics of the DNA i-motif by stopped-flow circular dichroism. Langmuir 51:17743–17748. https://doi.org/10.1021/la303851a
Choi J, Kim S, Tachikawa T, Fujitsuka M, Majima T (2011) pH-induced intramolecular folding dynamics of i-motif DNA. J Am Chem Soc 133:16146–16153. https://doi.org/10.1021/ja2061984
Elbaz J, Wang ZG, Orbach R, Willner I (2009) pH-stimulated concurrent mechanical activation of two DNA “tweezers”. A “SET-RESET” logic gate system. Nano Lett 9:4510–4514. https://doi.org/10.1021/nl902859m
Fu XL, Chen LX, Choo J (2017) Optical nanoprobes for ultrasensitive immunoassay. Anal Chem 89(1):124–137. https://doi.org/10.1021/acs.analchem.6b02251
Rica R, Stevens MM (2012) Plasmonic ELISA for the ultrasensitive detection of disease biomarkers with the naked eye. Nat Nanotechnol 7(12):821–824. https://doi.org/10.1038/nnano.2012.186
Day HA, Huguin C, Waller ZAE (2013) Silver cations fold i-motif at neutral pH. Chem Commun 49:7696–7698. https://doi.org/10.1039/C3CC43495H
Liu D, Wang Z, Jiang X (2011) Gold nanoparticles for the colorimetric and fluorescent detection of ions and small organic molecules. Nanoscale 3:1421–1433. https://doi.org/10.1039/C0NR00887G
Deng JJ, Yu P, Wang YX, Yang LF, Mao LQ (2014) Nanoparticles: visualization and quantification of neurochemicals with gold nanoparticles: opportunities and challenges. Adv Mater 26(40):6984. https://doi.org/10.1002/adma.201470279
Rust IM, Goran JM, Stevenson KJ (2015) Amperometric detection of aqueous silver ions by inhibition of glucose oxidase immobilized on nitrogen-doped carbon nanotube electrodes. Anal Chem 87(14):7250–7257. https://doi.org/10.1021/acs.analchem.5b01224
Guascito MR, Malitesta C, Mazzotta E, Turco A (2008) Inhibitive determination of metal ions by an amperometric glucose oxidase biosensor study of the effect of hydrogen peroxide decomposition. Sensors Actuators B Chem 131:394–402. https://doi.org/10.1016/j.snb.2007.11.049
Vilela D, González MC (2012) A Escarpa sensing colorimetric approaches based on gold and silver nanoparticles aggregation: chemical creativity behind the assay. A review. Anal Chim Acta 751:24–43. https://doi.org/10.1016/j.aca.2012.08.043
Liu Y, Liu YL, Xu L, Li J, Liu XY, Liu J, Li GY (2017) Highly selective, colorimetric detection of Hg2+ based on three color changes of AuNPs solution from red through sandy beige to celandine green. Sensors Actuators B Chem 249:331–338. https://doi.org/10.1016/j.snb.2017.04.116
Deng JJ, Yu P, Yang LF, Mao LQ (2013) Competitive coordination of Cu2+ between cysteine and pyrophosphate ion: toward sensitive and selective sensing of pyrophosphate ion in synovial fluid of arthritis patients. Anal Chem 85(4):2516–2522. https://doi.org/10.1021/ac303698p
Peng MP, Ma W, Long YT (2015) Alcohol dehydrogenase-catalyzed gold nanoparticle seed-mediated growth allows reliable detection of disease biomarkers with the naked eye. Anal Chem 87(12):5891–5896. https://doi.org/10.1021/acs.analchem.5b00287
Wang H, Zhang LY, Jiang Y, Chen LJ, Zhu SY (2018) A sandwiched electroanalysis method for probing Anthrax DNAs based on glucose-induced gold growth and catalytic coupling of tyramine using gold-mineralized glucose oxidase. Sensors Actuators B Chem 261:441–450. https://doi.org/10.1016/j.snb.2018.01.171
Reichmann ME, Rice SA, Thomas CA, Doty PA (1954) Further examination of the molecular weight and size of desoxypentose nucleic acid. J Am Chem Soc 76:3047–3053
Dixon M (1953) The determination of enzyme inhibitor constants. Biochem J 55:170–171. https://doi.org/10.1042/bj0550170
Chipman DM, Grisaro V, Sharon N (1967) The binding of oligosaccharides containing N-acetylglucosamine and N-acetylmuramic acid to lysozyme. J Biol Chem 242:4388–4394
Lin YH, Tseng WL (2009) Highly sensitive and selective detection of silver ions and silver nanoparticles in aqueous solution using an oligonucleotide-based fluorogenic probe. Chem Commun 43:6619–6621. https://doi.org/10.1039/B915990H
Acknowledgements
This work was financially supported by National Natural Science Foundation of China (No. 21765014, 21864018, and 21505067).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The author(s) declare that they have no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOC 1094 kb)
Rights and permissions
About this article
Cite this article
Liu, BW., Wu, YY., Huang, PC. et al. Colorimetric determination of cytosine-rich ssDNA by silver(I)-modulated glucose oxidase-catalyzed growth of gold nanoparticles. Microchim Acta 186, 467 (2019). https://doi.org/10.1007/s00604-019-3591-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00604-019-3591-6