Abstract
Background
The Wilms’ tumor 1 (WT1) gene is known to be overexpressed in hepatocellular carcinoma (HCC) and to upregulate tumor growth and oncogenic potential, although the detailed mechanisms remain to be elucidated.
Methods
We identified host genes involved in WT1 gene modulation of human liver cancer cell lines in vitro, and further characterized genes related to apoptosis. Moreover, we evaluated the alteration of genes by WT1 in 40 HCC and 58 non-HCC human liver samples collected at resection.
Results
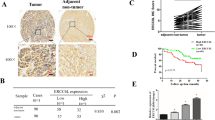
Analysis of the effect of small interfering RNAs-mediated knock-down of WT1 on apoptosis using an annexin V labeling assay, and on modulation of the activity of caspases-3, -8 and -9, indicated that WT1 has an anti-apoptotic role. We identified three apoptosis-related genes that were modulated by WT1; the cellular FLICE-inhibitory proteins (cFLIP) gene was upregulated, and Fas-associated death domain (FADD) and nuclear factor kappa B (NF-κB) were downregulated. Interestingly, knock-down of FADD or NF-κB resulted in the upregulation of WT1, and the expression of cFLIP changed in parallel with WT1 expression. We further evaluated WT1-mediated alteration of genes in HCC and non-HCC human liver samples. Both HCC and non-HCC tissues that expressed relatively high levels of WT1 showed cFLIP overexpression.
Conclusions
WT1 modulates cFLIP, FADD and NF-κB, and has an anti-apoptotic role in HCC. This mechanism of action of WT1 could be related to the tumor growth and oncogenic potential of HCC.






Similar content being viewed by others
Abbreviations
- cFLIP:
-
Cellular FLICE-inhibitory proteins
- FADD:
-
Fas-associated death domain protein
- GAPDH:
-
Glyceraldehyde phosphate dehydrogenase
- HCC:
-
Hepatocellular carcinoma
- NF-κB:
-
Nuclear factor kappa-light-chain-enhancer of activated B cells
- WT1:
-
Wilms’ tumor 1 gene
References
Call KM, Glaser T, Ito CY, Buckler AJ, Pelletier J, Haber DA, et al. Isolation and characterization of a zinc finger polypeptide gene at the human chromosome 11 Wilms’ tumor locus. Cell. 1990;60:509–20.
Scharnhorst V, van der Eb AJ, Jochemsen AG. WT1 proteins functions: functions in growth and differentiation. Gene. 2001;273:141–61.
Yang L, Han Y, Suarez Saiz F, Minden MD. A tumor suppressor and oncogene: the WT1 story. Leukemia. 2007;21:868–76.
Hohenstein P, Hastie ND. The many faces of the Wilms’ tumor gene, WT1. Hum Mol Genet. 2006;15:R196–201.
Tatsumi N, Oji Y, Tsuji N, Tsuda A, Higashio M, Aoyagi S, et al. Wilms’ tumor gene WT1-shRNA as a potent apoptosis-inducing agent for solid tumors. Int J Oncol. 2008;32:701–11.
Ariyaratana S, Loeb DM. The role of the Wilms tumor gene (WT1) in normal and malignant haematopoiesis. Expert Rev Mol Med. 2007;9:1–17.
Morrison AA, Viney RL, Ladomery MR. The post transcriptional roles of WT1, a multifunctional zinc-finger protein. Biochim Biophys Acta. 2008;1785:55–62.
Inoue K, Ogawa H, Sonoda Y, Kimura T, Sakabe H, Oka Y, et al. Aberrant overexpression of the Wilms tumor gene (WT1) in human leukemia. Blood. 1997;89:1405–12.
Loeb DM, Evron E, Patel CB, Sharma PM, Niranjan B, Buluwela L, et al. Wilms’ tumor suppressor gene (WT1) is expressed in primary breast tumors despite tumor-specific promoter methylation. Cancer Res. 2001;61:921–5.
Oji Y, Miyoshi S, Maeda H, Hayashi S, Tamaki H, Nakatsuka S, et al. Overexpression of the Wilms’ tumor gene WT1 in de novo lung cancers. Int J Cancer. 2002;100:297–303.
Netinatsunthorn W, Hanprasertpong J, Dechsukhum C, Leetanaporn R, Geater A. WT1 gene expression as a prognostic marker in advanced serous epithelial ovarian carcinoma: an immunohistochemical study. BMC Cancer. 2006;6:90–8.
Sera T, Hiasa Y, Mashiba T, Tokumoto Y, Hirooka M, Konishi I, et al. Wilms’ tumor 1 gene expression is increased in hepatocellular carcinoma and associated with poor prognosis. Eur J Cancer. 2008;44:600–8.
Berasain C, Herrero JI, Garcia-Trevijano ER, Avila MA, Esteban JI, Mato JM, et al. Expression of Wilms’ tumor suppressor in the liver with cirrhosis: relation to hepatocyte nuclear factor 4 and hepatocellular function. Hepatology. 2003;38:148–57.
Mohammed D, John H, Jerry P. Activation of the wt1 Wilm’s tumor suppressor gene by NF-κBp65. Oncogene. 1998;16:2033–9.
Maheswarnan S, Englert C, Bennett P, Heinrich G, Haber DA. The WT1 gene product stabilizes p53 and inhibits p53-mediated apoptosis. Genes Dev. 1995;9:2143–56.
Perugorria MJ, Castillo J, Ujue Latasa M, Goni S, Segura V, Sangro B, et al. Wilms’ tumor 1 gene expression in hepatocellular carcinoma promotes cell dedifferentiation and resistance to chemotherapy. Cancer Res. 2009;69:1353–67.
Okamoto K, Fujisawa J, Reth M, Yonehara S. Human T-cell leukemia virus type-I oncoprotein Tax inhibits Fas-mediated apoptosis by inducing cellular FLIP through activation of NF-κBp65. Genes Cells. 2006;11:177–91.
Duckett CS. IAP proteins: sticking it to Smac. Biochem J. 2005;385(Pt 1):e1–2.
Silke J, Kratina T, Chu D, Ekert PG, Day CL, Pakusch M, et al. Determination of cell survival by RING-mediated regulation of inhibitor of apoptosis (IAP) protein abundance. Proc Natl Acad Sci USA. 2005;102:16182–7.
Budd RC, Yeh WC, Tschopp J. cFLIP regulation of lymphocyte activation and development. Nat Rev Immunol. 2006;6:196–204.
Kataoka T. The caspase-8 modulator c-FLIP. Crit Rev Immunol. 2005;25:31–58.
Yu JW, Shi Y. FLIP and the death effector domain family. Oncogene. 2008;27:6216–7.
Tuna M, Cavez-Reyes A, Tari AM. HER2/neu increases the expression of Wilms’ tumor 1 (WT1) protein to stimulate S-phase proliferation and inhibit apoptosis in breast cancer cells. Oncogene. 2005;24:1648–52.
Ito K, Oji Y, Tatsumi N, Shimizu S, Kanai Y, Nakazawa T, et al. Antiapoptotic function of 17AA(+)WT1 (Wilms’ tumor gene) isoforms on the intrinsic apoptosis pathway. Oncogene. 2006;25:4217–29.
Scaffidi C, Schmitz I, Krammer PH, Peter ME. The role of c-FLIP in modulation of CD95-induced apoptosis. Biol Chem J. 1999;274:1541–8.
Erwert RD, Winn RK, Harlan JM, Bannerman DD. Shiga-like toxin inhibition of FLICE-like inhibitory protein expression sensitizes endothelial cells to bacterial lipopolysaccharide-induced apoptosis. J Biol Chem. 2002;277:40567–74.
Kreuz S, Siegmund D, Rumpf JJ, Samel D, Leverkus M, Janssen O, et al. NF-κB activation by Fas is mediated through FADD, caspase-8, and RIP and is inhibited by cFLIP. J Cell Biol. 2004;166:369–80.
Chinnaiyan AM, O’Rourke K, Tewari M, Dixit VM. FADD, a novel death domain-containing protein, interacts with the death domain of Fas and initiates apoptosis. Cell. 1995;81:505–12.
Van Antwerp DJ, Martin SJ, Kafri T, Green DR, Verma IM. Suppression of TNF-alpha-induced apoptosis by NF-kappaB. Science. 1996;274:787–9.
Oka Y, Tsuboi A, Taguchi T, Osaki T, Kyo T, Nakajima H, et al. Induction of WT1 (Wilms’ tumor gene)-specific cytotoxic T lymphocytes by WT1 peptide vaccine and the resultant cancer regression. Proc Natl Acad Sci USA. 2004;38:13885–90.
Acknowledgments
We thank Dr. Shigeki Higashiyama (Department of Biochemistry and Molecular Genetics), and Dr. Mamoru Aoto (Department of Physiology) in Ehime University Graduate School of Medicine (Ehime, Japan) and Dr. Keiko Udaka (Department of Immunology, Kochi University School of Medicine, Kochi, Japan) for valuable advice, and also Ms. Satomi Yamanaka, Ms. Chie Takeichi and Ms. Sakiko Inoh (in our department) for valuable technical assistance. We thank Dr. Francis V. Chisari (Department of Immunology and Microbial Science, The Scripps Research Institute, La Jolla, CA, USA) for providing Huh7.5.1 cells, and Dr. Haruo Sugiyama (Department of Functional Diagnostic Science, Graduate School of Medicine, Osaka University, Osaka, Japan) for providing the plasmids encoding WT1 genes. This work was supported in part by a Grant-in-Aid for Scientific Research (Japan Society for the Promotion of Science, KAKENHI 24590980 to Y.H.) and the Program for Enhancing Systematic Education in Graduate School (to K.U.) from the Ministry of Education, Culture, Sports, Science and Technology, Japan, and by a Grant-in-Aid for Scientific Research and Development (to Y.H.) from the Japanese Ministry of Health, Labor and Welfare, Japan.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
535_2012_708_MOESM1_ESM.tif
Supplementary Figure 1. Down- and upregulation of WT1 gene expression in liver cancer cell lines by WT1 siRNA and by a WT1 plasmid, respectively. WT1 mRNA was efficiently knocked-down in all three cell lines (Huh7, Huh7.5.1 and HLE cells) after WT1 siRNA transfection (black bars) compared to control siRNA transfection (white bars) as assessed by real-time RT-PCR (a). Expression of the WT1 protein was also downregulated by WT1 siRNA transfection as assessed by western blotting (b). WT1 mRNA levels remained upregulated for at least 3 days after transfection of the WT1-expressing plasmid (pWT1) compared to cells transfected with a control plasmid (p3.1) (c). Western blotting indicated that WT1 proteins were overexpressed in each cell line following plasmid transfection. (d). The mRNA data are means ± SEM of 4 independent experiments. *p < 0.05 versus control (TIFF 5883 kb)
535_2012_708_MOESM2_ESM.tif
Supplementary Figure 2. Upregulation of WT1 mRNA expression following transfection of plasmids encoding the four splicing variants of WT1 into liver cancer cell lines. Plasmids expressing the four splicing variant forms of WT1 were transfected into the indicated cell lines and WT1 mRNA was then analyzed by RT-PCR 2 days following transfection. Data are indicated as the copy ratio of WT1 mRNA/GAPDH mRNA, and each value represents the fold of the pWT1 (+/+) value, which is normalized as 1.0. Data are means ± SEM of three independent experiments (TIFF 1295 kb)
Rights and permissions
About this article
Cite this article
Uesugi, K., Hiasa, Y., Tokumoto, Y. et al. Wilms’ tumor 1 gene modulates Fas-related death signals and anti-apoptotic functions in hepatocellular carcinoma. J Gastroenterol 48, 1069–1080 (2013). https://doi.org/10.1007/s00535-012-0708-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00535-012-0708-7