Abstract
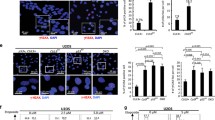
VACM-1/CUL5 is a member of the cullin family of proteins involved in the E3 ligase-dependent degradation of diverse proteins that regulate cellular proliferation. The ability of VACM-1/CUL5 to inhibit cellular growth is affected by its posttranslational modifications and its localization to the nucleus. Since the mechanism of VACM-1/CUL5 translocation to the nucleus is not clear, the goal of this project was to determine the role that the putative nuclear localization signal (NLS) we identified in the VACM-1/CUL5 (640PKLKRQ646) plays in the cellular localization of VACM-1/CUL5 and its effect on cellular growth. We used site-directed mutagenesis to change Lys642 and Lys644 to Gly and the mutated cDNA constructs were transfected into COS-1 cells. Mutation of the NLS in VACM-1/CUL5 significantly reduced its localization to the nucleus and compromised its effect on cellular growth. We have shown previously that the antiproliferative effect of VACM-1/CUL5 could be reversed by mutation of PKA-specific phosphorylation sequence (S730AVACM-1/CUL5), which was associated with its increased nuclear localization and modification by NEDD8. Thus, we examined whether these properties can be controlled by the NLS. The mutation of NLS in S730AVACM-1/CUL5 cDNA compromised its proliferative effect and reduced its localization to the nucleus. The immunocytochemistry results showed that, in cells transfected with the mutant cDNAs, the nuclear NEDD8 signal was decreased. Western blot analysis of total cell lysates, however, showed that VACM-1/CUL5 neddylation was not affected. Together, these results suggest that the presence of the NLS, both in VACM-1/CUL5 and in S730AVACM-1/CUL5 sequences, is critical for their control of cell proliferation.





Similar content being viewed by others
Abbreviations
- NEDD8:
-
Neural precursor cell expressed, developmentally down-regulated protein 8
- NLS:
-
Nuclear localization signal
- PKA:
-
Protein kinase A
- RAMEC:
-
Rat adrenal medullary endothelial cells
- VACM-1/CUL5:
-
Vasopressin-activated calcium-mobilizing (VACM-1) protein/cullin5
References
Baxter SS, Carlson LA, Mayer AM, Gall ML, Fay MJ (2009) Granulocytic differentiation of HL-60 promyelocytic leukemia cells is associated with increased expression of Cul5. In Vitro Cell Dev Biol Anim 45:264–274
Bradley SE, Johnson AE, Le IP, Oosterhouse E, Hledin MP, Marquez GA, Burnatowska-Hledin M (2010) Phosphorylation of VACM-1/Cul5 by protein kinase A regulates its neddylation and antiproliferative effect. J Biol Chem 285:4883–4895
Burnatowska-Hledin MA, Spielman WS, Smith WL, Shi P, Meyer JM, Dewitt DL (1995) Expression cloning of an AVP-activated, calcium-mobilizing receptor from rabbit kidney medulla. Am J Physiol 268:F1198–F1210
Burnatowska-Hledin M, Zeneberg A, Roulo A, Grobe J, Zhao P, Lelkes PI, Clare P, Barney C (2001) Expression of VACM-1 protein in cultured rat adrenal endothelial cells is linked to the cell cycle. Endothelium 8:49–63
Byrd PJ, Stankovic T, McConville CM, Smith AD, Cooper PR, Taylor AM (1997) Identification and analysis of expression of human VACM-1, a cullin gene family member located on chromosome. Genome Res 7:71–75
d’Azzo A, Bongiovanni A, Nastasi T (2005) E3 ubiquitin ligases as regulators of membrane protein trafficking and degradation. Traffic 6:429–441
Deshaies RJ, Emberley ED, Saha A (2010) Control of cullin-RING ubiquitin ligase activity by Nedd8. Conjugation and deconjugation of ubiquitin family modifiers. Subcell Biochem 54:41–56
Duda DM, Borg LA, Scott DC, Hunt HW, Hammel M, Schulman BA (2008) Structural insights into NEDD8 activation of CULLIN-RING ligases: conformational control of conjugation. Cell 134:995–1006
Fay MJ, Longo KA, Karathanasis GA, Shope DM, Mandernach CJ, Leong JR, Hicks A, Pherson K, Husian A (2003) Analysis of cul-5 expression in breast epithelial cells, breast cancer cell lines, normal tissues and tumor tissues. Mol Cancer 2:40–56
Furukawa M, Zhang Y, McCarville J, Ohta T, Xiong Y (2000) The CUL1 C-Terminal sequence and ROC1 are required for efficient nuclear accumulation, NEDD8 modification, and ubiquitin ligase activity of CUL1. Mol Cell Biol 20:8195–8197
Harreman MT, Kline TM, Milford HG, Harben MB, Hodel AE, Corbett AH (2004) Regulation of nuclear import by phosphorylation adjacent to nuclear localization signals. J Biol Chem 279:20613–20621
Hori T, Osaka F, Miyamoto C, Okabayashi K, Shimbara N, Kato S, Tanaka K (1999) Covalent modification of all members of human cullin family proteins by NEDD8. Oncogene 18:6829–6834
Johnson AE, Le IP, Buchwalter A, Burnatowska-Hledin M (2007) Estrogen dependent growth and estrogen receptor (ER)-α concentration in T47D Breast cancer cells are inhibited by VACM-1, a cul-5 Gene. Mol Cell Biochem 301:13–20
Kalla C, Scheuermann MO, Kube I, Schlotter M, Mertens D, Döhner H, Stilgenbauer S, Lichter P (2007) Analysis of 11q22-q23 deletion target genes in B-cell chronic lymphocytic leukemia: evidence for a pathogenic role of NPAT, CUL5, and PPP2R1B. Eur J Cancer 43:1328–1335
Kamitani T, Kito K, Nguyen HP, Yeh ET (1997) Characterization of NEDD8, a developmentally down-regulated ubiquitin- like protein. J Biol Chem 272:28557–28562
Kipreos ET, Lander LE, Wing JP, He WW, Hedgecock EM (1996) Cul-1 is required for cell cycle exit in C. elegans and identifies a novel gene family. Cell 85:829–839
Kurz T, Chou Y, Willems AR, Meyer-Schaller N, Hecht ML, Tyers M, Peter M, Sicheri F (2008) Dcn1 functions as a scaffold-type E3 ligase for cullin neddylation. Mol Cell 29:23–35
Lange A, Mills RE, Lange CJ, Stewart M, Devine SE, Corbett AH (2007) Classical nuclear localization signals: definition, function, and interaction with importin α. J Biol Chem 282:5101–5105
Laszlo GS, Cooper JA (2009) Restriction of Src activity by Cullin-5. Curr Biol 19:157–162
Liu B, Sarkis PT, Luo K, Yu Y, Yu XF (2005) Regulation of Apobec3F and human immunodeficiency virus type 1 Vif by Vif-Cul5-ElonB/C E3 ubiquitin ligase. J Virol 79:9579–9587
Luo M, Jones S, Flamand N, Aronoff DM, Peters-Golden M, Brock TG (2005) Phosphorylation by protein kinase A inhibits nuclear import of 5-lipoxygenase. J Biol Chem 280:40609–40616
Mathias N, Johnson SL, Winey M, Adams AE, Goetsch L, Pringle JR, Byers B, Goebl MG (1996) Cdc53p acts in concert with Cdc4p and Cdc34p to control the G1to-S-phase transition and identifies a conserved family of proteins. Mol Cell Biol 16:6634–6643
Nakai K, Kanehisa M (1992) A knowledge base for predicting protein localization sites in eucaryotic cells. Genomics 14:897–911
Nardozzi JD, Lott K, Cingolani G (2010) Phosphorylation meetsnuclear import: a review. Cell Commun Signal 8:32
Petroski MD, Deshaies RJ (2005) Function and regulation of cullin-RING ubiquitin ligases. Nat Rev Mol Cell Biol 6:9–20
Rowland RR, Yoo D (2003) Nucleolar-cytoplasmic shuttling of PRRSV nucleocapsid protein: a simple case of molecular mimicry or the complex regulation by nuclear import, nucleolar localization and nuclear export signal sequences. Virus Res 95:23–33
Soucy TA, Smith PG, Milhollen MA, Berger AJ, Gavin JM, Adhikari S, Brownell JE, Burke KE, Cardin DP, Critchley S, Cullis CA, Douchette A, Garnsey JJ, Gaulin JL, Gershman RE, Lublinsky AR, McDonald A, Mizutani H, Narayanan U, Olhava EJ, Peluso S, Rezaei M, Sintchak MD, Talreja T, Thomas MP, Traore T, Vyskocil S, Weatherhead GS, Yu J, Zhang J, Dick LR, Claiborne CF, Rolfe M, Bolen JB, Langston SP (2009) An inhibitor of NEDD8-activating enzyme as a new approach to treat cancer. Nature 458:732–736
Swords RT, Kelly KR, Smith PG, Garnsey JJ, Mahalingam D, Medina E, Oberheu K, Padmanabhan S, O’Dwyer M, Nawrocki ST, Giles FJ, Carew JS (2010) Inhibition of NEDD8-activating enzyme: a novel approach for the treatment of acute myeloid leukemia. Blood 115:3796–3800
Tateishi K, Omata M, Tanaka K, Chiba T (2001) The NEDD8 system is essential for cell cycle progression and morphogenetic pathway in mice. J Cell Biol 155:571–579
Van Dort C, Zhao P, Parmelee K, Capps B, Poel A, Listenberger L, Kossoris J, Wasilevich B, Murrey D, Clare P, Burnatowska-Hledin M (2003) VACM-1, a cul-5 gene, inhibits cellular growth by a mechanism that involves MAPK and p53 signaling pathways. Am J Physiol 285:C1386–C1396
Wormald S, Hilton DJ (2004) Inhibitors of cytokine signal transduction. J Biol Chem 279:821–824
Yu X, Yu Y, Liu B, Luo K, Kong W, Mao P, Yu XF (2003) Induction of APOBEC3G ubiquitination and degradation by an HIV-1 Vif-Cul5-SCF complex. Science 302:1056–1060
Zou Y, Mi J, Cui J, Lu D, Zhang X, Guo C, Gao G, Liu Q, Chen B, Shao C, Gong Y (2009) Characterization of nuclear localization signal in the N terminus of Cul4B and its essential role in cyclin E degradation and cell cycle progression. J Biol Chem 284:33320–33332
Acknowledgments
This work was supported by NIH (RO1DK47199) and by NCI (R15CA104014). In addition, we acknowledge a Beckman Foundation Award to S. Dean and grants from the National Science Foundation Research Experience for Undergraduates Programs (DBI-0754293, CHE-851194) that supported A. Willis. We also acknowledge the editorial assistance by Elizabeth Brigger.
Authors’ contributions
ANW and JH designed and constructed all mutant cDNAs and SH prepared and screened all plasmids. SEBD and MLL performed growth assays while BTK, ADS and SPL performed transfections, Western blot analysis and immuno staining. RG performed western blot analysis and MABH performed cytotoxicity assay. The first three authors were involved in writing and editing the manuscript. All authors read and approved the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing interests.
Additional information
Angelica N. Willis, Shirley E. Bradley Dean and Joe A. Habbouche contributed equally to this work.
Rights and permissions
About this article
Cite this article
Willis, A.N., Dean, S.E.B., Habbouche, J.A. et al. Nuclear localization signal sequence is required for VACM-1/CUL5-dependent regulation of cellular growth. Cell Tissue Res 368, 105–114 (2017). https://doi.org/10.1007/s00441-016-2522-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-016-2522-7