Abstract
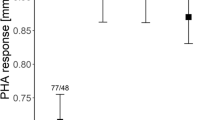
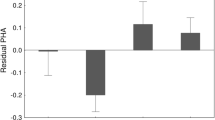
Immunocompetence may be a good measure of offspring quality, however, factors affecting variation in immune responses are not clear. Research suggests that immune function can vary due to differences in genetics, development conditions and individual quality. Here, I examined factors affecting variation in immune response among nestling European starlings through a split-nest cross-fostering brood manipulation that included two important covariates: spleen size and nest temperatures. Immunocompetence was assessed via a cell-mediated immune response to phytohaemagglutinin (PHA). This paper provides the first direct evidence that individuals with large spleens also mount strong immune responses. Exposure to PHA did not cause splenomegaly, as there was no difference in spleen size between control birds and those injected with PHA. Offspring immune function was affected by common origin and by rearing environment, though rearing environment appeared to exert its influence only through nest temperatures. A comparison of the immune performance of siblings reared in their home nest versus those reared in other nests revealed a strong effect of maternal quality. As the difference in natal clutch size increased, the magnitude of the difference in immune performance between home-reared nestlings versus out-reared nestlings increased. Overall, nestling immune function appears to be determined by the combination of genetic, maternal and environmental effects.


Similar content being viewed by others
References
Adriaensen F, Dhondt AA, Van Dongen S, Lens S, Matthysen E (1998) Stabilizing selection on blue tit fledgling mass in the presence of sparrowhawks. Proc R Soc Lond B Biol Sci 265:1011–1016
Apanius V (1998) Ontogeny of immune function. In: Starck JM, Ricklefs RE (eds) Evolution within the altricial-precocial spectrum. Oxford University Press, Oxford, pp 203–222
Blanco G, de la Puente J, Corroto M, Baz A, Cola J (2001) Condition-dependent immune defence in the Magpie: how important is ectoparasitism? Biol J Linnean Soc 72:279–286
Blount JD, Houston DC, Møller AP, Wright J (2003) Do individual branches of immune defence correlate? A comparative case study of scavenging and non-scavenging birds. Oikos 102:340–350
Brinkhof MWG, Heeb P, Kölliker M, Richner H (1999) Immunocompetence of nestling great tits in relation to rearing environment and parentage. Proc R Soc Lond B Biol Sci 266: 2315–2322
Brown CR, Brown MB (2002) Spleen volume varies with colony size and parasite load in a colonial bird. Proc R Soc Lond B 269:1367–1373
Brown CR, Sherman LC (1989) Variation in the appearance of swallow eggs and the detection of intraspecific brood parasitism. Condor 91:620–627
Cheng S, Lamont SJ (1988) Genetic analysis of immunocompetence measures in a white leghorn chicken line. Poul Sci 67:989–995
Christe P, Møller AP, de Lope F (1998) Immunocompetence and nestling survival in the house martin: the tasty chick hypothesis. Oikos 83:175–179
Christians JK, Evanson M, Aiken JJ (2001) Seasonal decline in clutch size in European starlings: A novel randomization test to distinguish between the timing and quality hypotheses. J Anim Ecol 70:1080–1087
Darlington RB, Smulders TV (2001) Problems with residual analysis. Anim Behav 62:599–602
Dhondt AA (1971) The regulation of numbers in Belgian populations of great tits. In: den Boer PJ, Gradwell JR (eds) Dynamics of populations. Advanced Study Institute, Wageningen, pp 532–547
Dietert RR, Golemboski KA, Austic RE (1994) Environment-immune interactions. Poult Sci 73:1062–1076
Donker RA, Nieuwland MGB, Vanderzijpp AJ (1990) Heat-stress influences on antibody-production in chicken lines selected for high and low immune responsiveness. Poult Sci 69:599–607
Flux JEC, Flux MM (1981) Artificial selection and gene flow in wild starlings, Sturnus vulgaris. Naturwissenschaften 69: 96–97
Hõrak P, Tegelmann L, Ots I, Møller AP (1999) Immune function and survival of great tit nestlings in relation to growth conditions. Oecologia 121:316–322
Henken AM, Schaarsberg AMJG, Nieuwland MGB (1983) The effect of environmental-temperature on immune-response and metabolism of the young chicken 3: Effect of environmental-temperature on the humoral immune-response following injection of sheep red-blood-cells. Poult Sci 62:51–58
Hochachka W, Smith JNM (1991) Determinants and consequences of nestling condition in song sparrows. J Anim Ecol 60:995–1008
Hoi-Leitner M, Romero-Pujante M, Hoi H, Pavlova A (2001) Food availability and immune capacity in serin nestlings. Behav Ecol Sociobiol 49:333–339
John JL (1994) The avian spleen: a neglected organ. Q Rev Biol 69:327–351
John JL (1995) Parasites and the avian spleen: Helminths. Biol J Linnean Soc 54:87–106
Kirkley JS, Gessaman JA (1990) Ontogeny of thermoregulation in red-tailed hawks and Swainson’s hawks. Wilson Bull 102:71–83
Klasing KC (1998) Nutritional modulation of resistance to infectious disease. Poult Sci 77:1119–1125
Liker A, Màrkus M, Vozár A, Zemankovics E, Rózsa L (2001) Distribution of Carnus hemapterus in a starling colony. Can J Zool 79:574–580
Linden M, Gustafsson L, Part T (1992) Selection on fledging mass in the collared flycatcher and the great tit. Ecology 73:336–343
Littell RC, Milliken GA, Stroup WW, Wolfinger RD (1996) SAS system for mixed models. SAS Institute, Cary
Lochmiller RL, Vestey MR, Boren JC (1993) Relationship between protein nutritional status and immunocompetence in northern bobwhite chicks. Auk 110:503–510
Miller L, Qureshi MA (1992) Induction of heat-shock proteins and phagocytic function of chicken macrophages following in vitro heat exposure. Vet Immunol Immunopathol 30:179–192
Møller AP, Erritzøe J (2000) Predation against birds with low immmunocompetence. Oecologia 122:500–504
Møller AP, Sorci G, Erritzøe J (1998a) Host immune function and sexual selection in birds. J Evol Biol 11:703–719
Møller AP, Sorci G, Erritzøe J, Mavarez J (1998b) Condition, disease, and immune defence. Oikos 83:301–306
Perrins CM (1964) Survival of young swifts in relation to brood size. Nature 201:1147
Saino N, Calza S, Møller AP (1997) Immunocompetence of nestling barn swallows in relation to brood size and parental effort. J Anim Ecol 66: 827–836
Smith HG (2004) Selection for synchronous breeding in the European starling. Oikos 105:301–311
Smith KG, Hunt JL (2004) On the use of the avian spleen as a measure of avian immune strength. Oecologia 138:28–31
Smits JE, Williams TD (1999) Validation of ecotoxicology techniques in passerine chicks exposed to oil sands tailings water. Ecotoxicol Environ Saf 44:105–112
Taylor RLJ, Cotter PF, Wing TL, Briles WE (1987) Major histocompatibility B complex and sex effects on the phytohaemagglutinin wattle response. Anim Genet 18:343–350
Tella JL, Bortolotti GR, Dawson RD, Forero MG (2000) The T-cell-mediated immune response of fledgling American kestrels are positively correlated with parental clutch size. Proc R Soc Lond B 267:891–895
Thompson CF, Flux JEC (1988) Body mass and lipid content at nest-leaving of European starlings in New Zealand. Ornis Scandinavica 19:1–6
Thompson CF, Flux JEC, Tetzlaff VT (1993) The heaviest nestlings are not necessarily the fattest nestlings. J Field Ornithol 64:426–432
Visser GH (1998) Development of temperature regulation. In: Starck JM, Ricklefs RE (eds) Avian growth and development: Evolution within the altricial-precocial spectrum. Oxford Ornithology Series, Oxford University Press, Oxford, pp 117–156
Warner CM, Meeker DL, Rothschild MF (1987) Genetic control of immune responsiveness: a review of its use as a tool for selection for disease resistance. J Anim Sci 64:394–406
Acknowledgements
I am grateful to John and Meg Flux for letting me work at the Belmont field site and for their generosity of time and advice, and Nathaniel Taylor for assistance under difficult field conditions. Elizabeth Atkinson and Fabio provided additional support. This manuscript was improved through discussions with Andre Dhondt, David Winkler, Wesley Hochachka, Karel Schat, Matt Wasson, Dana Hawley, Becca Safran and Mark Hauber. Victor Apanius, Charles Brown and Carol Vleck made comments that substantively improved this manuscript. The U.S. Environmental Protection Agency provided financial support. This work was carried out under an Animal Care Protocol approved by the Cornell Center for Research Animal Resources.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Carol Vleck
Rights and permissions
About this article
Cite this article
Ardia, D.R. Cross-fostering reveals an effect of spleen size and nest temperatures on immune responses in nestling European starlings. Oecologia 145, 326–333 (2005). https://doi.org/10.1007/s00442-005-0120-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-005-0120-6