Abstract
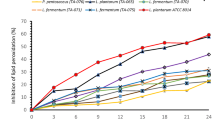
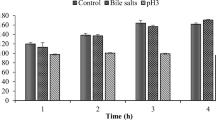
This study aimed to investigate the effects of temperature (4, 85, 121 °C), pH (2, 4, 11), and bile salt (0.3, 0.6, 1%)—as common conditions of gastrointestinal tract or thermal food processing—on antimicrobial activity of bacteriocin-like substances (BLS) obtained from barley sourdough lactic acid bacteria (LAB) against some of food-borne pathogens. In addition, the safety of LAB isolates was also evaluated. The isolates were identified by sequencing of PCR products as Lactobacillus brevis, Lactobacillus curieae, Pediococcus stilesii and Weissella cibaria. At low and high temperatures and pH values and at the presence of bile salt, BLS of the LAB isolates showed remarkable antimicrobial activity against studied indicator bacteria. Furthermore, BLS of L. curieae treated in pH 4 among all pH treatments, BLS of L. brevis and P. stilesii in 1% bile salt among all bile salt treatments, and finally BLS of L. curieae and P. stilesii treated at 4 °C among all temperature treatments were the most effective, respectively, against Aspergillus niger and A. flavus. The safety results demonstrated that consumption of LAB isolates had no adverse effects on general health of mice. Body weight of mice was increased by administration of LAB during 3 weeks, such as control group. Liver enzyme activities of mice treated with the LAB isolates had also insignificant difference with control mice.



Similar content being viewed by others
References
Abnous K, Brooks SPJ, Kwan J, Matias F, Johnson JG, Selinger LB, Thomas M, Kalmokoff M (2009) Diets enriched in oat bran or wheat bran temporally and differentially alter the composition of the fecal community of rats. J Nutr 139(11):2024–2031. https://doi.org/10.3945/jn.109.109470
Alakomi HL, Skytta E, Saarela M, Mattila-Sandholm T, Latva-Kala K, Helander IM (2000) Lactic acid permeabilizes gram-negative bacteria by disrupting the outer membrane. Appl Environ Microbiol 66(5):2001–2005. https://doi.org/10.1128/AEM.66.5.2001-2005.2000
Assefa E, Beyene F, Santhanam A (2008) Effect of temperature and pH on the antimicrobial activity of inhibitory substances produced by lactic acid bacteria isolated from ergo, an Ethiopian traditional fermented milk. Afr J Microbiol Res 2:229–234
Bernardeau M, Vernoux JP, Gueguen M (2002) Safety and efficacy of probiotic lactobacilli in promoting growth in post-weaning Swiss mice. Int J Food Microbiol 77(1-2):19–27. https://doi.org/10.1016/S0168-1605(02)00059-4
Cavicchioli VQ, Camargo AC, Todorov SD, Nero LA (2017) Novel bacteriocinogenic Enterococcus hirae and Pediococcus pentosaceus strains with antilisterial activity isolated from Brazilian artisanal cheese. J Dairy Sci 100(4):2526–2535. https://doi.org/10.3168/jds.2016-12049
Corsetti A, Settanni L, Van Sinderen D (2004) Characterization of bacteriocin-like inhibitory substances (BLIS) from sourdough lactic acid bacteria and evaluation of their in vitro and in situ activity. J Appl Microbiol 96(3):521–534. https://doi.org/10.1111/j.1365-2672.2004.02171.x
Cotter PD, Ross RP, Hill C (2013) Bacteriocins, a viable alternative to antibiotics? Nat Rev Microbiol 11(2):95–105. https://doi.org/10.1038/nrmicro2937
De Vuyst L, Neysens P (2005) The sourdough microflora: biodiversity and metabolic interactions. Trends Food Sci Technol 16(1-3):43–56. https://doi.org/10.1016/j.tifs.2004.02.012
Deegan LH, Cotter PD, Hill C, Ross P (2006) Bacteriocins: biological tools for bio-preservation and shelf-life extension. Int Dairy J 16(9):1058–1071. https://doi.org/10.1016/j.idairyj.2005.10.026
Dicks LM, Botes M (2010) Probiotic lactic acid bacteria in the gastro-intestinal tract: health benefits, safety and mode of action. Benefic Microbes 1(1):11–29. https://doi.org/10.3920/BM2009.0012
Dobson A, Cotter PD, Ross RP, Hill C (2012) Bacteriocin production: a probiotic trait? Appl Environ Microbiol 78(1):1–6. https://doi.org/10.1128/AEM.05576-11
Fayol-Messaoudi D, Berger CN, Coconnier-Polter MH, Liévin-Le Moal V, Servin AL (2005) pH, lactic acid, and non-lactic acid-dependent activities of probiotic Lactobacilli against Salmonella enterica Serovar typhimurium. Appl Environ Microbiol 71(10):6008–6013. https://doi.org/10.1128/AEM.71.10.6008-6013.2005
Giannini EG, Testa R, Savarino V (2005) Liver enzyme alteration: a guide for clinicians. Can Med Assoc J 172:367–379s
Hassan YI, Bullerman LB (2008) Antifungal activity of Lactobacillus paracasei ssp. tolerans isolated from a sourdough bread culture. Int J Food Microbiol 121(1):112–115. https://doi.org/10.1016/j.ijfoodmicro.2007.11.038
Hernandez D, Cardell E, Zarate V (2005) Antimicrobial activity of lactic acid bacteria isolated from Tenerife cheese: initial characterization of Plantaricin TF711, a bacteriocin-like substance produced by Lactobacillus plantarum TF711. J Appl Microb 99(1):77–84. https://doi.org/10.1111/j.1365-2672.2005.02576.x
Jenssen H, Hamill P, Hancock REW (2006) Peptide antimicrobial agents. Clin Microbiol Rev 19(3):491–511. https://doi.org/10.1128/CMR.00056-05
Jorgensen JH, Turnidge JD (2015) Susceptibility test methods: dilution and disk diffusion methods. In: Jorgensen JM, Pfaller MA, Carroll KC, Funke G, Landry ML, Richter SS, Warnock DW (eds) Manual of clinical microbiology, 11rd edn. American Society of Microbiology, New York, pp 1253–1273. https://doi.org/10.1128/9781555817381.ch71
Juarez Tomás MS, Bru E, Wiese B, de Ruiz Holgado AA, Nader-Macías ME (2002) Influence of pH, temperature and culture media on the growth and bacteriocin production by vaginal Lactobacillus salivarius CRL 1328. J Appl Microbiol 93(4):714–724. https://doi.org/10.1046/j.1365-2672.2002.01753.x
Kaur K, Andrew LC, Wishart DS, Vederas JC (2004) Dynamic relationships among type IIa bacteriocins: temperature effects on antimicrobial activity and on structure of the C-terminal amphipathic α helix as a receptor-binding region. Biochemist 43(28):9009–9020. https://doi.org/10.1021/bi036018e
Marianelli C, Cifani N, Pasquali P (2010) Evaluation of antimicrobial activity of probiotic bacteria against Salmonella enterica subsp. enterica Serovar typhimurium 1344 in a common medium under different environmental conditions. Res Microbiol 161(8):673–680. https://doi.org/10.1016/j.resmic.2010.06.007
Messens W, De Vuyst L (2002) Inhibitory substances produced by Lactobacilli isolated from sourdoughs: a review. Int J Food Microbiol 72(1-2):31–43. https://doi.org/10.1016/S0168-1605(01)00611-0
Messens W, Neysens P, Vansieleghem W, Vanderhoeven J, De Vuyst L (2002) Modeling growth and bacteriocin production by Lactobacillus amylovorus DCE 471 in response to temperature and pH values used for sourdough fermentations. Appl Environ Microbiol 68(3):1431–1435. https://doi.org/10.1128/AEM.68.3.1431-1435.2002
Neysens P, Messens W, De Vuyst L (2003) Effect of sodium chloride on growth and bacteriocin production by Lactobacillus amylovorus DCE 471. Int J Food Microbiol 88(1):29–39. https://doi.org/10.1016/S0168-1605(03)00079-5
Organization for Economic Co-operation and Development: OECD (1999) Guidance document on humane endpoints for experimental animals used in safety evaluation studies. Paris
Pavan S, Desreumaux P, Mercenier A (2003) Use of mouse models to evaluate the persistence, safety, and immune modulation capacities of lactic acid bacteria. Clin Diagn Lab Immunol 10(4):696–701
Rodriguez E, Martinez MI, Horn N, Dodd HM (2003) Heterologous production of bacteriocins by lactic acid bacteria. Int J Food Microbiol 80(2):101–116. https://doi.org/10.1016/S0168-1605(02)00153-8
Sadeghi A, Raeisi M, Ebrahimi M, Sadeghi B (2016) Antifungal activity of Pediococcus pentosaceus isolated from whole barley sourdough. J Food Qual Hazards Control 3:30–36
Sadeghi A, Raeisi M, Ebrahimi M (2017) Sourdough: functional feathers and healthy benefits. Golestan University of Medical Sciences Press, Gorgan (ISSN 97860095473-33, in Persian)
Schnürer J, Magnusson J (2005) Antifungal lactic acid bacteria as biopreservatives. Trends Food Sci Technol 16(1-3):70–78. https://doi.org/10.1016/j.tifs.2004.02.014
Sharma N, Gupta A, Gautam N (2014) Characterization of bacteriocin like inhibitory substance produced by a new strain Brevibacillus borstelensis AG1 isolated from ‘Marcha’. Braz J Microbiol 45(3):1007–1015. https://doi.org/10.1590/S1517-83822014000300033
Ullah N, Wang X, Wu J, Guo Y, Ge H, Li T, Khan S, Li Z, Feng X (2017) Purification and primary characterization of a novel bacteriocin, LiN333, from Lactobacillus casei, an isolate from a Chinese fermented food. LWT Food Sci Technol 84:867–875. https://doi.org/10.1016/j.lwt.2017.04.056
Vogelmann SA, Hertel C (2011) Impact of ecological factors on the stability of microbial associations in sourdough fermentation. Food Microbiol 28(3):583–589. https://doi.org/10.1016/j.fm.2010.11.010
Wang H, Yan Y, Wang J, Zhang H, Qi W (2012) Production and characterization of antifungal compounds produced by Lactobacillus plantarum IMAU10014. PLoS One 7(1):e29452. https://doi.org/10.1371/journal.pone.0029452
Yang E, Fan L, Jiang Y, Doucette C, Fillmore S (2012) Antimicrobial activity of bacteriocin-producing lactic acid bacteria isolated from cheeses and yogurts. AMB Express 2(1):48. https://doi.org/10.1186/2191-0855-2-48
Zacharof MP, Lovitt RW (2012) Bacteriocins produced by lactic acid bacteria, a review article. APCBEE Procedia 2:50–56. https://doi.org/10.1016/j.apcbee.2012.06.010
Zannini E, Garofalo C, Aquilanti L, Santarelli S, Silvestri G, Clementi F (2009) Microbiological and technological characterization of sourdoughs destined for bread-making with barley flour. Food Microbiol 26(7):744–753. https://doi.org/10.1016/j.fm.2009.07.014
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sadeghi, A., Raeisi, M., Ebrahimi, M. et al. Effects of temperature, pH, and bile salt on antimicrobial activity of bacteriocin-like substances obtained from barley sourdough LAB. Comp Clin Pathol 27, 611–619 (2018). https://doi.org/10.1007/s00580-017-2635-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-017-2635-8