Abstract
Aim
Exendin-4, a glucagon-like peptide-1 (GLP-1) analog, has been used for treating diabetes mellitus (DM). However, its effects on improving the dysfunction of high glucose (HG)-induced endothelial progenitor cells (EPCs) remain unclear. The present study explored the effects of Exendin-4 on improving dysfunction of EPCs and the underlying mechanism.
Methods
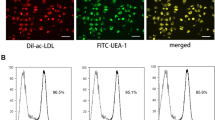
EPCs were isolated from SD rats and identified by flow cytometry. Next, the EPCs were treated by HG and high or low concentration of Exendin-4, and cell viability, migration and tube formation were, respectively, examined by performing MTT assay, wound-healing assay and tube formation assay. Interleukin-6 (IL-6) secretion was measured by enzyme-linked immunosorbent assay (ELISA). The protein expressions of relative stromal-derived growth factor-1β (SDF-1β), C-X-C chemokine receptor type 7 (CXCR7), AMP-activated protein kinase (AMPK), p38 and expressions of CXCR7 and IL-6 in EPCs were measured by Western blot. The cell behaviors of EPCs treated by HG and Exendin-4 with or without silencing of CXCR7 and IL-6 were detected.
Results
Exendin-4 reversed the inhibitory effects of HG on viability, migration and tube formation of EPCs and on SDF-1β/CXCR7–AMPK pathway in EPCs in a dose-dependent manner. Moreover, Exendin-4 promoted the effects of HG on IL-6 level in EPCs through the promotion of p38-MAPK phosphorylation and reduction of cleaved caspase-3 protein expressions in EPCs. However, silencing of CXCR7 and IL-6 reversed the effects of Exendin-4 on cell behaviors, inactivated SDF-1β/CXCR7–AMPK pathway and increased cleaved caspase-3 expression in EPCs.
Conclusions
Exendin-4 could ameliorate HG-induced EPC dysfunction through regulating the production of IL-6 via SDF-1β/CXCR7–AMPK/p38-MAPK axis.





Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Pippitt K, Li M, Gurgle HE (2016) Diabetes mellitus: screening and diagnosis. Am Fam Physician 93(2):103–109
Lipska KJ, Krumholz H, Soones T, Lee SJ (2016) Polypharmacy in the aging patient: a review of glycemic control in older adults with type 2 diabetes. JAMA 315(10):1034–1045. https://doi.org/10.1001/jama.2016.0299
Hinnen D (2017) Glucagon-like peptide 1 receptor agonists for type 2 diabetes. Diabetes Spectr 30(3):202–210. https://doi.org/10.2337/ds16-0026
D'Alessio D (2016) Is GLP-1 a hormone: whether and when? J Diabetes Investig. https://doi.org/10.1111/jdi.12466
Cao Y, Gao W, Jusko WJ (2012) Pharmacokinetic/pharmacodynamic modeling of GLP-1 in healthy rats. Pharm Res 29(4):1078–1086. https://doi.org/10.1007/s11095-011-0652-x
Jansen TJP, van Lith SAM, Boss M, Brom M, Joosten L, Béhé M, Buitinga M, Gotthardt M (2019) Exendin-4 analogs in insulinoma theranostics. J Labelled Comp Radiopharm 62(10):656–672. https://doi.org/10.1002/jlcr.3750
Athauda D, Foltynie T (2018) Protective effects of the GLP-1 mimetic exendin-4 in Parkinson's disease. Neuropharmacology 136(Pt B):260–270. https://doi.org/10.1016/j.neuropharm.2017.09.023
Kang H-M, Sohn I, Jung J, Jeong J-W, Park C (2015) Exendin-4 protects hindlimb ischemic injury by inducing angiogenesis. Biochem Biophys Res Commun 465(4):758–763. https://doi.org/10.1016/j.bbrc.2015.08.080
Ventorp F, Bay-Richter C, Nagendra AS et al (2017) Exendin-4 Treatment Improves LPS-Induced Depressive-Like Behavior Without Affecting Pro-Inflammatory Cytokines. J Parkinsons Dis 7(2):263–273. https://doi.org/10.3233/JPD-171068
Abdelwahed OM, Tork OM, Gamal El Din MM, Rashed L, Zickri M (2018) Effect of glucagon-like peptide-1 analogue; Exendin-4, on cognitive functions in type 2 diabetes mellitus; possible modulation of brain derived neurotrophic factor and brain Visfatin. Brain Res Bull 139:67–80. https://doi.org/10.1016/j.brainresbull.2018.02.002
Ozkok A, Yildiz A (2018) Endothelial progenitor cells and kidney diseases. Kidney Blood Press Res 43(3):701–718. https://doi.org/10.1159/000489745
Yao Y, Li Y, Song Q, Hu C, Xie W, Xu C, Chen Q, Wang QK (2019) Angiogenic factor AGGF1-primed endothelial progenitor cells repair vascular defect in diabetic mice. Diabetes 68(8):1635–1648. https://doi.org/10.2337/db18-1178
Hu L, Dai S-C, Luan X, Chen J, Cannavicci A (2018) Dysfunction and therapeutic potential of endothelial progenitor cells in diabetes mellitus. J Clin Med Res 10(10):752–757. https://doi.org/10.14740/jocmr3581w
Kadam S, Kanitkar M, Dixit K, Deshpande R, Seshadri V, Kale V (2018) Curcumin reverses diabetes-induced endothelial progenitor cell dysfunction by enhancing MnSOD expression and activity in vitro and in vivo. J Tissue Eng Regen Med 12(7):1594–1607. https://doi.org/10.1002/term.2684
Yang Y, Zhou Y, Wang Y et al (2020) Exendin-4 regulates endoplasmic reticulum stress to protect endothelial progenitor cells from high-glucose damage. Mol Cell Probes. https://doi.org/10.1016/j.mcp.2020.101527
Hu J, Wang S, Xiong Z et al (1864) Sun D (2018) Exosomal Mst1 transfer from cardiac microvascular endothelial cells to cardiomyocytes deteriorates diabetic cardiomyopathy. Biochim Biophys Acta Mol Basis Dis 11:3639–3649. https://doi.org/10.1016/j.bbadis.2018.08.026
Qian N, Li X, Wang X, Wu C, Yin L, Zhi X (2018) Tryptase promotes breast cancer angiogenesis through PAR-2 mediated endothelial progenitor cell activation. Oncol Lett 16(2):1513–1520. https://doi.org/10.3892/ol.2018.8856
Zhao Y, Tan Y, Xi S et al (2013) A novel mechanism by which SDF-1β protects cardiac cells from palmitate-induced endoplasmic reticulum stress and apoptosis via CXCR7 and AMPK/p38 MAPK-mediated interleukin-6 generation. Diabetes 62(7):2545–2558. https://doi.org/10.2337/db12-1233
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods (San Diego, Calif) 25(4):402–408. https://doi.org/10.1006/meth.2001.1262
Bai C, Hou L, Zhang M, Pu Y, Guan W, Ma Y (2012) Characterization of vascular endothelial progenitor cells from chicken bone marrow. BMC Vet Res 8:54. https://doi.org/10.1186/1746-6148-8-54
Rehman K, Akash MSH, Liaqat A, Kamal S, Qadir MI, Rasul A (2017) Role of Interleukin-6 in development of insulin resistance and type 2 diabetes mellitus. Crit Rev Eukaryot Gene Expr 27(3):229–236. https://doi.org/10.1615/CritRevEukaryotGeneExpr.2017019712
Li H, O'Meara M, Zhang X et al (2019) Ameliorating methylglyoxal-induced progenitor cell dysfunction for tissue repair in diabetes. Diabetes 68(6):1287–1302. https://doi.org/10.2337/db18-0933
Berezin AE (2017) Endothelial progenitor cells dysfunction and impaired tissue reparation: The missed link in diabetes mellitus development. Diabetes Metab Syndr 11(3):215–220. https://doi.org/10.1016/j.dsx.2016.08.007
Chen S, Wang Z, Zhou H, He B, Hu D, Jiang H (2019) Icariin reduces high glucose-induced endothelial progenitor cell dysfunction via inhibiting the p38/CREB pathway and activating the Akt/eNOS/NO pathway. Exp Ther Med 18(6):4774–4780. https://doi.org/10.3892/etm.2019.8132
Zeng Y-C, Peng L-S, Zou L et al (2017) Protective effect and mechanism of lycopene on endothelial progenitor cells (EPCs) from type 2 diabetes mellitus rats. Biomed Pharmacother 92:86–94. https://doi.org/10.1016/j.biopha.2017.05.018
Liu Z, Zhang M, Zhou T, Shen Q, Qin X (2018) Exendin-4 promotes the vascular smooth muscle cell re-differentiation through AMPK/SIRT1/FOXO3a signaling pathways. Atherosclerosis 276:58–66. https://doi.org/10.1016/j.atherosclerosis.2018.07.016
Hussien NI, Sorour SM, El-Kerdasy HI, Abdelrahman BA (2018) The glucagon-like peptide-1 receptor agonist Exendin-4, ameliorates contrast-induced nephropathy through suppression of oxidative stress, vascular dysfunction and apoptosis independent of glycaemia. Clin Exp Pharmacol Physiol 45(8):808–818. https://doi.org/10.1111/1440-1681.12944
Guo Z, Chen R, Zhang F, Ding M, Wang P (2018) Exendin-4 relieves the inhibitory effects of high glucose on the proliferation and osteoblastic differentiation of periodontal ligament stem cells. Arch Oral Biol. https://doi.org/10.1016/j.archoralbio.2018.03.014
Fallahi P, Corrado A, Di Domenicantonio A, Frenzilli G, Antonelli A, Ferrari SM (2016) CXCR3, CXCR5, CXCR6, and CXCR7 in Diabetes. Curr Drug Targets 17(5):515–519
Hao H, Hu S, Chen H et al (2017) Loss of endothelial CXCR7 impairs vascular homeostasis and cardiac remodeling after myocardial infarction: implications for cardiovascular drug discovery. Circulation 135(13):1253–1264. https://doi.org/10.1161/CIRCULATIONAHA.116.023027
Tang Y, Liu J, Yan Y, Fang H, Guo C, Xie R, Liu Q (2018) 1,25-dihydroxyvitamin-D3 promotes neutrophil apoptosis in periodontitis with type 2 diabetes mellitus patients via the p38/MAPK pathway. Medicine (Baltimore) 97(52):e13903. https://doi.org/10.1097/MD.0000000000013903
Tomita T (2010) Immunocytochemical localization of cleaved caspase-3 in pancreatic islets from type 1 diabetic subjects. Islets 2(1):24–29. https://doi.org/10.4161/isl.2.1.10041
Nakagami H, Morishita R, Yamamoto K et al (2001) Phosphorylation of p38 mitogen-activated protein kinase downstream of bax-caspase-3 pathway leads to cell death induced by high D-glucose in human endothelial cells. Diabetes 50(6):1472–1481
Akbari M, Hassan-Zadeh V (2018) IL-6 signalling pathways and the development of type 2 diabetes. Inflammopharmacology 26(3):685–698. https://doi.org/10.1007/s10787-018-0458-0
Pedersen BK (2017) Anti-inflammatory effects of exercise: role in diabetes and cardiovascular disease. Eur J Clin Invest 47(8):600–611. https://doi.org/10.1111/eci.12781
Fan Y, Ye J, Shen F et al (2008) Interleukin-6 stimulates circulating blood-derived endothelial progenitor cell angiogenesis in vitro. J Cereb Blood Flow Metab 28(1):90–98. https://doi.org/10.1038/sj.jcbfm.9600509
Acknowledgements
Not applicable.
Funding
Not applicable.
Author information
Authors and Affiliations
Contributions
YY and YZ substantially contributed to conception and design. YW and XW contributed to data acquisition. LW, TW and AM contributed to data analysis and interpretation. YY and YZ drafted the article or critically revised it for important intellectual content. All authors contributed to final approval of the version to be published. All authors contributed to the agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval and consent to participate
All animal experiments were conducted following the guidelines of China Council on Animal Care and Use. This study has obtained approval from the Committee of Experimental Animals of The First Affiliated Hospital of Xi’an Jiaotong University (approval serial number: XNK20190806). No humans are involved in this study.
Consent for publication
Not applicable.
Additional information
Managed by Antonio Secchi.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yang, Y., Zhou, Y., Wang, Y. et al. Exendin-4 reverses high glucose-induced endothelial progenitor cell dysfunction via SDF-1β/CXCR7–AMPK/p38-MAPK/IL-6 axis. Acta Diabetol 57, 1315–1326 (2020). https://doi.org/10.1007/s00592-020-01551-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-020-01551-3