Abstract
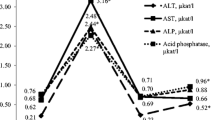
The aim of this study was to determine the protective action of silymarin on mebendazole-induced hepatotoxicity in cats. Twenty five healthy cats were randomly allotted into five equal groups. Cats in group A were given mebendazole (single dose 200 mg⁄kg, p.o.); group B consisted of cats that received silymarin (single dose 30 mg⁄kg, p.o.) concurrent with mebendazole administration; groups C, D and E were treated as group B, but silymarin was administered 2, 12 and 24 h after mebendazole administration, respectively. The serum concentrations of alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), lactate dehydrogenase (LDH) and total and direct bilirubin were measured before mebendazole administration and 2, 12, 24 and 72 h later as indices of liver injury. A single oral administration of mebendazole significantly elevated serum concentrations of ALT, AST, ALP, LDH (in all cases), and total and direct bilirubin in one cat in group A, after 24 h (P < 0.05). In groups B and C, levels of serum enzyme activities and total and direct bilirubin remained within normal values, but in group D, levels of serum enzyme activity (in four cases) were higher than normal values and total and direct bilirubin remained within the normal range. In group E, levels of serum enzyme activities (in all cats) and total and direct bilirubin (in one cat) were higher than normal values. In conclusion, silymarin can protect liver tissue against oxidative stress in cats with mebendazole intoxication particularly in the first 2 h after exposure.




Similar content being viewed by others
References
Avizeh R, Najafzadeh H, Razi Jalali M, Shirali S (2010) Evaluation of prophylactic and therapeutic effects of silymarin and N-acetylcysteine in acetaminophen-induced hepatotoxicity in cats. J Vet Pharmacol Ther 33(1):95–99
Bircher A (2007) Approach to the patient with a drug hypersensitivity reaction—clinical perspectives. In: Pichler WJ (ed) drug hypersensitivity. Basel, Karger, pp 352–365
Boothe DM (2001) Small animal clinical pharmacology and therapeutics, 1st edn. W.B. Saunders Co, Philadelphia, pp 444–538
Cotler S, Gustafson JH, Colburn WA (2006) Pharmacokinetics of diazepam and nordiazepam in the cat. J Pharm Sci 73(3):348–351
Das SK, Vasudevan DM (2006) Protective effects of silymarin, a milk thistle (Silybium marianum) derivative on ethanol-induced oxidative stress in liver. Indian J Biochem Biophys 43:306–311
EMEA, The European Agency for the Evaluation of Medical Products Veterinary Medicines Evaluation Unit (2001) EMEA/MRL/625/99-FINAL July 1999. http://www.emea.eu.int Seven West ferry Circus, Canary Wharf, London, UK
Hsu WH (2008) Handbook of veterinary pharmacology, 1st edn. Wiley-Blackwell, Iowa, pp 84–112
Janssen-Cilag P (2005) Information for health professionals. Ltd. Newmarket, Auckland
Kahn CM (2007) The Merck/Merial manual for pet health. Home Edition. 1st edn. pp 77–126
Kalendera Y, Yelb M, Kalenderb S (2005) Doxorubicin hepatotoxicity and hepatic free radical metabolism in rats. Toxicology 209(1):39–45
Kayne SB, Jepson MH (2004) Veterinary Pharmacy. 1st edn. Publications division of the Royal Pharmaceutical Society of Great Britain, pp 343–423
Lappin MR (2001) Feline internal medicine secrets, 2nd edn. Hanley Belfus Inc, Philadelphia, pp 158–161
Maddison J, Page S, Church D (2002) Small Animal Clinical Pharmacology. 1st edn. W.B. Saunders Co, Harcourt Publishers, pp 106–339
McConkey SE, Grant DM, Cribb AE (2009) The role of para-aminophenol in acetaminophen-induced methemoglobinemia in dogs and cats. J Vet Pharmacol Ther 32(6):585–595
Mira L, Silva M, Manso CF (1994) Scavenging of reactive oxygen species by silibinin dihemisuccinate. Biochem Pharmacol 48:753–759
Oliveira CP, Lopasso FP, Laurindo FR, Leitao RM, Laudanna AA (2001) Protection against liver ischemia-reperfusion injury in rats by silymarin or verapamil. Transplant Proc 33:3010–3314
Paulova J, Dvorak M, Kolouch F, Vanova L, Janeckova L (1990) Verification of the hepatoprotective and therapeutic effect of silymarin in experimental liver injury with tetrachloromethane in dogs. Vet Med (Praha) 35:629–635
Reinhard S, Remy M, Reto B (2001) The use of silymarin in the treatment of liver diseases. Drugs 61:2035–2063
Speare R, Skerratt LF, Berger L, Johnson PM (2004) Toxic effects of mebendazole at high dose on the haematology of red-legged pademelons (Thylogale stigmatica). Aust Vet J 82:300–303
Stokol T, Randolph JF, Nachbar S, Rodi C, Barr SC (1997) Development of bone marrow toxicosis after albendazole administration in a dog and cat. J Am Vet Med Assoc 210:1753–1756
Sumathy T, Subramanian S, Govindasamy S, Balakrishna K, Veluchamy G (2001) Protective role of Bacopa monniera on morphine induced hepatotoxicity in rats. Phytother Res 15:643–645
Tilley LP, Smith FWK (2005) The 5-minute veterinary consult, 3rd edn. Lippincott Williams and Wilkins, Maryland, pp 1325–1326
Valenzuela A, Garrido A (1994) Biochemical bases of the pharmacological action of the flavonoid silymarin and of its structural isomer silibinin. Biol Res 27:105–112
Varzi HN, Esmailzadeh S, Morovvati H, Avizeh R, Shahriari A, Givi ME (2007) Effect of silymarin and vitamin E on gentamicin-induced nephrotoxicity in dogs. J Vet Pharmacol Ther 30:477–481
Webster CR, Cooper J (2009) Therapeutic use of cytoprotective agents in canine and feline hepatobiliary disease. Vet Clin North Am Small Anim Pract 39(3):631–652
Wellington K, Jarvis B (2001) Silymarin: a review of its clinical properties in the management of hepatic disorders. Bio Drugs 15:465–489
Acknowledgment
The authors wish to express their gratitude to the research council of Shahid Chamran University of Ahvaz for their financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mosallanejad, B., Avizeh, R., Najafzadeh Varzi, H. et al. Evaluation of prophylactic and therapeutic effects of silymarin on mebendazole-induced hepatotoxicity in cats. Comp Clin Pathol 21, 681–685 (2012). https://doi.org/10.1007/s00580-010-1157-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-010-1157-4