Abstract
This report presents the case of an 84-year-old woman who developed tetanus 3 days after the resection of a gangrenous small intestine caused by obturator hernia incarceration. The diagnosis of tetanus was clinically made after the appearance of generalized spastic contractions with opisthotonus. Clostridium tetani organisms residing in the gastrointestinal tract were presumed to have been endogenously inoculated into the strangulated intestine, where it produced tetanospasmin, causing tetanus. The patient successfully recovered after aggressive intensive care. There have been 16 case reports of tetanus occurring after gastrointestinal surgical procedures. Primary care physicians should thus be aware of the fact that, although extremely rare, C. tetani residing in the gastrointestinal tract can provide a possible endogenous source of tetanus infection.
Similar content being viewed by others
Introduction
Tetanus is a nervous system disorder characterized by muscle spasms that are caused by the toxin-producing anaerobe, Clostridium tetani. Tetanus is often a severe and fatal disease, although it is currently rare in developed countries where active immunization programs provide immunity to the infection in the general population. The incidence of tetanus in Japan has been reported to range from 66 to 123 cases per year during the period between 1999 and 2009 [1].
Tetanus usually occurs after C. tetani of exogenous origin enters through acute injuries or chronic wounds. Postoperative tetanus after a gastrointestinal surgical procedure is uncommon. This report presents a case of generalized tetanus after an emergency operation for strangulated ileus caused by obturator hernia incarceration.
Case report
An 84-year-old woman presented at a community hospital with right groin pain. Vomiting with abdominal pain appeared 2 days later, and she was referred to our hospital. She had a history of hypertension and bilateral knee osteoarthritis for more than 20 years. She had no history of recent trauma or tetanus toxoid immunization.
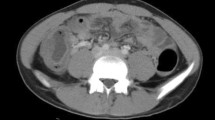
Her blood pressure at the time of admission was 104/59 mmHg, her pulse rate was 80 beats/min, and her body temperature was 38.0°C. The abdomen was distended and there was tenderness around the navel, but no rebound tenderness was found. There were no external injuries or wounds on her body, and her neurological examination was normal. Laboratory findings showed leukocytosis (16900/mm3) and an elevated serum C-reactive protein level (21.3 mg/dl), but no hypoxia or metabolic acidosis was observed. An X-ray of the abdomen showed distended loops of the small bowel with air fluid levels. Computed tomography (CT) revealed diffuse dilatation of the small intestine, and protrusion of the bowel into the right obturator foramen (Fig. 1). She was diagnosed with small bowel obstruction caused by incarceration of a right obturator hernia, and a long intestinal suction tube was inserted to induce intestinal decompression, followed by laparoscopic surgery the next day.
Laparoscopic evaluation showed an incarcerated hernia of the ileum (about 20 cm from the terminal ileum) in the right obturator canal (Fig. 2a), and this was reduced by tractioning the effluent intestine with atraumatic grasping forceps. A white-colored purulent fluid was discharged into the peritoneal cavity during the traction (Fig. 2b), indicating that an abscess had formed in the right obturator canal (Fig. 2c). The pus was obtained for bacteriological examination. The incarcerated ileum was resected because the strangulated region of the ileum was apparently gangrenous, and the bowel was anastomosed extracorporeally.
Intraoperative photographs. a Incarcerated small bowel at right obturator foramen (arrow). Asterisk indicates distended affluent intestine, double asterisk indicates effluent intestine. b A white-colored pus (white arrow) was discharged during traction of the intestine. c A defect in the right obturator canal (arrowhead)
The culture reports on the purulent discharge revealed identification of Proteus vulgaris, Enterobacter cloacae, and Citrobacter sp., which are inhabitants of the intestinal tract in humans, but C. tetani was not isolated. The pathology disclosed that the incarcerated region of the ileum was transmurally necrotic, with hemorrhage and inflammation.
The patient’s postoperative course was uneventful until the evening of the 3rd postoperative day, when both of the patient’s knees were difficult to bend because of muscular rigidity. She complained of pain in her right groin region, but her inguinal region had no signs of redness or swelling. The muscle around the patient’s neck was found to be rigid 2 days later (on the 5th postoperative day), accompanied by a fever of 38°C and excessive sweating. Her abdominal wall was rigid and the groin pain was increasing. These findings led to an examination by abdominal CT to rule out recurrence of the obturator hernia, postoperative peritonitis, or abscess formation. No diagnosis of tetanus was seriously considered at that time. Abdominal CT revealed soft tissue density on the right obturator canal, compatible with the postoperative changes and/or infection, but no abscess was detected. She subsequently presented with generalized tonic and periodic spastic muscular contractions with opisthotonus, accompanied by risus sardonicus. A diagnosis of generalized tetanus was finally made, followed by the administration of human tetanus immune globulin, tetanus toxoid vaccination, and penicillin G administration. She was intubated, artificially ventilated, and sedated with propofol in the intensive care unit. Vecuronium was continuously infused for 21 days following the 7th postoperative day.
The patient successfully recovered following a long period of intensive care and two episodes of mechanical ventilation-related pneumonia. The ventilator was disconnected on the 41st postoperative day. She was transferred to the convalescence ward for rehabilitation.
Discussion
Tetanus occurs when spores of C. tetani, an obligate anaerobe normally present in the gut of mammals and widely found in soil, gains access to damaged human tissue. Clostridium tetani transforms into a vegetative rod-shaped bacterium after inoculation, and produces the metalloprotease, tetanospasmin (also known as tetanus toxin) [2].
It is known that a diagnosis of tetanus is usually made clinically based on the characteristic symptoms of tetanus, and C. tetani is not isolated from wounds in many cases. Likewise, the diagnosis of tetanus in the present case was based upon the clinical findings, but it was not recognized in the early stage, until generalized spastic contractions with opisthotonus appeared. Tetanus has become an uncommon disease in developed countries, including Japan, so many practicing primary care physicians have never experienced a case of the disease during their career [3]. Furthermore, the possibility of tetanus occurring after gastrointestinal surgical procedures in the absence of external primary injuries is not widely known. The medical staff in this hospital had never experienced a case of tetanus. The initial symptom of muscular rigidity in both of the patient’s legs was assumed to be due to chronic osteoarthritis in her knees, which had been difficult to bend for years.
We searched the English-language articles from 1966 to December 2010 in the MeSH system of PubMed for relevant case reports and articles. The MeSH keywords that were used included “tetanus,” combined with “digestive system surgical procedures” and “surgical procedures operative.” All relevant articles were reviewed, and their reference lists were examined for other relevant articles. Other articles were obtained and reviewed from these reference lists. We found a total of only 16 case reports of tetanus after gastrointestinal surgical procedures. The procedures included open cholecystectomy (four cases) [4–6] cholecystectomy with exploration of the bile duct (two cases) [7], resection for a gangrenous small intestine (two cases) [8, 9], rubber band ligation of hemorrhoids (two cases) [10, 11], cryosurgery for internal hemorrhoids [12], drainage of an anorectal abscess [13], sigmoidoscopic polypectomy [14], gastrectomy and transverse colectomy [15], pyloroplasty and oversewing of a bleeding duodenal ulcer [16], and exploratory laparotomy for carcinoma in the omentum and liver [5]. These cases are summarized in Table 1. Tetanus can therefore occur after a wide variety of gastrointestinal surgical procedures.
The entry point of C. tetani in the current patient was presumed to be the strangulated necrotic ileum incarcerated in the right obturator canal. There had been no other focus of infection where anaerobic bacteria could grow during the possible incubation period in this patient. There is evidence that C. tetani spores exist in the gut of asymptomatic individuals. For example, Meyer et al. [17] reported that the stools of healthy individuals often contain tetanus organisms. A convergence of factors must occur for the tetanus toxin to be elaborated in the human host, such as a penetrating injury, coinfection with other bacteria, devitalized tissue, a foreign body, and localized ischemia [2]. The spores of C. tetani residing in the gut of the current patient were thought to be inoculated into the devitalized, necrotic strangulated intestine, resulting in bacterial growth and production of tetanospasmin. Postsurgical immunosuppression [18] might also affect the progression of the disease. Primary care physicians should be aware that, although extremely rare, C. tetani organisms residing in the gastrointestinal tract represent a possible endogenous source of tetanus infection.
The number of reported cases of tetanus has declined dramatically over the past 50 years in Japan, due to the routine immunization of the diphtheria–tetanus–pertussis combined vaccine (DTP) beginning in 1968 [19]. However, many adults who were born before the introduction of routine DTP immunization are inadequately vaccinated against tetanus. The provisional report of the Japanese National Epidemiological Surveillance of Vaccine-Preventable Diseases in 2008 found that the rate of tetanus antitoxin positivity (those with an antitoxin titer higher than 0.01 IU/ml, which is the minimum level of protective immunity to tetanus) was as high as ≥92% among individuals up to 35–39 years of age, but the positive rate was 25% in the 45- to 59-year-old individuals, and for individuals older than 60 years was as low as 11% [1]. These findings are strongly correlated with the report on the age distribution of tetanus cases, which found that the majority of tetanus patients were older than 40 years during the period from 2004 to 2008; i.e., 94% were older than 40 and 75% were older than 60 years [1]. Tetanus is a preventable, but possibly lethal disease. Immunization of the population ≥40 years of age is recommended for elimination of the disease in Japan because tetanus can occur in the absence of external primary injuries, as in the present case.
References
Tetanus in Japan as of December 2008. Infectious Agent Surveillance Report (IASR). 2009;30:65–6.
Sexton DJ, Bartlett JG, Thorner AR. Tetanus. http://www.uptodate.com. Accessed December 20, 2010.
Cannarella R, Agbayani E. Tetanus: a case report, epidemiology review and recommendations for immunization compliance. W V Med J. 2001;97:253–6.
Crokaert F, Glupczynski Y, Fastrez R, Allé JL, Yourassowsky E. Postoperative tetanus. Lancet. 1984;323:1466.
Parker L, Mandal BK. Postoperative tetanus. Lancet. 1984;324:407.
O’Riordain DD, Buckley DJ, Kirwan WO. Tetanus following cholecystectomy. Ir J Med Sci. 1991;160:310.
Lennard TWJ, Gunn A, Sellers J, Stoddart JC. Tetanus after elective cholecystectomy and exploration of the common bile duct. Lancet. 1984;323:1466–7.
Clay RC, Bolton JW. Tetanus arising from gangrenous unperforated small intestine. JAMA. 1964;187:856–8.
Furui J, Enjyoji A, Susumu S, Okudaira S, Takayama K, Kanematsu T. Tetanus after a resection for a gangrenous perforated small intestine: report of a case. Surg Today. 1999;29:626–8.
Murphy KJ. Tetanus after rubber-band ligation of haemorrhoids. Br Med J. 1978;1:1590–1.
Kasher JA, Mathisen G. Acquiring tetanus after hemorrhoid banding and other gastrointestinal procedures. J Gastrointest Surg. 2007;11:515–9.
Singh K, Chhina RS, Kaul TK. Postoperative tetanus following cryosurgery for haemorrhoids. Trop Doct. 1992;22:135–6.
Myers KJ, Heppell T, Bode WE, Culp CE, Thurber DL, Van Scoy RE. Tetanus after anorectal abscess. Mayo Clin Proc. 1984;59:429–30.
Segel J, Shaff BL. An unusual case of postoperative tetanus. S Afr Med J. 1969;43:1556–7.
Fleshner PR, Hunter JG, Rudick J. Tetanus after gastrointestinal surgery. Am J Gastroenterol. 1988;83:298–300.
Katz KC, Walmsley SL. Postoperative tetanus: a case report. CMAJ. 2000;163:571–3.
Meyer KA, Spector BK. The incidence of tetanus bacilli in the stools and on the regional skin of one hundred urban herniotomy cases. Surg Gynecol Obstet. 1932;54:785–9.
Kimura F, Shimizu H, Yoshidome H, Ohtsuka M, Miyazaki M. Immunosuppression following surgical and traumatic injury. Surg Today. 2010;40:793–808.
Tetanus in Japan as of 2001. Infectious Agent Surveillance Report (IASR). 2002;23:1–2.
Acknowledgments
The authors sincerely appreciate the kind cooperation and great support of the entire medical and technical staff of Shirane Tokushukai Hospital.
Conflict of interest
The authors do not have any financial relationships with organizations, and have no conflict of interest with regard to this report.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mori, M., Iida, H., Miki, K. et al. Postoperative tetanus after laparoscopic obturator hernia repair for strangulated ileus: report of a case. Surg Today 42, 470–474 (2012). https://doi.org/10.1007/s00595-011-0023-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00595-011-0023-6