Abstract
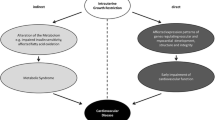
Epidemiological and experimental studies show that the risk of cardiovascular and metabolic diseases at adulthood is inversely related to the weight at birth. Although with less evidence, low birth weight has been suggested to increase the risk of chronic kidney disease (CKD). It is well established that the developmental programming of arterial hypertension and of renal disease involves in particular renal factors, especially nephron endowment, which is reduced in low birth weight and maternal diabetes situations. Experimental studies, especially in rodents, have demonstrated the long-term influence of postnatal nutrition and/or postnatal growth on cardiovascular, metabolic and renal functions, while human data are scarce on this issue. Vascular and renal diseases appear to have a “multihits” origin, with reduced nephron number the initial hit and rapid postnatal growth the second hit. This review addresses the current understanding of the role of the kidney, both as a mechanism and as a target, in the developmental origins of adult disease theory, with a particular focus on the long-term effects of postnatal growth and nutrition.


Similar content being viewed by others
References
Barker DJ, Osmond C, Golding J, Kuh D, Wadsworth ME (1989) Growth in utero, blood pressure in childhood and adult life, and mortality from cardiovascular disease. BMJ 298:564–567
Barker DJ (1990) The fetal and infant origins of adult disease. BMJ 301:1111
Newsome CA, Shiell AW, Fall CH, Phillips DI, Shier R, Law CM (2003) Is birth weight related to later glucose and insulin metabolism?—A systematic review. Diabet Med 20:339–348
Hales CN, Barker DJ, Clark PM, Cox LJ, Fall C, Osmond C, Winter PD (1991) Fetal and infant growth and impaired glucose tolerance at age 64. BMJ 303:1019–1022
Stein CE, Fall CH, Kumaran K, Osmond C, Cox V, Barker DJ (1996) Fetal growth and coronary heart disease in south India. Lancet 348:1269–1273
Curhan GC, Willett WC, Rimm EB, Spiegelman D, Ascherio AL, Stampfer MJ (1996) Birth weight and adult hypertension, diabetes mellitus, and obesity in US men. Circulation 94:3246–3250
Leon DA, Lithell HO, Vagero D, Koupilova I, Mohsen R, Berglund L, Lithell UB, McKeigue PM (1998) Reduced fetal growth rate and increased risk of death from ischaemic heart disease: cohort study of 15 000 Swedish men and women born 1915–29. BMJ 317:241–245
Huxley RR, Shiell AW, Law CM (2000) The role of size at birth and postnatal catch-up growth in determining systolic blood pressure: a systematic review of the literature. J Hypertens 18:815–831
Barker DJ, Eriksson JG, Forsen T, Osmond C (2002) Fetal origins of adult disease: strength of effects and biological basis. Int J Epidemiol 31:1235–1239
Barker DJ (2006) Adult consequences of fetal growth restriction. Clin Obstet Gynecol 49:270–283
Hoy WE, Douglas-Denton RN, Hughson MD, Cass A, Johnson K, Bertram JF (2003) A stereological study of glomerular number and volume: preliminary findings in a multiracial study of kidneys at autopsy. Kidney Int Suppl S31–S37
Lackland DT, Bendall HE, Osmond C, Egan BM, Barker DJ (2000) Low birth weights contribute to high rates of early-onset chronic renal failure in the Southeastern United States. Arch Intern Med 160:1472–1476
Keijzer-Veen MG, Finken MJ, Nauta J, Dekker FW, Hille ET, Frolich M, Wit JM, van der Heijden AJ (2005) Microalbuminuria and lower glomerular filtration rate at young adult age in subjects born very premature and after intrauterine growth retardation. J Am Soc Nephrol 16:2762–2768
Vikse BE, Irgens LM, Leivestad T, Hallan S, Iversen BM (2008) Low birth weight increases risk for end-stage renal disease. J Am Soc Nephrol 19:151–157
Kuzawa CW (2005) Fetal origins of developmental plasticity: are fetal cues reliable predictors of future nutritional environments? Am J Hum Biol 17:5–21
Bateson P (2007) Developmental plasticity and evolutionary biology. J Nutr 137:1060–1062
Gluckman PD, Hanson MA, Cooper C, Thornburg KL (2008) Effect of in utero and early-life conditions on adult health and disease. N Engl J Med 359:61–73
Brenner BM, Chertow GM (1994) Congenital oligonephropathy and the etiology of adult hypertension and progressive renal injury. Am J Kidney Dis 23:171–175
Vehaskari VM, Woods LL (2005) Prenatal programming of hypertension: lessons from experimental models. J Am Soc Nephrol 16:2545–2556
McMillen IC, Robinson JS (2005) Developmental origins of the metabolic syndrome: prediction, plasticity, and programming. Physiol Rev 85:571–633
Simeoni U, Barker DJ (2009) Offspring of diabetic pregnancy: long-term outcomes. Semin Fetal Neonatal Med 14:119–124
Lelièvre-Pégorier M, Merlet-Benichou C (2000) The number of nephrons in the mammalian kidney: environmental influences play a determining role. Exp Nephrol 8:63–65
Merlet-Benichou C, Gilbert T, Vilar J, Moreau E, Freund N, Lelièvre-Pégorier M (1999) Nephron number: variability is the rule. Causes and consequences. Lab Invest 79:515–527
Rodriguez MM, Gomez AH, Abitbol CL, Chandar JJ, Duara S, Zilleruelo GE (2004) Histomorphometric analysis of postnatal glomerulogenesis in extremely preterm infants. Pediatr Dev Pathol 7:17–25
Magaton A, Gil FZ, Casarini DE, Cavanal Mde F, Gomes GN (2007) Maternal diabetes mellitus—early consequences for the offspring. Pediatr Nephrol 22:37–43
Nehiri T, Duong Van Huyen JP, Viltard M, Fassot C, Heudes D, Freund N, Deschenes G, Houillier P, Bruneval P, Lelièvre-Pégorier M (2008) Exposure to maternal diabetes induces salt-sensitive hypertension and impairs renal function in adult rat offspring. Diabetes 57:2167–2175
Simeoni U, Zetterstrom R (2005) Long-term circulatory and renal consequences of intrauterine growth restriction. Acta Paediatr 94:819–824
Tauzin L, Rossi P, Giusano B, Gaudart J, Boussuges A, Fraisse A, Simeoni U (2006) Characteristics of arterial stiffness in very low birth weight premature infants. Pediatr Res 60:592–596
Martin H, Gazelius B, Norman M (2000) Impaired acetylcholine-induced vascular relaxation in low birth weight infants: implications for adult hypertension? Pediatr Res 47:457–462
Kistner A, Jacobson L, Jacobson SH, Svensson E, Hellstrom A (2002) Low gestational age associated with abnormal retinal vascularization and increased blood pressure in adult women. Pediatr Res 51:675–680
Lamireau D, Nuyt AM, Hou X, Bernier S, Beauchamp M, Gobeil F Jr, Lahaie I, Varma DR, Chemtob S (2002) Altered vascular function in fetal programming of hypertension. Stroke 33:2992–2998
Pladys P, Sennlaub F, Brault S, Checchin D, Lahaie I, Lê NL, Bibeau K, Cambonie G, Abran D, Brochu M, Thibault G, Hardy P, Chemtob S, Nuyt AM (2005) Microvascular rarefaction and decreased angiogenesis in rats with fetal programming of hypertension associated with exposure to a low-protein diet in utero. Am J Physiol Regul Integr Comp Physiol 289:R1580–R1588
Franco M, Martinez F, Rodriguez-Iturbe B, Johnson RJ, Santamaria J, Montoya A, Nepomuceno T, Bautista R, Tapia E, Herrera-Acosta J (2006) Angiotensin II, interstitial inflammation, and the pathogenesis of salt-sensitive hypertension. Am J Physiol Renal Physiol 291:F1281–F1287
Ligi I, Grandvuillemin I, Andres V, Dignat-George F, Simeoni U (2010) Low birth weight infants and the developmental programming of hypertension: a focus on vascular factors. Semin Perinatol 34:188–192
Rasch R, Skriver E, Woods LL (2004) The role of the RAS in programming of adult hypertension. Acta Physiol Scand 181:537–542
Yzydorczyk C, Comte B, Cambonie G, Lavoie JC, Germain N, Ting Shun Y, Wolff J, Deschepper C, Touyz RM, Lelièvre-Pégorier M, Nuyt AM (2008) Neonatal oxygen exposure in rats leads to cardiovascular and renal alterations in adulthood. Hypertension 52:889–895
Brenner BM, Garcia DL, Anderson S (1988) Glomeruli and blood pressure. Less of one, more the other? Am J Hypertens 1:335–347
Van Huyen JP, Viltard M, Nehiri T, Freund N, Belair MF, Martinerie C, Lelongt B, Bruneval P, Lelièvre-Pégorier M (2007) Expression of matrix metalloproteinases MMP-2 and MMP-9 is altered during nephrogenesis in fetuses from diabetic rats. Lab Invest 87:680–689
Tran S, Chen YW, Chenier I, Chan JS, Quaggin S, Hebert MJ, Ingelfinger JR, Zhang SL (2008) Maternal diabetes modulates renal morphogenesis in offspring. J Am Soc Nephrol 19:943–952
Nyengaard JR, Bendtsen TF (1992) Glomerular number and size in relation to age, kidney weight, and body surface in normal man. Anat Rec 232:194–201
Manalich R, Reyes L, Herrera M, Melendi C, Fundora I (2000) Relationship between weight at birth and the number and size of renal glomeruli in humans: a histomorphometric study. Kidney Int 58:770–773
Hughson M, Farris AB 3rd, Douglas-Denton R, Hoy WE, Bertram JF (2003) Glomerular number and size in autopsy kidneys: the relationship to birth weight. Kidney Int 63:2113–2122
Hoy W, Kondalsamy-Chennakesavan S, Scheppingen J, Sharma S (2005) Kidney and related chronic disease profiles and risk factors in three remote Australian Aboriginal communities. Adv Chronic Kidney Dis 12:64–70
Konje JC, Bell SC, Morton JJ, de Chazal R, Taylor DJ (1996) Human fetal kidney morphometry during gestation and the relationship between weight, kidney morphometry and plasma active renin concentration at birth. Clin Sci (Lond) 91:169–175
Hinchliffe SA, Lynch MR, Sargent PH, Howard CV, Van Velzen D (1992) The effect of intrauterine growth retardation on the development of renal nephrons. Br J Obstet Gynaecol 99:296–301
Boubred F, Vendemmia M, Garcia-Meric P, Buffat C, Millet V, Simeoni U (2006) Effects of maternally administered drugs on the fetal and neonatal kidney. Drug Saf 29:397–419
Buffat C, Boubred F, Mondon F, Chelbi ST, Feuerstein JM, Lelièvre-Pégorier M, Vaiman D, Simeoni U (2007) Kidney gene expression analysis in a rat model of intrauterine growth restriction reveals massive alterations of coagulation genes. Endocrinology 148:5549–5557
Boubred F, Daniel L, Buffat C, Feuerstein JM, Tsimaratos M, Oliver C, Dignat-George F, Lelièvre-Pégorier M, Simeoni U (2009) Early postnatal overfeeding induces early chronic renal dysfunction in adult male rats. Am J Physiol Renal Physiol 297:F943–F951
Welham SJ, Wade A, Woolf AS (2002) Protein restriction in pregnancy is associated with increased apoptosis of mesenchymal cells at the start of rat metanephrogenesis. Kidney Int 61:1231–1242
Welham SJ, Riley PR, Wade A, Hubank M, Woolf AS (2005) Maternal diet programs embryonic kidney gene expression. Physiol Genomics 22:48–56
Woods LL, Ingelfinger JR, Nyengaard JR, Rasch R (2001) Maternal protein restriction suppresses the newborn renin-angiotensin system and programs adult hypertension in rats. Pediatr Res 49:460–467
Segar JL, Bedell K, Page WV, Mazursky JE, Nuyt AM, Robillard JE (1995) Effect of cortisol on gene expression of the renin-angiotensin system in fetal sheep. Pediatr Res 37:741–746
Wintour EM, Moritz KM, Johnson K, Ricardo S, Samuel CS, Dodic M (2003) Reduced nephron number in adult sheep, hypertensive as a result of prenatal glucocorticoid treatment. J Physiol 549:929–935
Vehaskari VM, Stewart T, Lafont D, Soyez C, Seth D, Manning J (2004) Kidney angiotensin and angiotensin receptor expression in prenatally programmed hypertension. Am J Physiol Renal Physiol 287:F262–F267
Dickinson H, Walker DW, Wintour EM, Moritz K (2007) Maternal dexamethasone treatment at midgestation reduces nephron number and alters renal gene expression in the fetal spiny mouse. Am J Physiol Regul Integr Comp Physiol 292:R453–R461
Figueroa JP, Rose JC, Massmann GA, Zhang J, Acuna G (2005) Alterations in fetal kidney development and elevations in arterial blood pressure in young adult sheep after clinical doses of antenatal glucocorticoids. Pediatr Res 58:510–515
Singh RR, Moritz KM, Bertram JF, Cullen-McEwen LA (2007) Effects of dexamethasone exposure on rat metanephric development: in vitro and in vivo studies. Am J Physiol Renal Physiol 293:F548–F554
Abdel-Hakeem AK, Henry TQ, Magee TR, Desai M, Ross MG, Mansano RZ, Torday JS, Nast CC (2008) Mechanisms of impaired nephrogenesis with fetal growth restriction: altered renal transcription and growth factor expression. Am J Obstet Gynecol 199:e251–e257
Vilar J, Lalou C, Duong VH, Charrin S, Hardouin S, Raulais D, Merlet-Benichou C, Lelièvre-Pégorier M (2002) Midkine is involved in kidney development and in its regulation by retinoids. J Am Soc Nephrol 13:668–676
Gilbert T, Merlet-Benichou C (2000) Retinoids and nephron mass control. Pediatr Nephrol 14:1137–1144
Woods LL, Ingelfinger JR, Rasch R (2005) Modest maternal protein restriction fails to program adult hypertension in female rats. Am J Physiol Regul Integr Comp Physiol 289:R1131–R1136
Vehaskari VM, Aviles DH, Manning J (2001) Prenatal programming of adult hypertension in the rat. Kidney Int 59:238–245
Celsi G, Kistner A, Aizman R, Eklof AC, Ceccatelli S, de Santiago A, Jacobson SH (1998) Prenatal dexamethasone causes oligonephronia, sodium retention, and higher blood pressure in the offspring. Pediatr Res 44:317–322
Woods LL, Weeks DA, Rasch R (2004) Programming of adult blood pressure by maternal protein restriction: role of nephrogenesis. Kidney Int 65:1339–1348
Moritz KM, Mazzuca MQ, Siebel AL, Mibus A, Arena D, Tare M, Owens JA, Wlodek ME (2009) Uteroplacental insufficiency causes a nephron deficit, modest renal insufficiency but no hypertension with ageing in female rats. J Physiol 587:2635–2646
Schmidt IM, Chellakooty M, Boisen KA, Damgaard IN, Mau Kai C, Olgaard K, Main KM (2005) Impaired kidney growth in low-birth-weight children: distinct effects of maturity and weight for gestational age. Kidney Int 68:731–740
Drougia A, Giapros V, Hotoura E, Papadopoulou F, Argyropoulou M, Andronikou S (2009) The effects of gestational age and growth restriction on compensatory kidney growth. Nephrol Dial Transplant 24:142–148
Moritz KM, Singh RR, Probyn ME, Denton KM (2009) Developmental programming of a reduced nephron endowment: more than just a baby's birth weight. Am J Physiol Renal Physiol 296:F1–F9
Schreuder MF, Nyengaard JR, Remmers F, van Wijk JA, Delemarre-van de Waal HA (2006) Postnatal food restriction in the rat as a model for a low nephron endowment. Am J Physiol Renal Physiol 291:F1104–F1107
Boubred F, Buffat C, Feuerstein JM, Daniel L, Tsimaratos M, Oliver C, Lelièvre-Pégorier M, Simeoni U (2007) Effects of early postnatal hypernutrition on nephron number and long-term renal function and structure in rats. Am J Physiol Renal Physiol 293:F1944–F1949
Mansano R, Desai M, Garg A, Chol G, Ross M (2007) Enhanced nephrogenesis in offspring of water-restricted rat dams. Am J Obstet Gynecol 196:e481–e486
Barker DJ, Bull AR, Osmond C, Simmonds SJ (1990) Fetal and placental size and risk of hypertension in adult life. BMJ 301:259–262
Curhan GC, Chertow GM, Willett WC, Spiegelman D, Colditz GA, Manson JE, Speizer FE, Stampfer MJ (1996) Birth weight and adult hypertension and obesity in women. Circulation 94:1310–1315
Law CM, de Swiet M, Osmond C, Fayers PM, Barker DJ, Cruddas AM, Fall CH (1993) Initiation of hypertension in utero and its amplification throughout life. BMJ 306:24–27
Irving RJ, Belton NR, Elton RA, Walker BR (2000) Adult cardiovascular risk factors in premature babies. Lancet 355:2135–2136
Singhal A, Cole TJ, Lucas A (2001) Early nutrition in preterm infants and later blood pressure: two cohorts after randomised trials. Lancet 357:413–419
Keijzer-Veen MG, Finken MJ, Nauta J, Dekker FW, Hille ET, Frolich M, Wit JM, van der Heijden AJ (2005) Is blood pressure increased 19 years after intrauterine growth restriction and preterm birth? A prospective follow-up study in The Netherlands. Pediatrics 116:725–731
Langley-Evans SC, Phillips GJ, Jackson AA (1994) In utero exposure to maternal low protein diets induces hypertension in weanling rats, independently of maternal blood pressure changes. Clin Nutr 13:319–324
Symonds ME, Budge H (2009) Nutritional models of the developmental programming of adult health and disease. Proc Nutr Soc 68:173–178
Singh RR, Denton KM, Bertram JF, Jefferies AJ, Head GA, Lombardo P, Schneider-Kolsky M, Moritz KM (2009) Development of cardiovascular disease due to renal insufficiency in male sheep following fetal unilateral nephrectomy. J Hypertens 27:386–396
Woods LL (1999) Neonatal uninephrectomy causes hypertension in adult rats. Am J Physiol 276:R974–R978
Grond J, Beukers JY, Schilthuis MS, Weening JJ, Elema JD (1986) Analysis of renal structural and functional features in two rat strains with a different susceptibility to glomerular sclerosis. Lab Invest 54:77–83
Kreutz R, Kovacevic L, Schulz A, Rothermund L, Ketteler M, Paul M (2000) Effect of high NaCl diet on spontaneous hypertension in a genetic rat model with reduced nephron number. J Hypertens 18:777–782
Thorner PS, Arbus GS, Celermajer DS, Baumal R (1984) Focal segmental glomerulosclerosis and progressive renal failure associated with a unilateral kidney. Pediatrics 73:806–810
Zucchelli P, Cagnoli L (1985) Proteinuria and hypertension after unilateral nephrectomy. Lancet 2:212
Rugiu C, Oldrizzi L, Lupo A, Valvo E, Loschiavo C, Tessitore N, Gammaro L, Ortalda V, Fabris A, Panzetta G (1986) Clinical features of patients with solitary kidneys. Nephron 43:10–15
Wikstad I, Celsi G, Larsson L, Herin P, Aperia A (1988) Kidney function in adults born with unilateral renal agenesis or nephrectomized in childhood. Pediatr Nephrol 2:177–182
Wikstad I, Celsi G, Larsson L, Herin P, Aperia A (2001) Ambulatory blood pressure monitoring in children with a solitary kidney—a comparison between unilateral renal agenesis and uninephrectomy. Blood Press Monit 6:263–267
Keller G, Zimmer G, Mall G, Ritz E, Amann K (2003) Nephron number in patients with primary hypertension. N Engl J Med 348:101–108
Hughson MD, Douglas-Denton R, Bertram JF, Hoy WE (2006) Hypertension, glomerular number, and birth weight in African Americans and white subjects in the Southeastern United States. Kidney Int 69:671–678
Gossmann J, Wilhelm A, Kachel HG, Jordan J, Sann U, Geiger H, Kramer W, Scheuermann EH (2005) Long-term consequences of live kidney donation follow-up in 93% of living kidney donors in a single transplant center. Am J Transplant 5:2417–2424
Bidani AK, Mitchell KD, Schwartz MM, Navar LG, Lewis EJ (1990) Absence of glomerular injury or nephron loss in a normotensive rat remnant kidney model. Kidney Int 38:28–38
Griffin KA, Picken MM, Churchill M, Churchill P, Bidani AK (2000) Functional and structural correlates of glomerulosclerosis after renal mass reduction in the rat. J Am Soc Nephrol 11:497–506
Hoppe CC, Evans RG, Moritz KM, Cullen-McEwen LA, Fitzgerald SM, Dowling J, Bertram JF (2007) Combined prenatal and postnatal protein restriction influences adult kidney structure, function, and arterial pressure. Am J Physiol Regul Integr Comp Physiol 292:R462–R469
Brenner BM, Meyer TW, Hostetter TH (1982) Dietary protein intake and the progressive nature of kidney disease: the role of hemodynamically mediated glomerular injury in the pathogenesis of progressive glomerular sclerosis in aging, renal ablation, and intrinsic renal disease. N Engl J Med 307:652–659
Nenov VD, Taal MW, Sakharova OV, Brenner BM (2000) Multi-hit nature of chronic renal disease. Curr Opin Nephrol Hypertens 9:85–97
Bagby SP (2007) Maternal nutrition, low nephron number, and hypertension in later life: pathways of nutritional programming. J Nutr 137:1066–1072
Vehaskari VM (2007) Developmental origins of adult hypertension: new insights into the role of the kidney. Pediatr Nephrol 22:490–495
Sahajpal V, Ashton N (2003) Renal function and angiotensin AT1 receptor expression in young rats following intrauterine exposure to a maternal low-protein diet. Clin Sci (Lond) 104:607–614
McMullen S, Langley-Evans SC (2005) Maternal low-protein diet in rat pregnancy programs blood pressure through sex-specific mechanisms. Am J Physiol Regul Integr Comp Physiol 288:R85–R90
Shaltout HA, Figueroa JP, Rose JC, Dl D, Chapell MC (2009) Alterations in circulatory and renal angiotensin-converting enzyme and angiotensin-converting enzyme 2 in fetal programmed hypertension. Hypertension 53:404–408
Sherman RC, Langley-Evans SC (1998) Early administration of angiotensin-converting enzyme inhibitor captopril, prevents the development of hypertension programmed by intrauterine exposure to a maternal low-protein diet in the rat. Clin Sci (Lond) 94:373–381
Sherman RC, Langley-Evans SC (2000) Antihypertensive treatment in early postnatal life modulates prenatal dietary influences upon blood pressure in the rat. Clin Sci (Lond) 98:269–275
Manning J, Vehaskari VM (2005) Postnatal modulation of prenatally programmed hypertension by dietary Na and ACE inhibition. Am J Physiol Regul Integr Comp Physiol 288:R80–R84
Brandon AE, Boyce AC, Lumbers ER, Gibson KJ (2009) Maternal renal dysfunction in sheep is associated with salt insensitivity in female offspring. J Physiol 587:261–270
Bertram C, Trowern AR, Copin N, Jackson AA, Whorwood CB (2001) The maternal diet during pregnancy programs altered expression of the glucocorticoid receptor and type 2 11beta-hydroxysteroid dehydrogenase: potential molecular mechanisms underlying the programming of hypertension in utero. Endocrinology 142:2841–2853
Manning J, Beutler K, Knepper MA, Vehaskari VM (2002) Upregulation of renal BSC1 and TSC in prenatally programmed hypertension. Am J Physiol Renal Physiol 283:F202–F206
Alexander BT (2003) Placental insufficiency leads to development of hypertension in growth-restricted offspring. Hypertension 41:457–462
Stewart T, Jung FF, Manning J, Vehaskari VM (2005) Kidney immune cell infiltration and oxidative stress contribute to prenatally programmed hypertension. Kidney Int 68:2180–2188
Ojeda NB, Johnson WR, Dwyer TM, Alexander BT (2007) Early renal denervation prevents development of hypertension in growth-restricted offspring. Clin Exp Pharmacol Physiol 34:1212–1216
Stewart T, Ascani J, Craver RD, Vehaskari VM (2009) Role of postnatal dietary sodium in prenatally programmed hypertension. Pediatr Nephrol 24:1727–1733
Alwasel SH, Ashton N (2009) Prenatal programming of renal sodium handling in the rat. Clin Sci (Lond) 117:75–84
Simonetti GD, Raio L, Surbek D, Nelle M, Frey FJ, Mohaupt MG (2008) Salt sensitivity of children with low birth weight. Hypertension 52:625–630
Coresh J (2007) Prevalence of chronic kidney disease in the United States. JAMA 298:2038–2047
White SL, Perkovic V, Cass A, Chang CL, Poulter NR, Spector T, Haysom L, Craig JC, Salmi IA, Chadban SJ, Huxley RR (2009) Is low birth weight an antecedent of CKD in later life? A systematic review of observational studies. Am J Kidney Dis 54:248–261
Fagerudd J, Forsblom C, Pettersson-Fernholm K, Saraheimo M, Waden J, Ronnback M, Rosengard-Barlund M, Bjorkesten CG, Thorn L, Wessman M, Groop PH (2006) Low birth weight does not increase the risk of nephropathy in Finnish type 1 diabetic patients. Nephrol Dial Transplant 21:2159–2165
Li S, Chen SC, Shlipak M, Bakris G, McCullough PA, Sowers J, Stevens L, Jurkovitz C, McFarlane S, Norris K, Vassalotti J, Klag MJ, Brown WW, Narva A, Calhoun D, Johnson B, Obialo C, Whaley-Connell A, Becker B, Collins AJ (2008) Low birth weight is associated with chronic kidney disease only in men. Kidney Int 73:637–642
Hallan S, Euser AM, Irgens LM, Finken MJ, Holmen J, Dekker FW (2008) Effect of intrauterine growth restriction on kidney function at young adult age: the Nord Trondelag Health (HUNT 2) study. Am J Kidney Dis 51:10–20
Gielen M, Pinto-Sietsma SJ, Zeegers MP, Loos RJ, Fagard R, de Leeuw PW, Beunen G, Derom C, Vlietinck R (2005) Birth weight and creatinine clearance in young adult twins: influence of genetic, prenatal, and maternal factors. J Am Soc Nephrol 16:2471–2476
Painter RC, Roseboom TJ, van Montfrans GA, Bossuyt PM, Krediet RT, Osmond C, Barker DJ, Bleker OP (2005) Microalbuminuria in adults after prenatal exposure to the Dutch famine. J Am Soc Nephrol 16:189–194
Zidar N, Avgustin Cavic M, Kenda RB, Ferluga D (1998) Unfavorable course of minimal change nephrotic syndrome in children with intrauterine growth retardation. Kidney Int 54:1320–1323
Zidar N, Cavic MA, Kenda RB, Koselj M, Ferluga D (1998) Effect of intrauterine growth retardation on the clinical course and prognosis of IgA glomerulonephritis in children. Nephron 79:28–32
Plank C, Ostreicher I, Dittrich K, Waldherr R, Voigt M, Amann K, Rascher W, Dotsch J (2007) Low birth weight, but not postnatal weight gain, aggravates the course of nephrotic syndrome. Pediatr Nephrol 22:1881–1889
Keijzer-Veen MG, Kleinveld HA, Lequin MH, Dekker FW, Nauta J, de Rijke YB, van der Heijden BJ (2007) Renal function and size at young adult age after intrauterine growth restriction and very premature birth. Am J Kidney Dis 50:542–551
Rodriguez-Soriano J, Aguirre M, Oliveros R, Vallo A (2005) Long-term renal follow-up of extremely low birth weight infants. Pediatr Nephrol 20:579–584
Rakow A, Johansson S, Legnevall L, Sevastik R, Celsi G, Norman M, Vanpee M (2008) Renal volume and function in school-age children born preterm or small for gestational age. Pediatr Nephrol 23:1309–1315
Kistner A, Celsi G, Vanpee M, Jacobson SH (2000) Increased blood pressure but normal renal function in adult women born preterm. Pediatr Nephrol 15:215–220
Iacobelli S, Loprieno S, Bonsante F, Latorre G, Esposito L, Gouyon JB (2007) Renal function in early children in very low birthweight infants. Am J Perinatol 24:587–592
Hodgin JB, Rasoulpour M, Markowitz GS, D'Agati VD (2009) Very low birth weight is a risk factor for secondary focal segmental glomerulosclerosis. Clin J Am Soc Nephrol 4:71–76
Abitbol CL, Chandar J, Rodriguez MM, Berho M, Seeherunvong W, Freundlich M, Zilleruelo G (2009) Obesity and preterm birth: additive risks in the progression of kidney disease in children. Pediatr Nephrol 24:1363–1370
Nwagwu MO, Cook A, Langley-Evans SC (2000) Evidence of progressive deterioration of renal function in rats exposed to a maternal low-protein diet in utero. Br J Nutr 83:79–85
Gluckman PD, Hanson MA, Buklijas T, Low FM, Beedle AS (2009) Epigenetic mechanisms that underpin metabolic and cardiovascular diseases. Nat Rev Endocrinol 5:401–408
Pham TD, MacLennan NK, Chiu CT, Laksana GS, Hsu JL, Lane RH (2003) Uteroplacental insufficiency increases apoptosis and alters p53 gene methylation in the full-term IUGR rat kidney. Am J Physiol Regul Integr Comp Physiol 285:R962–R970
Harrison M, Langley-Evans SC (2009) Intergenerational programming of impaired nephrogenesis and hypertension in rats following maternal protein restriction during pregnancy. Br J Nutr 101:1020–1030
Ehrenkranz RA, Dusick AM, Vohr BR, Wright LL, Wrage LA, Poole WK (2006) Growth in the neonatal intensive care unit influences neurodevelopmental and growth outcomes of extremely low birth weight infants. Pediatrics 117:1253–1261
Poindexter BB, Langer JC, Dusick AM, Ehrenkranz RA (2006) Early provision of parenteral amino acids in extremely low birth weight infants: relation to growth and neurodevelopmental outcome. J Pediatr 148:300–305
Stephens BE, Walden RV, Gargus RA, Tucker R, McKinley L, Mance M, Nye J, Vohr BR (2009) First-week protein and energy intakes are associated with 18-month developmental outcomes in extremely low birth weight infants. Pediatrics 123:1337–1343
McCance R (1962) Food, growth and time. Lancet 2:621–626
Plagemann A, Harder T, Rake A, Voits M, Fink H, Rohde W, Dorner G (1999) Perinatal elevation of hypothalamic insulin, acquired malformation of hypothalamic galaninergic neurons, and syndrome x-like alterations in adulthood of neonatally overfed rats. Brain Res 836:146–155
Boullu-Ciocca S, Dutour A, Guillaume V, Achard V, Oliver C, Grino M (2005) Postnatal diet-induced obesity in rats upregulates systemic and adipose tissue glucocorticoid metabolism during development and in adulthood: its relationship with the metabolic syndrome. Diabetes 54:197–203
Velkoska E, Cole TJ, Dean RG, Burrell LM, Morris MJ (2008) Early undernutrition leads to long-lasting reductions in body weight and adiposity whereas increased intake increases cardiac fibrosis in male rats. J Nutr 138:1622–1627
Jennings BJ, Ozanne SE, Dorling MW, Hales CN (1999) Early growth determines longevity in male rats and may be related to telomere shortening in the kidney. FEBS Lett 448:4–8
Vickers MH, Breier BH, Cutfield WS, Hofman PL, Gluckman PD (2000) Fetal origins of hyperphagia, obesity, and hypertension and postnatal amplification by hypercaloric nutrition. Am J Physiol Endocrinol Metab 279:E83–E87
Desai M, Babu J, Ross M (2007) Programming metabolic syndrome: prenatal undernutrition and postweaning overnutrition. Am J Physiol Regul Integr Comp Physiol 293:R2306–R2314
Mühle A, Mühle C, Amann K, Dötsch J, Nüsken KD, Boltze J, Schneider H (2010) No juvenile arterial hypertension in sheep multiples despite reduced nephron numbers. Pediatr Nephrol 25:1653–1661
De Matteo R, Stacy V, Probyn M, Brew N, Harding R (2008) The perinatal development of arterial pressure in sheep: effects of low birth weight due to twinning. Reprod Sci 15:66–74
Petry C, Jennings B, James L, Hales C, Ozanne S (2006) Suckling a protein restricted rat dam leads to diminished albuminuria in her male offspring in adult life: a longitudinal study. BMC Nephrol 29:7–14
Tarry-Adkins JL, Joles JA, Chen JH, Martin-Gronet MS, van der Giezen DM, Goldschmeding RR, Hales CN, Ozanne SE (2007) Protein restriction in lactation confers nephroprotective effects in the male rat and is associated with increased antioxidant expression. Am J Physiol Regul Integr Comp Physiol 293:R1259–R1266
Silbiger S, Neugarten J (2008) Gender and human chronic renal disease. Gend Med 5:S3–S10
Wang J, Bingaman S, Huxley VH (2010) Intrinsic sex-specific differences in microvascular endothelial cell phosphodiesterase. Am J Physiol Heart Circ Physiol 298:H1146–H1154
Woods LL, Morgan TK, Resko JA (2010) Castration fails to prevent prenatally programmed hypertension in male rats. Am J Physiol Regul Integr Comp Physiol 298:R1111–R1116
Baserga M, Kaur R, Hale MA, Bares A, Yu X, Callaway CW, McKnight RA, Lane RH (2010) Fetal growth restriction alters transcription factor binding and epigenetic mechanisms of renal 11beta-hydroxysteroid dehydrogenase type 2 in a sex-specific manner. Am J Physiol Regul Integr Comp Physiol 299:R334–R342
Durcova-Hills G, Burgoyne P, McLaren A (2004) Analysis of sex differences in EGC imprinting. Dev Biol 268:105–110
Gabory A, Attig L, Junien C (2009) Sexual dimorphism in environmental epigenetic programming. Mol Cell Endocrinol 304:8–18
Lucas A, Fewtrell M, Morley R, Singhal A, Abbott R, Isaac E, Stephenson T, MacFadyen UM, Clements H (2001) Randomized trial of nutrient-enriched formula versus standard formula for postdischarge preterm infants. Pediatrics 108:703–711
Griffin KA, Kramer H, Bidani AK (2008) Adverse renal consequences of obesity. Am J Physiol Renal Physiol 294:F685–F696
Sanders MW, Fazzi GE, Janssen GM, Blanco CE, De Mey JG (2005) High sodium intake increases blood pressure and alters renal function in intrauterine growth-retarded rats. Hypertension 46:71–75
Correa-Rotter R, Hostetter TH, Rosenberg ME (1992) Effect of dietary protein on renin and angiotensinogen gene expression after renal ablation. Am J Physiol 262:F631–F638
Pavenstadt H, Kriz W, Kretzler M (2003) Cell biology of the glomerular podocyte. Physiol Rev 83:253–307
Chevalier RL, Kaiser DL (1983) Autoregulation of renal blood flow in the rat: effects of growth and uninephrectomy. Am J Physiol 244:F483–F487
Shinozaki K, Kashiwagi A, Massada M, Okamura T (2004) Molecular mechanism of impaired endothelial function associated with insulin resistance. Curr Drug Targets Cardiovasc Haematol Disord 4:1–11
Montarro M, Allen A, Oldfield B (2005) Structural and functional evidence supporting a role for leptin in central neural pathway influencing blood pressure in rats. Exp Physiol 90:683–688
Qian Y, Feldman E, Pennathur S, Kretzler M, Brosius FC 3rd (2008) From fibrosis to sclerosis: mechanisms of glomerulosclerosis in diabetic nephropathy. Diabetes 57:1439–1445
Adair LS, Cole TJ (2003) Rapid child growth raises blood pressure in adolescent boys who were thin at birth. Hypertension 41:451–456
Eriksson JG, Forsen T, Tuomilehto J, Winter PD, Osmond C, Barker DJ (1999) Catch-up growth in childhood and death from coronary heart disease: longitudinal study. BMJ 318:427–431
Barker DJ, Osmond C, Forsen TJ, Kajantie E, Eriksson JG (2005) Trajectories of growth among children who have coronary events as adults. N Engl J Med 353:1802–1809
Stettler N, Stallings VA, Troxel AB, Zhao J, Schinnar R, Nelson SE, Ziegler EE, Strom BL (2005) Weight gain in the first week of life and overweight in adulthood: a cohort study of European American subjects fed infant formula. Circulation 111:1897–1903
Singhal A, Fewtrell M, Cole TJ, Lucas A (2003) Low nutrient intake and early growth for later insulin resistance in adolescents born preterm. Lancet 361:1089–1097
Singhal A, Cole TJ, Fewtrell M, Deanfield J, Lucas A (2004) Is slower early growth beneficial for long-term cardiovascular health? Circulation 109:1108–1113
Herin P, Zetterstrom R (1987) Studies in renal response to various protein intakes in preterm infants. Acta Paediatr Scand 76:447–452
Schmidt IM, Damgaard IN, Boisen KA, Mau C, Chellakooty M, Olgaard K, Main KM (2004) Increased kidney growth in formula-fed versus breast-fed healthy infants. Pediatr Nephrol 19:1137–1144
Bacchetta J, Harambat J, Dubourg L, Guy B, Liutkus A, Canterino I, Kassai B, Putet G, Cochat P (2009) Both extrauterine and intrauterine growth restriction impair renal function in children born very preterm. Kidney Int 76:445–452
Lopez-Bermejo A, Sitjar C, Cabacas A, Vazquez-Ruiz M, Garcia-Gonzalez MM, Mora C, Soriano P, Calvo M, Ibanez L (2008) Prenatal programming of renal function: the estimated glomerular filtration rate is influenced by size at birth in apparently healthy children. Pediatr Res 64:97–99
Langley-Evans SC, Jackson AA (1994) Increased systolic blood pressure in adult rats induced by fetal exposure to maternal low protein diets. Clin Sci 86:217–222
Zimanyi MA, Bertram JF, Black MJ (2004) Does a nephron deficit in rats predispose to salt-sensitive hypertension ? Kidney Blood Pressure Res 27:239–247
Langley-Evans SC, Welham SJ, Jackson AA (1999) Fetal exposure to a maternal low protein diet impairs nephrogenesis and promotes hypertension in the rat. Life Sci 64:965–974
Jackson AA, Dunn RL, Marchand MC, Langley-Evans SC (2002) Increased systolic blood pressure in rats induced by a maternal low-protein diet is reversed by dietary supplementation with glycine. Clin Sci (Lond) 103:633–639
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Simeoni, U., Ligi, I., Buffat, C. et al. Adverse consequences of accelerated neonatal growth: cardiovascular and renal issues. Pediatr Nephrol 26, 493–508 (2011). https://doi.org/10.1007/s00467-010-1648-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-010-1648-1