Abstract
Aims
Diabetic cardiomyopathy (DCM) is a specific myocardial alteration in patients with diabetics. LncRNA KCNQ1OT1 has been previously demonstrated to be involved in various diabetic complications. Our aims are to further investigate the underlying regulatory mechanisms/pathways of KCNQ1OT1 in DCM.
Methods
In vitro and in vivo models of DCM were established in high glucose (HG)-treated human cardiomyocytes and in streptozotocin (STZ)-induced diabetic mice, respectively. Gene and protein expressions were examined by qPCR, western blotting and ELISA. Cell proliferation and apoptosis were determined by CCK8 assay, flow cytometry and TUNEL staining. The association between KCNQ1OT1 and miR-181a-5p, miR-181a-5p and PDCD4 was predicted using bioinformatics methods and subsequently confirmed by dual luciferase reporter and RNA immunoprecipitation assays. Mouse cardiac tissues were collected and analysed using HE staining, Masson’s staining and immunohistochemical analysis.
Results
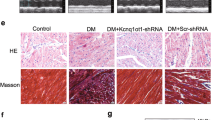
KCNQ1OT1 and PDCD4 were upregulated in HG-treated human cardiomyocytes, while miR-181a-5p was downregulated. In addition, KCNQ1OT1 could negatively regulate miR-181a-5p expression; meanwhile, miR-181a-5p also negatively regulated PDCD4 expression. KCNQ1OT1 silencing suppressed the expression of inflammatory cytokines and cell apoptosis in vitro, whereas inhibition of miR-181a-5p abrogated those effects of KCNQ1OT1 knockdown. Moreover, overexpressed PDCD4 abolished the inhibition on inflammation and apoptosis caused by miR-181a-5p overexpression. Finally, KCNQ1OT1 knockdown reduced the expression of PDCD4 via regulating miR-181a-5p and inhibited myocardial inflammation and cardiomyocyte apoptosis in the in vivo DCM model.
Conclusions
Our findings suggest that KCNQ1OT1 and its target gene miR-181a-5p regulate myocardial inflammation and cardiomyocyte apoptosis by modulating PDCD4 in DCM.








Similar content being viewed by others
Availability of data and materials
All data generated or analysed during this study are included in this article. The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- 3′-UTR:
-
3′-Untranslated region
- DCM:
-
Diabetic cardiomyopathy
- HG:
-
High glucose
- IL:
-
Interleukin
- LncRNA:
-
Long non-coding RNA
- miRNA:
-
MicroRNA
- OD:
-
Optical density
- qPCR:
-
Quantitative real-time PCR
- sh:
-
Short-hairpin
- STZ:
-
Streptozotocin
References
Alonso N, Moliner P, Mauricio D (2018) Pathogenesis, clinical features and treatment of diabetic cardiomyopathy. Adv Exp Med Biol 1067:197–217. https://doi.org/10.1007/5584_2017_105
Bhan A, Soleimani M, Mandal SS (2017) Long noncoding RNA and cancer: a new paradigm. Cancer Res 77:3965–3981. https://doi.org/10.1158/0008-5472.Can-16-2634
Bugger H, Abel ED (2014) Molecular mechanisms of diabetic cardiomyopathy. Diabetologia 57:660–671. https://doi.org/10.1007/s00125-014-3171-6
Chandramouli C, Reichelt ME, Curl CL et al (2018) Diastolic dysfunction is more apparent in STZ-induced diabetic female mice, despite less pronounced hyperglycemia. Sci Rep 8:2346. https://doi.org/10.1038/s41598-018-20703-8
Chang CP, Han P (2016) Epigenetic and lncRNA regulation of cardiac pathophysiology. Biochem Biophys Acta 1863:1767–1771. https://doi.org/10.1016/j.bbamcr.2016.03.005
Chen Y, Tan S, Liu M, Li J (2018) LncRNA TINCR is downregulated in diabetic cardiomyopathy and relates to cardiomyocyte apoptosis. Scand Cardiovasc J SCJ 52:335–339. https://doi.org/10.1080/14017431.2018.1546896
Chen Y, Zhang Z, Zhu D, Zhao W, Li F (2019) Long non-coding RNA MEG3 serves as a ceRNA for microRNA-145 to induce apoptosis of AC16 cardiomyocytes under high glucose condition. Biosci Rep. https://doi.org/10.1042/bsr20190444
Cheng Y, Li J, Wang C et al (2019) Inhibition of long non-coding RNA Malat1 attenuates high glucose-induced cardiomyocyte apoptosis via regulation of miR-181a-5p. Exp Anim. https://doi.org/10.1538/expanim.19-0058
Cheng Y, Li J, Wang C et al (2020) Inhibition of long non-coding RNA metastasis-associated lung adenocarcinoma transcript 1 attenuates high glucose-induced cardiomyocyte apoptosis via regulation of miR-181a-5p. Exp Anim 69:34–44. https://doi.org/10.1538/expanim.19-0058
Ding G, Zhou L, Shen T, Cao L (2018) IFN-gamma induces the upregulation of RFXAP via inhibition of miR-212-3p in pancreatic cancer cells: a novel mechanism for IFN-gamma response. Oncol Lett 15:3760–3765. https://doi.org/10.3892/ol.2018.7777
Drilon A, Nagasubramanian R, Blake JF et al (2017) A next-generation TRK kinase inhibitor overcomes acquired resistance to prior TRK kinase inhibition in patients with TRK fusion-positive solid tumors. Cancer Discov 7:963–972. https://doi.org/10.1158/2159-8290.Cd-17-0507
Feng SD, Yang JH, Yao CH et al (2017) Potential regulatory mechanisms of lncRNA in diabetes and its complications. Biochem Cell Biol Biochim Biol Cell 95:361–367. https://doi.org/10.1139/bcb-2016-0110
Iorio MV, Croce CM (2012) MicroRNA dysregulation in cancer: diagnostics, monitoring and therapeutics. A comprehensive review. EMBO Mol Med 4:143–159. https://doi.org/10.1002/emmm.201100209
Jia G, DeMarco VG, Sowers JR (2016) Insulin resistance and hyperinsulinaemia in diabetic cardiomyopathy. Nat Rev Endocrinol 12:144–153. https://doi.org/10.1038/nrendo.2015.216
Jia G, Whaley-Connell A, Sowers JR (2018) Diabetic cardiomyopathy: a hyperglycaemia- and insulin-resistance-induced heart disease. Diabetologia 61:21–28. https://doi.org/10.1007/s00125-017-4390-4
Jiang Y, Du W, Chu Q et al (2018) Downregulation of long non-coding RNA Kcnq1ot1: an important mechanism of arsenic trioxide-induced long QT syndrome. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharmacol 45:192–202. https://doi.org/10.1159/000486357
Kanduri C (2011) Kcnq1ot1: a chromatin regulatory RNA. Semin Cell Dev Biol 22:343–350. https://doi.org/10.1016/j.semcdb.2011.02.020
Latronico MV, Elia L, Condorelli G, Catalucci D (2008) Heart failure: targeting transcriptional and post-transcriptional control mechanisms of hypertrophy for treatment. Int J Biochem Cell Biol 40:1643–1648. https://doi.org/10.1016/j.biocel.2008.03.002
Li JZ, Gao W, Ho WK et al (2016) The clinical association of programmed cell death protein 4 (PDCD4) with solid tumors and its prognostic significance: a meta-analysis. Chin J Cancer 35:95. https://doi.org/10.1186/s40880-016-0158-3
Li Y, Lu J, Bao X et al (2016) MiR-499-5p protects cardiomyocytes against ischaemic injury via anti-apoptosis by targeting PDCD4. Oncotarget 7:35607–35617. https://doi.org/10.18632/oncotarget.9597
Liu HJ, Fan YL, Liao HH et al (2017) Apigenin alleviates STZ-induced diabetic cardiomyopathy. Mol Cell Biochem 428:9–21. https://doi.org/10.1007/s11010-016-2913-9
Lo Furno D, Graziano AC, Caggia S et al (2013) Decrease of apoptosis markers during adipogenic differentiation of mesenchymal stem cells from human adipose tissue. Apoptosis Int J Program Cell Death 18:578–588. https://doi.org/10.1007/s10495-013-0830-x
Lozano-Bartolome J, Llaurado G, Portero-Otin M et al (2018) Altered expression of miR-181a-5p and miR-23a-3p is associated with obesity and TNFalpha-induced insulin resistance. J Clin Endocrinol Metab 103:1447–1458. https://doi.org/10.1210/jc.2017-01909
Lozano-Bartolomé J, Llauradó G, Portero-Otin M et al (2018) Altered expression of miR-181a-5p and miR-23a-3p is associated with obesity and TNFα-induced insulin resistance. J Clin Endocrinol Metab 103:1447–1458. https://doi.org/10.1210/jc.2017-01909
Meng Q, Li S, Liu Y et al (2019) Circular RNA circSCAF11 accelerates the glioma tumorigenesis through the miR-421/SP1/VEGFA axis. Mol Ther Nucleic Acids 17:669–677. https://doi.org/10.1016/j.omtn.2019.06.022
Pant T, Dhanasekaran A, Fang J et al (2018) Current status and strategies of long noncoding RNA research for diabetic cardiomyopathy. BMC Cardiovasc Disord 18:197. https://doi.org/10.1186/s12872-018-0939-5
Raut SK, Singh GB, Rastogi B et al (2016) miR-30c and miR-181a synergistically modulate p53–p21 pathway in diabetes induced cardiac hypertrophy. Mol Cell Biochem 417:191–203. https://doi.org/10.1007/s11010-016-2729-7
Romaine SP, Tomaszewski M, Condorelli G, Samani NJ (2015) MicroRNAs in cardiovascular disease: an introduction for clinicians. Heart (Br Card Soc) 101:921–928. https://doi.org/10.1136/heartjnl-2013-305402
Rotini A, Martinez-Sarra E, Pozzo E, Sampaolesi M (2018) Interactions between microRNAs and long non-coding RNAs in cardiac development and repair. Pharmacol Res 127:58–66. https://doi.org/10.1016/j.phrs.2017.05.029
Ruan Q, Wang T, Kameswaran V et al (2011) The microRNA-21-PDCD4 axis prevents type 1 diabetes by blocking pancreatic beta cell death. Proc Natl Acad Sci USA 108:12030–12035. https://doi.org/10.1073/pnas.1101450108
Schmitz SU, Grote P, Herrmann BG (2016) Mechanisms of long noncoding RNA function in development and disease. Cell Mol Life Sci CMLS 73:2491–2509. https://doi.org/10.1007/s00018-016-2174-5
Shao J, Pan X, Yin X et al (2019) KCNQ1OT1 affects the progression of diabetic retinopathy by regulating miR-1470 and epidermal growth factor receptor. J Cell Physiol 234:17269–17279. https://doi.org/10.1002/jcp.28344
Shirjang S, Mansoori B, Asghari S et al (2019) MicroRNAs in cancer cell death pathways: apoptosis and necroptosis. Free Radic Biol Med 139:1–15. https://doi.org/10.1016/j.freeradbiomed.2019.05.017
Thomas CM, Yong QC, Seqqat R et al (2013) Direct renin inhibition prevents cardiac dysfunction in a diabetic mouse model: comparison with an angiotensin receptor antagonist and angiotensin-converting enzyme inhibitor. Clin Sci (Lond Engl) 124:529–541. https://doi.org/10.1042/cs20120448
van den Bosch MW, Palsson-Mcdermott E, Johnson DS, O’Neill LA (2014) LPS induces the degradation of programmed cell death protein 4 (PDCD4) to release Twist2, activating c-Maf transcription to promote interleukin-10 production. J Biol Chem 289:22980–22990. https://doi.org/10.1074/jbc.M114.573089
Vausort M, Wagner DR, Devaux Y (2014) Long noncoding RNAs in patients with acute myocardial infarction. Circ Res 115:668–677. https://doi.org/10.1161/circresaha.115.303836
Wang Q, Dong Z, Liu X et al (2013) Programmed cell death-4 deficiency prevents diet-induced obesity, adipose tissue inflammation, and insulin resistance. Diabetes 62:4132–4143. https://doi.org/10.2337/db13-0097
Wang Y, Yang X, Jiang A, Wang W, Li J, Wen J (2019) Methylation-dependent transcriptional repression of RUNX3 by KCNQ1OT1 regulates mouse cardiac microvascular endothelial cell viability and inflammatory response following myocardial infarction. FASEB J 33:13145–13160. https://doi.org/10.1096/fj.201900310R
Xiao J, Pan Y, Li XH et al (2016) Cardiac progenitor cell-derived exosomes prevent cardiomyocytes apoptosis through exosomal miR-21 by targeting PDCD4. Cell Death Dis 7:e2277. https://doi.org/10.1038/cddis.2016.181
Xue M, Zhuo Y, Shan B (2017) MicroRNAs, long noncoding RNAs, and their functions in human disease. Methods Mol Biol (Clifton, NJ) 1617:1–25. https://doi.org/10.1007/978-1-4939-7046-9_1
Yang F, Qin Y, Lv J et al (2018) Silencing long non-coding RNA Kcnq1ot1 alleviates pyroptosis and fibrosis in diabetic cardiomyopathy. Cell Death Dis 9:1000. https://doi.org/10.1038/s41419-018-1029-4
Yang F, Qin Y, Wang Y et al (2018) LncRNA KCNQ1OT1 mediates pyroptosis in diabetic cardiomyopathy. Cell Physiol Biochem 50:1230–1244. https://doi.org/10.1159/000494576
Yang M, Zhai X, Ge T, Yang C, Lou G (2018) miR-181a-5p promotes proliferation and invasion and inhibits apoptosis of cervical cancer cells via regulating inositol polyphosphate-5-phosphatase A (INPP5A). Oncol Res 26:703–712. https://doi.org/10.3727/096504017x14982569377511
Zhang J, Zhang M, Yang Z et al (2020) PDCD4 deficiency ameliorates left ventricular remodeling and insulin resistance in a rat model of type 2 diabetic cardiomyopathy. BMJ Open Diabetes Res Care. https://doi.org/10.1136/bmjdrc-2019-001081
Zhang Y, Zhang L, Wang Y et al (2019) KCNQ1OT1, HIF1A-AS2 and APOA1-AS are promising novel biomarkers for diagnosis of coronary artery disease. Clin Exp Pharmacol Physiol 46:635–642. https://doi.org/10.1111/1440-1681.13094
Zheng D, Ma J, Yu Y et al (2015) Silencing of miR-195 reduces diabetic cardiomyopathy in C57BL/6 mice. Diabetologia 58:1949–1958. https://doi.org/10.1007/s00125-015-3622-8
Zhou Q, Lv D, Chen P et al (2014) MicroRNAs in diabetic cardiomyopathy and clinical perspectives. Front Genet 5:185. https://doi.org/10.3389/fgene.2014.00185
Zhu J, Yao K, Wang Q et al (2016) Ischemic postconditioning-regulated miR-499 protects the rat heart against ischemia/reperfusion injury by inhibiting apoptosis through PDCD4. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharmacol 39:2364–2380. https://doi.org/10.1159/000452506
Acknowledgements
We would like to give our sincere gratitude to the reviewers for their constructive comments. The study was supported by Guangdong Natural Science Foundation (2018A030313535, Guangdong, China), Guangdong Medical Research Fund (A2020038) and Foundation for Basic and Applied Basic Research of Guangdong Province (2019A1515110063), Natural Science Foundation of Tibet Autonomous Region [XZ2020ZR-ZY55(Z)],National Key Research and Development Program (2018YFC2001803),National Natural Science Foundation of China(82002095).
Author information
Authors and Affiliations
Contributions
SFZ helped in conception and study design; YXY and JDX contributed to data acquisition; YH and DWZ were involved in data analysis; ZYX helped in manuscript drafting; SW contributed to manuscript revising. All authors have read and approved the final version of this manuscript to be published.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
All animal study protocols approved by the Animal Ethics Committee of Guangdong Provincial People’s Hospital were performed in accordance with the guidelines by the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
Human and animal rights
Not applicable. This article does not contain any studies with human participants performed by any of the authors.
Informed consent
The informed consent was obtained from the Animal Ethics Committee of Guangdong Provincial People’s Hospital.
Additional information
Managed by Massimo Porta.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhao, SF., Ye, YX., Xu, JD. et al. Long non-coding RNA KCNQ1OT1 increases the expression of PDCD4 by targeting miR-181a-5p, contributing to cardiomyocyte apoptosis in diabetic cardiomyopathy. Acta Diabetol 58, 1251–1267 (2021). https://doi.org/10.1007/s00592-021-01713-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-021-01713-x



