Abstract
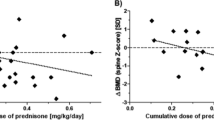
Long-term glucocorticoid treatment contributes to the growth retardation in children after renal transplantation. We investigated whether determination of prednisone (PN) and prednisolone (PL) in plasma and PN, PL, and 6-β-hydroxyprednisolone (βOH-PL) in urine could help to predict growth. PN and PL pharmacokinetics were studied in 36 children, from 5 to 15 years of age, receiving daily (D) or alternate-day (AD) oral PN treatment. Statural growth velocity was evaluated over a 1-year period. We compared three groups of children according to the growth kinetics during the study year (catch-up, stable, or decline) for clinical and pharmacokinetic parameters. A multiple linear regression analysis was performed in order to determine pharmacokinetic parameters able to explain height 1 year after inclusion. Height at the beginning of the study, creatinine clearance, and type of D or AD treatment explained 94.2% of height variance 1 year after inclusion. Only PL clearance was associated with growth evolution, but introduction of PL clearance in the multivariate model did not improve the variance of height accounted for by the previous model. We, therefore, do not recommend using glucocorticoid pharmacokinetics to predict growth retardation in children with renal transplantation.


Similar content being viewed by others
References
Pickup ME (1979) Clinical pharmacokinetics of prednisone and prednisolone. Clin Pharmacokinet 4:111–128
Gambertoglio JG, Amend WJ Jr, Benet LZ (1980) Pharmacokinetics and bioavailability of prednisone and prednisolone in healthy volunteers and patients: a review. J Pharmacokinet Biopharm 8:1–52
Escher G, Frey FJ, Frey BM (1994) 11 beta-Hydroxysteroid dehydrogenase accounts for low prednisolone/prednisone ratios in the kidney. Endocrinology 135:101–106
Renner E, Horber FF, Jost G, Frey BM, Frey FJ (1986) Effect of liver function on the metabolism of prednisone and prednisolone in humans. Gastroenterology 90:819–828
Loo JC, McGilveray IJ, Jordan N, Moffat J, Brien R (1978) Dose-dependent pharmacokinetics of prednisone and prednisolone in man. J Pharm Pharmacol 30:736
Frey BM, Frey FJ (1990) Clinical pharmacokinetics of prednisone and prednisolone. Clin Pharmacokinet 19:126–146
Frey FJ, Schaad HJ, Renner EL, Horber FF, Frey BM, Preisig R (1989) Impaired liver function in stable renal allograft recipients. Hepatology 9:606–613
Frey FJ, Frey BM, Schnetzer A, Horber FF (1987) Evidence that cyclosporin A does not affect the metabolism of prednisolone after renal transplantation. Transplantation 43:494–498
Guest G, Broyer M (1991) Growth after renal transplantation: correlation with immunosuppressive therapy. Pediatr Nephrol 5:143–146
Sarna S, Hoppu K, Neuvonen PJ, Laine J, Holmberg C (1997) Methylprednisolone exposure, rather than dose, predicts adrenal suppression and growth inhibition in children with liver and renal transplants. J Clin Endocrinol Metab 82:75–77
Sarna S, Sipilä I, Vihervuori E, Koistinen R, Holmberg C (1995) Growth delay after liver transplantation in childhood: studies of underlying mechanisms. Pediatr Res 38:366–372
Travis LB, Chesney R, McEnery P, Moel D, Pennisi A, Potter D, Talwalkar YB, Wolff E (1978) Growth and glucocorticoids in children with kidney disease. Kidney Int 14:365–368
Meacham LR, Culler FL, Abdul-Latif H, Sullivan KM, Bowers CY (1999) Preservation of growth hormone secretion in response to growth hormone-releasing peptide-2 during prednisone therapy. Metabolism 48:585–589
Hokken-Koelega AC, Saenger P, Cappa M, Greggio N (2001) Unresolved problems concerning optimal therapy of puberty in children with chronic renal diseases. J Pediatr Endocrinol Metab 14 [Suppl 2]:945–952
Qvist E, Marttinen E, Ronnholm K, Antikainen M, Jalanko H, Sipila I, Holmberg C (2002) Growth after renal transplantation in infancy or early childhood. Pediatr Nephrol 17:438–443
Sempe M, Pedron G, Roy-Pernot MP (1979) Auxologie, méthode et séquence. Théraplix Ed., Paris, 35
Schwartz GJ, Haycock GB, Edelmann CM Jr, Spitzer A (1976) A simple estimate of glomerular filtration rate in children derived from body length and plasma creatinine. Pediatrics 58:259–263
Uribe M, Schalm SW, Summerskill WH, Go VL (1978) Oral prednisone for chronic active liver disease: dose responses and bioavailability studies. Gut 19:1131–1135
Pickup ME, Lowe JR, Leatham PA, Rhind VM, Wright V, Downie WW (1977) Dose dependent pharmacokinetics of prednisolone. Eur J Clin Pharmacol 12:213–219
Kaiser BA, Polinsky MS, Palmer JA, Dunn S, Mochon M, Flynn JT, Baluarte HJ (1994) Growth after conversion to alternate-day corticosteroids in children with renal transplants: a single-center study. Pediatr Nephrol 8:320–325
Hokken-Koelega AC, Zaal MAE van, Bergen W van, Ridder MAJ de, Stijnen T, Wolff ED, Jong RCJW de, Donckerwolcke RA, Muinck Keizer-Schrama SMPF de, Drop SLS (1994) Final height and its predictive factors after renal transplantation in childhood. Pediatr Res 36:323–328
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chavatte, C., Guest, G., Proust, V. et al. Glucocorticoid pharmacokinetics and growth retardation in children with renal transplants. Pediatr Nephrol 19, 898–904 (2004). https://doi.org/10.1007/s00467-004-1497-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-004-1497-x