Abstract
Introduction
Harvested biological tissue is a common medium for surgical device assessment in a laboratory setting; this study aims to differentiate between surgical device performance in the clinical and laboratory environments prior to and following tissue storage. Vascular tissue fusion devices are sensitive to tissue-device temperature gradients, tissue pre-stretch in vivo and tissue water content, each of which can vary during tissue storage. In this study, we compare the results of tissue fusion prior to and following storage using a standardized bursting pressure protocol.
Methods
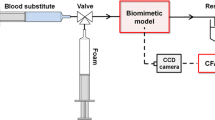
Epigastric veins from seven porcine models were subject to identical bursting pressure protocols after fusion. One half of each vein was fused in vivo, harvested and immediately analyzed for burst pressure; the remainder was stored (0.9% Phosphate Buffered Saline, 24h, 4 °C) and then analyzed ex vivo. Histological slides were prepared for qualitative analysis of in versus ex vivo fusions.
Results
Bursting pressures of vessels fused ex vivo (514.7 ± 187.0 mmHg) were significantly greater than those of vessels fused in vivo (310 ± 127.7 mmHg, p = 2.06 E-10). Histological imaging of venous axial cross-sections indicated the lamination of adventitia and media layers ex vivo, whereas in vivo samples consisted only of adventitia.
Conclusion
These findings suggest that the fusion of porcine venous tissue ex vivo may overestimate the clinical performance of fusion devices. Prior work has indicated that increased tissue hydration and the lamination of tissue layers both positively affect arterial fusion bursting pressures. The bursting pressure increase observed herein may therefore be due to storage-induced alterations in tissue composition and mechanics of the fusion interface. While harvested tissue provides an accessible medium for comparative study, the fusion of vascular tissue in vivo may avoid storage-induced biomechanical alterations and is likely a better indicator of fusion device performance in a clinical setting.





Similar content being viewed by others
References
Carbonell AM, Joels CS, Kercher KW, Matthews BD, Sing RF, Heniford BT (2003) A comparison of laparoscopic bipolar vessel sealing devices in the hemostasis of small-, medium-, and large-sized arteries. J Laparoendosc Adv Surg Tech A 13(6):377–380
Person B, Vivas DA, Ruiz D, Talcott M, Coad JE, Wexner SD (2008) Comparison of four energy-based vascular sealing and cutting instruments: a porcine model. Surg Endosc 22(2):534–538
Lamberton GR, Hsi RS, Jin DH, Lindler TU, Jellison FC, Baldwin DD (2008) Prospective comparison of four laparoscopic vessel ligation devices. J Endourol Endourol Soc 22(10):2307–2312
Harold KL, Pollinger H, Matthews BD, Kercher KW, Sing RF, Heniford BT (2003) Comparison of ultrasonic energy, bipolar thermal energy, and vascular clips for the hemostasis of small-, medium-, and large-sized arteries. Surg Endosc 17(8):1228–1230
Wallwiener CW, Rajab TK, Zubke W, Isaacson KB, Enderle M, Schäller D, Wallwiener M (2008) Thermal conduction, compression, and electrical current—an evaluation of major parameters of electrosurgical vessel sealing in a porcine in vitro model. J Minim Invasive Gynecol 15(5):605–610
Brossollet LJ, Vito RP (1997) The Effects of cryopreservation on the biaxial mechanical properties of canine saphenous veins. J Biomech Eng 119:1–5
Stemper BD, Yoganandan N, Stineman MR, Gennarelli TA, Baisden JL, Pintar FA (2007) Mechanics of fresh, refrigerated, and frozen arterial tissue. J Surg Res 139(2):236–242
Neil DAH, Maguire SH, Walsh M, Lynch SV, Hardie IR, Effeney DJ (1997) Progression of changes in arteries following cold storage preservation in UW and collins solution in a syngeneic aortic transplant model. Transpl Proc 29:2561–2562
J. Cezo, E. Kramer, K. Taylor, V. Ferguson, and M. Rentschler (2013) Tissue fusion bursting pressure and the role of tissue water content. In: Proceedings of the BiOS SPIE Photonics West, San Fransisco, CA
Wille T, de Groot H, Rauen U (2008) Improvement of the cold storage of blood vessels with a vascular preservation solution. Study in porcine aortic segments. J Vasc Surg 47(2):422–431
Neil DA, Lynch SV, Hardie IR, Effeney DJ (2002) Cold storage preservation and warm ischaemic injury to isolated arterial segments: endothelial cell injury. Am J Transplant 2(5):400–409
Cezo JD, Kramer E, Taylor KD, Ferguson V, Rentschler ME (2013) Temperature measurement methods during direct heat arterial tissue fusion. IEEE Trans Biomed Eng 60(9):2552–2558
Cezo JD, Passernig AC, Ferguson VL, Taylor KD, Rentschler ME (2014) Evaluating temperature and duration in arterial tissue fusion to maximize bond strength. J Mech Behav Biomed Mater 30:41–49
Cavallari N, Abebe W, Mingoli A, Sapienza P, Hunter WJ III, Agrawal DK, Cavallaro A, Edwards JD (1997) Short-term preservation of autogenous vein grafts: effectiveness of University of Wisconsin solution. Surgery 121(1):64–71
Southard JH (2005) The right solution for organ preservation. Bus Briefing: US Kidney Urol Dis 2005:1–4
Zatschler B, Dieterich P, Müller B, Kasper M, Rauen U, Deussen A (2009) Improved vessel preservation after 4 days of cold storage: experimental study in rat arteries. J Vasc Surg 50(2):397–406
Garbe S, Zatschler B, Müller B, Dieterich P, Ebner A, Rauen U, Matschke K, Deussen A (2011) Preservation of human artery function following prolonged cold storage with a new solution. J Vasc Surg 53(4):1063–1070
Cardamone L, Valentín A, Eberth JF, Humphrey JD (2009) Origin of axial prestretch and residual stress in arteries. Biomech Model Mechanobiol 8(6):431–446
Richter S, Kollmar O, Neunhoeffer E, Schilling MK, Menger MD, Pistorius G (2006) Differential response of arteries and veins to bipolar vessel sealing: evaluation of a novel reusable device. J Laparoendosc Adv Surg Tech A 16(2):149–155
Hruby GW, Marruffo FC, Durak E, Collins SM, Pierorazio P, Humphrey PA, Mansukhani MM, Landman J (2007) Evaluation of surgical energy devices for vessel sealing and peripheral energy spread in a porcine model. J Urol 178(6):2689–2693
Acknowledgments
The authors would like to thank Nicholas Anderson, Kelli Barnes, Dave Stafford, Roberto del Cid, ConMed Electrosurgery, Premier Labs, PreClinical Research Services, Inc., and the University of Colorado Denver Anschutz Medical Campus Large Animal Operating Room staff. This publication is funded by grant support from ConMed Electrosurgery Corporation, Centennial, CO, to the University of Colorado at Boulder.
Disclosures
Drs. Rentschler, Ferguson, Cezo, and Eric Kramer receive materials, supplies, and travel funding under the ConMed Corporation grant. Dr. Taylor is the Vice President of research and development at ConMed Corporation. Dr. Schoen has no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cezo, J.D., Kramer, E.A., Schoen, J.A. et al. Tissue storage ex vivo significantly increases vascular fusion bursting pressure. Surg Endosc 29, 1999–2005 (2015). https://doi.org/10.1007/s00464-014-3900-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-014-3900-4