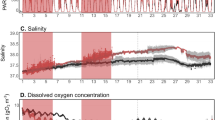
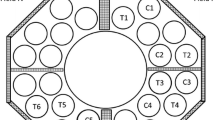
Abstract
An indoor mesocosm system was set up to study the response of phytoplankton and zooplankton spring succession to winter and spring warming of sea surface temperatures. The experimental temperature regimes consisted of the decadal average of the Kiel Bight, Baltic Sea, and three elevated regimes with 2°C, 4°C, and 6°C temperature difference from that at baseline. While the peak of the phytoplankton spring bloom was accelerated only weakly by increasing temperatures (1.4 days per degree Celsius), the subsequent biomass minimum of phytoplankton was accelerated more strongly (4.25 days per degree Celsius). Phytoplankton size structure showed a pronounced response to warming, with large phytoplankton being more dominant in the cooler mesocosms. The first seasonal ciliate peak was accelerated by 2.1 days per degree Celsius and the second one by 2.0 days per degree Celsius. The over-wintering copepod populations declined faster in the warmer mesocosm, and the appearance of nauplii was strongly accelerated by temperature (9.2 days per degree Celsius). The strong difference between the acceleration of the phytoplankton peak and the acceleration of the nauplii could be one of the “Achilles heels” of pelagic systems subject to climate change, because nauplii are the most starvation-sensitive life cycle stage of copepods and the most important food item of first-feeding fish larvae.







Similar content being viewed by others
References
Aberle N, Lengfellner K, Sommer U (2006) Spring bloom succession, grazing impact and herbivore selectivity of ciliate communities in response to winter warming. Oecologia (this issue)
Bautista B, Harris RP, Tranter PRG, Harbour D (1992) In situ copepod feeding and grazing rates during a spring bloom dominated by Phaeocystis sp. in the English Channel. J Plankton Res 14:691–703
Behrends G (1996) Long-term investigation of seasonal mesozooplankton dynamics in Kiel Bight, Germany. In: Proceedings of the 13th symposium on Baltic and Marine Biology, pp 93–98
Brock TD (1981) Calculating solar radiation for ecological studies. Ecol Model 14:1–19
Cushing DH (1975) Marine ecology and fisheries. Cambridge University Press, Cambridge
Edwards M, Beaugrand G, Reid PC, Rowden AA, Joes MB (2002) Ocean climate anomalies and the ecology of the North Sea. Mar Ecol Progr Ser 239:1–10
Edwards M, Richardson AJ (2004) Impact of climate change on marine pelagic phenology and trophic mismatch. Nature 430:881–884
Feuchtmayr H (2004) Mesozooplankton impacts on lower trophic levels from freshwater, marine and brackish systems in spring—a comparative study. PhD thesis, University of Kiel
Fromentin JM, Planque B (1996) Calanus and the environment in the eastern North Atlantic. II: Influence of the North Atlantic Oscillation on C. finnmarchicus and C. helgolandicus. Mar Ecol Progr Ser 134:111–118
Goldman JC, McCarthy JJ, Peavey DG (1979) Growth rate influence on the chemical composition of phytoplankton in oceanic waters. Nature 279:210–215
Granéli E, Turner JT (2002) Top-down regulation in ctenophore–copepod–ciliate–diatom–phytoflagellate communities in coastal waters: a mesocosm study. Mar Ecol Progr Ser 239:57–68
Greve W, Reiners F (1995) Biocoenotic process patterns in the German Bight. In: Eleftheriou A, et al (eds) Biology and ecology of shallow coastal waters. Olsen & Olsen, Fredensborg, pp 67–71
Hansen HP, Koroleff F (1999) Determination of nutrients. In: Grasshoff K, Kremling K, Ehrhardt M (eds) Methods of seawater analysis, 3rd edition. Wiley VCH, Weinheim pp 159–228
Hillebrand H, Dürselen CD, Kischtel D, Pollingher U (1999) Biovolume calculations for pelagic and benthic microalgae. J Phycol 35:403–424
Holmes RM, Aminot A, Kérouel R, Hooker BA, Peterson BJ (1999) A simple and precise method for measuring ammonium in marine and freshwater ecosystems. Can J Fish Aquat Sci 56:1801–1808
International Panel on Climate Change (IPCC) (2001) Climate change 2001: impacts, adaptations and vulnerability. UNEP and WHO, 2001
Irigoien X, Titelman J, Harris RP, Harbour D, Castellani C (2003) Feeding of Calanus finnmarchicus nauplii in the Irminger Sea. Mar Ecol Progr Ser 262:193–200
Katechakis A, Stibor H, Sommer U, Hansen T (2002) Changes in the phytoplankton community and microbial food web of Blanes Bay (Catalan Sea, NW Mediterranean) under prolonged grazing pressure by doliolids (Tunicata), cladocerans or copepods (Crustacea). Mar Ecol Progr Ser 234:55–69
Kleppel GS (1993) On the diet of calanoid copepods. Mar Ecol Progr Ser 99:183–195
Lehmann A, Krauss WW, Hinrichsen HH (2002) Effects of remote and local atmospheric forcing on circulation and upwelling in the Baltic Sea. Tellus 54A:299–316
Lopez MDG (1996) Effect of starvation on development and survivorship of nauplian Calanus pacificus (Brodsky). J Exp Mar Biol Ecol 203:133–146
Madhudatrap M, Nehring S, Lenz J (1996) Resting eggs of zooplankton (Copepoda and Cladocera) from the Kiel Bay and adjacent waters (southwestern Baltic). Mar Biol 125:77–87
Matthäus W., H. Schinke (1994) Mean atmospheric circulation patterns associated with Major Baltic Inflows. Dt Hydrogr Z 46:321–338
Menden-Deuer S, Lessard EJ (2000) Carbon to volume relationships in dinoflagellates, diatoms and other protist plankton. Limnol Oceanogr 45:569–579
Montagnes DJS, Lynn DH, Roff JC, Taylor WD (1988) The annual cycle of heterotrophic planktonic ciliates in the waters surrounding the Isles of Shoals, Gulf of Maine: an assessment of their trophic role. Mar Biol 99:21–30
Putt M, Stoecker DK (1989) An experimentally determined carbon:volume ratio for marine “oligotrichous” ciliates from estuarine and coastal waters. Limnol Oceanogr 34:1097–1103
Riley GA (1957) Phytoplankton of the North Central Sargasso Sea. Limnol Oceanogr 2:252–270
Scheffer M, Straile D, van Nes EH, Hosper H (2001) Climate warming causes regime shifts in lake food webs. Limnol Oceanogr 46:1780–1783
Sell AF, van Keuren D, Madin LP (2001) Predation by omnivorous copepods on early developmental stages of Calanus finnmarchicus and Pseudocalanus spp. Limnol Oceanogr 46:953–959
Sharp JH (1974) Improved analysis for particulate organic carbon and nitrogen from seawater. Limnol Oceanogr 19:984–989
Smayda TJ (1971) Normal and accelerated sinking of diatoms in the sea. Mar Geol 11:105–122
Smetacek V, v Bodungen B, Knoppers B, Peinert R, Pollehne F, Stegmann P, Zeitszschel B (1984) Seasonal stages characterizing the annual cycle of an inshore pelagic ecosystem. Rapp P V Reun Cons Int Explor Mer 183:126–135
Sommer U (1991) The application of the Droop model of nutrient limitation to natural phytoplankton. Verh Int Verein Limnol 24:791–794
Sommer U (1996) Plankton ecology: the last two decades of progress. Naturwiss 83:393–301
Sommer U, Stibor H (2002) Copepoda–Cladocera–Tunicata: the role of three major mesozooplankton groups in pelagic food webs. Ecol Res 17:161–174
Sommer U, Gliwicz ZM, Lampert W, Duncan A (1986) The PEG model of seasonal succession of planktonic events in fresh waters. Arch Hydrobiol 106:433–471
Sommer F, Hansen T, Feuchtmayr H, Santer B, Tokle N, Sommer U (2003a) Do calanoid copepods suppress appendicularians in the coastal ocean? J Plankton Res 25:869–871
Sommer U, Sommer F, Santer B, Zöllner E, Jürgens K, Jamieson C, Boersma M, Gocke K (2003b) Daphnia versus copepod impact on summer phytoplankton: functional compensation at both trophic levels. Oecologia 135:639–647
Sommer F, Saage A, Santer B, Hansen T, Sommer U (2005a) Linking foraging strategies of marine calanoid copepods to patterns of nitrogen stable isotope signatures in a mesocosm study. Mar Ecol Prog Ser 286:99–106
Sommer U, Hansen T, Blum O, Holzner N, Vadstein O, Stibor H (2005b) Copepod and microzooplankton grazing in mesocosms fertilised with different Si:N ratios: no overlap between food spectra and Si:N influence on zooplankton trophic level. Oecologia 142:274–283
Sterner RW, Elser JJ (2002) Ecological stoichiometry. Princeton University Press, Princeton
Straile D (2000) Meteorological forcing of plankton dynamics in a large and deep continental European lake. Oecologia 122:44–50
Straile D, Adrian R (2000) The North Atlantic Oscillation and plankton dynamics in two European lakes—two variations on a general theme. Global Change Biol 6:663–670
Sverdrup HU (1953) On conditions for the vernal blooming of phytoplankton. J Cons Explor Mer 18:287–295
Tilzer MM, Elbrächter M, Gieskes WW, Beese B (1986) Light–temperature interactions in the control of photosynthesis in Antarctic phytoplankton. Polar Biol 5:105–111
Urabe J, Clasen J, Sterner RW (1997) Phosphorus limitation of Daphnia growth Is it real? Limnol Oceanogr 42:1436–1443
Utermöhl H (1958) Zur Vervollkommnung der quantitativen Phytoplankton-Methodik. Mitt Int Ver Theor Angew Limnol 9:263–272
Walther GR, Post E, Convey P, Menzel A, Parmesan C, Beebee TC, Fromentin JM, Hoegh-Guldberg O, Bairlein F (2002) Ecological responses to recent climate change. Nature 416:389–395
White HH (1979) Effects of dinoflagellate bioluminescence on the ingestion rates of herbivorous zooplankton. J Exp Mar Biol Ecol 36:217–224
Yachi S, Loreau M (1999) Biodiversity and ecosystem productivity in a fluctuating environment: the insurance hypothesis. Proc Nat Acad Sci U S A 96:1463–1468
Acknowledgements
This work was supported by the DFG (Deutsche Forschungsgemeinschaft) within the priority program 1162 “AQUASHIFT” (The impact of climate variability on aquatic ecosystems). Constructive comments by Herwig Stibor and one anonymous reviewer are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Roland Brandl.
Priority programme of the German Research Foundation—contribution 3.
Rights and permissions
About this article
Cite this article
Sommer, U., Aberle, N., Engel, A. et al. An indoor mesocosm system to study the effect of climate change on the late winter and spring succession of Baltic Sea phyto- and zooplankton. Oecologia 150, 655–667 (2007). https://doi.org/10.1007/s00442-006-0539-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-006-0539-4