Abstract
Background
Rocuronium bromide (Rb) is a rapid onset, intermediate-acting neuromuscular blocking agent that is suitable for continuous administration. The appropriate rate of rocuronium administration is, however, difficult to determine due to large interindividual differences in sensitivity to rocuronium. The aim of this study was to clarify whether the simulated rocuronium concentration at the time of recovery to %T1 > 0 % after the initial administration of rocuronium is a good indicator of optimal effect-site concentrations during continuous rocuronium administration.
Methods
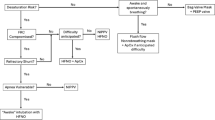
Twenty-one patients were anesthetized with propofol. After induction, Rb 0.6 mg/kg was administered intravenously, and nerve stimulation using the single stimulation mode was conducted every 15 s. When %T1 recovered to >0 % after the initial administration of Rb, the effect-site concentration of rocuronium, calculated by pharmacokinetic simulation with Wierda’s set of parameters, was recorded and defined as the recovery concentration (Rb r.c.). The administration rate of rocuronium was adjusted to maintain the Rb r.c. during surgery. Rb administration was discontinued just before the end of surgery, and the recovery time until %T1 > 25 % was recorded. Plasma Rb concentrations were measured at 1 and 3 h after the initiation of continuous Rb administration.
Result
The mean Rb r.c. was 1.56 ± 0.35 μg/ml, with minimum and maximum values of 1.09 and 2.08 μg/ml, respectively. The %T1 did not increase above 10 % in any of the patients during continuous administration of Rb, and the recovery period to %T1 > 25 % ranged from 9 to 29 min. The effect-site concentrations of Rb calculated with Wierda’s parameters significantly correlated with plasma concentrations (P < 0.01) at both 1 and 3 h after the initial administration of Rb.
Conclusion
The results suggest that our method may be one of the most reliable protocols for the continuous administration of Rb described to date for maintaining suitable muscle relaxation during surgery without excessively prolonged effects.




Similar content being viewed by others
References
Wierda JM, Kleef UW, Lambalk LM, Kloppenburg WD, Agoston S. The pharmacodynamics and pharmacokinetics of Org 9426, a new non-depolarizing neuromuscular blocking agent, in patients anaesthetized with nitrous oxide, halothane and fentanyl. Can J Anaesth. 1991;38:430–5.
Miller DR, Wherrett C, Hull K, Watson J, Legault S. Cumulation characteristics of cisatracurium and rocuronium during continuous infusion. Can J Anaesth. 2000;47:943–9.
Sparr HJ, Wierda JM, Proost JH, Keller C, Khuenl-Brady KS. Pharmacodynamics and pharmacokinetics of rocuronium in intensive care patients. Br J Anaesth. 1997;78:267–73.
Shanks CA, Fragen RJ, Ling D. Continuous intravenous infusion of rocuronium (ORG 9426) in patients receiving balanced, enflurane, or isoflurane anesthesia. Anesthesiology. 1993;78:649–51.
McCoy EP, Mirakhur RK, Maddineni VR, Wierda JM, Proost JH. Pharmacokinetics of rocuronium after bolus and continuous infusion during halothane anaesthesia. Br J Anaesth. 1996;76:29–33.
Xue FS, Tong SY, Liao X, Liu JH, An G, Luo LK. Dose-response and time course of effect of rocuronium in male and female. Anesth Analg. 1997;85:667–71.
Bevan DR, Fiset P, Balendran P, Law-Min JC, Ratcliffe A, Donati F. Pharmacodynamic behavior of rocuronium in the elderly. Can J Anaesth. 1993;40:127–32.
Leykin Y, Pellis T, Lucca M, Lomangino G, Marzano B, Gullo A. The pharmacodynamics effects of rocuronium when dosed according to real body weight or ideal body weight in mobidly obese patients. Anesth Analg. 2004;99:1086–9.
Meyhoff CS, Lund J, Jenstrup MT, Claudius C, Sorensen AM, Viby-Mogensen J, Rasmussen LS. Should dosing of rocuronium in obese patients be based on ideal or corrected body weight? Anesth Analg. 2009;109:787–92.
Dahaba AA, Perelman SI, Moskowitz DM, Bennett HL, Shander A, Xiao Z, Huang L, An G, Bornemann H, Wilfinger G, Hager B, Rehak PH, List WF, Metzler H. Geographic regional differences in rocuronium bromide dose-response relation and time course of action: an overlooked factor in determining recommended dosage. Anesthesiology. 2006;104:950–3.
Fuchs-Buder T, Claudis C, Skovgaard LT, Eriksson LI, Mirakhur RK, Viby-Monngensen J. 8th International Neuromuscular Meeting. Good clinical research practice in pharmacodynamics studies of neuromuscular blocking agents II: the Stockholm revision. Acta Anaesthesiol Scand. 2007;51:789–808.
Valadares de Moraes N, Rocha Lauretti G, Campos de Filgueira G, Carvalho Portes Lopes B, Lanchote VL. Analysis of rocuronium in human plasma by liquid chromatography-tandem mass spectrometry with application in clinical pharmacokinetics. J Pharm Biomed Anal. 2014; 90:180–5.
Kotake Y, Takeda J, Ozaki M, Saeki S, Otagiri T, Kiyama S, Uchimoto R, Iwao Y. Randamized, multicenter study of interaction between Org 9426 (rocuronium bromide) and anesthestic agents in Japanese population (in Japanese with English abstract). Masui (Jpn J Anesthesiol). 2006;55:873–9.
Bissinger U, Schimek F, Lenz G. Postoperative residual paralysis and respiratory status: a comparative study of pancuronium and vecuronium. Physiol Res. 2000;49:455–62.
Hayes AH, Mirakhur RK, Breslin DS, Reid JE, McCourt KC. Postoperative residual block after intermediate-acting neuromuscular blocking drugs. Anaesthesia. 2001;56:312–8.
Kim KS, Lew SH, Cho HY, Cheong MA. Residual paralysis induced by either vecuronium or rocuronium after reversal with pyridostigmine. Anesth Analg. 2002;95:1656–60.
Debaene B, Plaud B, Dilly MP, Donati F. Residual paralysis in the PACU after a single intubating dose of nondepolarizing muscle relaxant with an intermediate duration of action. Anesthesiology. 2003;98:1042–8.
Cammu G, De Witte J, De Veylder J, Bytebier G, Vandeput D, Foubert L, Vandenbroucke G, Deloof T. Postoperative residual paralysis in outpatients versus inpatients. Anesth Analg. 2006;102:426–9.
Eleveld DJ, Kuizenga K, Proost JH, Wierda JM. A temporary decrease in twich response during reversal of rocuronium-induced muscle relaxation with a small dose of sugammadex. Anesth Analg. 2007;104:582–4.
Takagi S, Ozaki M, Iwasaki H, Hatano Y, Takeda J. Effects of sevoflurane and propofol on neuromuscular blocking action of Org 9426 (rocuronium bromide) infused continuously in Japanese patients (in Japanese with English abstract). Masui (Jpn J Anesthesiol). 2006;55:963–7.
Fujimoto M, Tanahira C, Nishi M, Yamamoto T. In non-obese patients, duration of action of rocuronium is directly correlated with body mass index. Can J Anaesth. 2013;60:552–6.
Olkkola KT, Tammisto T. Quantifying the interaction of rocuronium (Org 9426) with etomidate, fentanyl, midazolam, propofol, thiopental, and isoflurane using closed-loop feedback control of rocuronium infusion. Anesth Analg. 1994;78:691–6.
Wulf H, Ledowski T, Linstedt U, Proppe D, Sitzlack D. Neuromuscular blocking effects of rocuronium during desflurane, isoflurane, and sevoflurane anaesthesia. Can J Anaesth. 1998;45:526–32.
Brock M, Kippel K, Nitsche B, Bach A, Martin E, Motsch J. Rocuronium potency and recovery characteristics during steady-state desflurane, sevoflurane, isoflurane or propofol anaesthesia. Br J Anaesth. 2000;84:43–7.
Acknowledgments
This study was supported by a Grant for research from Nihon Kohden.
Author information
Authors and Affiliations
Corresponding author
About this article
Cite this article
Moriyama, T., Matsunaga, A., Nagata, O. et al. Effective method of continuous rocuronium administration based on effect-site concentrations using a pharmacokinetic/pharmacodynamic model during propofol–remifentanil anesthesia. J Anesth 29, 593–599 (2015). https://doi.org/10.1007/s00540-015-1991-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-015-1991-2