Abstract
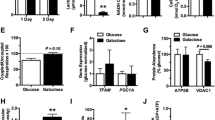
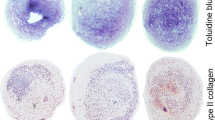
Low oxygen tension may change the dependence of chondrocytes on exogenous carbohydrate sources. In this study, we have investigated whether glucosamine sulphate (GS) stimulates proteoglycan synthesis, the mRNA expression of aggrecan and of type II collagen, and UDP-sugar levels in bovine primary chondrocytes under a low oxygen (O2) atmosphere. Chondrocytes from bovine femoral condyles were cultivated with or without GS or sulphate at various concentrations in low- (5.5 mM) or high-glucose (25 mM) DMEM under either a 5% or 20% O2 atmosphere for 2 or 8 days after isolation. The mRNA expression of aggrecan and type II collagen and the synthesis of glycosaminoglycan (GAG) were determined by quantitative real-time reverse transcription with polymerase chain reaction and a [35S]-sulphate incorporation assay, respectively. Aggrecan promoter activity was analysed by a dual-luciferase reporter gene assay. Intracellular UDP-N-acetylhexosamines (UDP-HexN), UDP-glucuronic acid and UDP-hexoses were analysed by reversed-phase high-performance liquid chromatography electrospray ionization mass spectrometry. A low (5%) O2 atmosphere significantly increased GAG synthesis, mRNA expression of aggrecan and of type II collagen and aggrecan promoter activity in bovine primary chondrocytes. A high (1 mM) concentration of GS was required to increase the level of UDP-HexN. However, GS did not increase GAG synthesis, aggrecan promoter activity or mRNA expression of aggrecan and of type II collagen. Interestingly, a 5% O2 atmosphere increased the level of UDP-HexN in 8-day cultures without GS treatment. Thus, exogenous GS does not change chondrocyte metabolism, whereas a 5% O2 atmosphere stimulates extracellular matrix production in bovine primary chondrocytes. The balance of UDP-sugars is changed under a 5% O2 atmosphere for longer culture periods.





Similar content being viewed by others
References
Bassleer C, Rovati L, Franchimont P (1998) Stimulation of proteoglycan production by glucosamine sulfate in chondrocytes isolated from human osteoarthritic articular cartilage in vitro. Osteoarthritis Cartilage 6:427–434
Beaudry F, Vachon P (2008) Determination of glucosamine in horse plasma by liquid chromatography tandem mass spectrometry. Biomed Chromatogr 22:1–4
Brighton CT, Heppenstall RB (1971) Oxygen tension in zones of the epiphyseal plate, the metaphysis and diaphysis. An in vitro and in vivo study in rats and rabbits. J Bone Joint Surg Am 53:719–728
Clegg DO, Reda DJ, Harris CL, Klein MA, O’Dell JR, Hooper MM, Bradley JD, Bingham CO 3rd, Weisman MH, Jackson CG, Lane NE, Cush JJ, Moreland LW, Schumacher HR Jr, Oddis CV, Wolfe F, Molitor JA, Yocum DE, Schnitzer TJ, Furst DE, Sawitzke AD, Shi H, Brandt KD, Moskowitz RW, Williams HJ (2006) Glucosamine, chondroitin sulfate, and the two in combination for painful knee osteoarthritis. N Engl J Med 354:795–808
Coimbra IB, Jimenez SA, Hawkins DF, Piera-Velazquez S, Stokes DG (2004) Hypoxia inducible factor-1 alpha expression in human normal and osteoarthritic chondrocytes. Osteoarthritis Cartilage 12:336–345
Dodge GR, Jimenez SA (2003) Glucosamine sulfate modulates the levels of aggrecan and matrix metalloproteinase-3 synthesized by cultured human osteoarthritis articular chondrocytes. Osteoarthritis Cartilage 11:424–432
Domm C, Schunke M, Christesen K, Kurz B (2002) Redifferentiation of dedifferentiated bovine articular chondrocytes in alginate culture under low oxygen tension. Osteoarthritis Cartilage 10:13–22
Etherington PJ, Winlove P, Taylor P, Paleolog E, Miotla JM (2002) VEGF release is associated with reduced oxygen tensions in experimental inflammatory arthritis. Clin Exp Rheumatol 20:799–805
Felson DT (2008) Glucosamine sulfate might have no effect on pain or structural changes associated with ostoearthritis. Nat Clin Pract Rheumatol 4:518–519
Fenton JI, Chlebek-Brown KA, Peters TL, Caron JP, Orth MW (2000) Glucosamine HCl reduces equine articular cartilage degradation in explant culture. Osteoarthritis Cartilage 8:258–265
Fermor B, Christensen SE, Youn I, Cernanec JM, Davies CM, Weinberg JB (2007) Oxygen, nitric oxide and articular cartilage. Eur Cell Mater 13:56–65
Ferrell WR, Najafipour H (1992) Changes in synovial PO2 and blood flow in the rabbit knee joint due to stimulation of the posterior articular nerve. J Physiol (Lond) 449:607–617
Gouze JN, Bianchi A, Becuwe P, Dauca M, Netter P, Magdalou J, Terlain B, Bordji K (2002) Glucosamine modulates IL-1-induced activation of rat chondrocytes at a receptor level, and by inhibiting the NF-kappaB pathway. FEBS Lett 510:166–170
Grimshaw MJ, Mason RM (2000) Bovine articular chondrocyte function in vitro depends upon oxygen tension. Osteoarthritis Cartilage 8:386–392
Grimshaw MJ, Mason RM (2001) Modulation of bovine articular chondrocyte gene expression in vitro by oxygen tension. Osteoarthritis Cartilage 9:357–364
Hansen U, Schunke M, Domm C, Ioannidis N, Hassenpflug J, Gehrke T, Kurz B (2001) Combination of reduced oxygen tension and intermittent hydrostatic pressure: a useful tool in articular cartilage tissue engineering. J Biomech 34:941–949
Hoffer LJ, Kaplan LN, Hamadeh MJ, Grigoriu AC, Baron M (2001) Sulfate could mediate the therapeutic effect of glucosamine sulfate. Metabolism 50:767–770
Kurz B, Domm C, Jin M, Sellckau R, Schunke M (2004) Tissue engineering of articular cartilage under the influence of collagen I/III membranes and low oxygen tension. Tissue Eng 10:1277–1286
Lammi MJ, Qu CJ, Laasanen MS, Saarakkala S, Rieppo J, Jurvelin JS, Töyräs J (2004) Undersulfated chondroitin sulfate does not increase in osteoarthritic cartilage. J Rheumatol 31:2449–2453
Lane JM, Brighton CT, Menkowitz BJ (1977) Anaerobic and aerobic metabolism in articular cartilage. J Rheumatol 4:334–342
Largo R, Alvarez-Soria MA, Diez-Ortego I, Calvo E, Sanchez-Pernaute O, Egido J, Herrero-Beaumont G (2003) Glucosamine inhibits IL-1beta-induced NFkappaB activation in human osteoarthritic chondrocytes. Osteoarthritis Cartilage 11:290–298
Lee RB, Urban JP (1997) Evidence for a negative Pasteur effect in articular cartilage. Biochem J 321:95–102
Lund-Olesen K (1970) Oxygen tension in synovial fluids. Arthritis Rheum 13:769–776
Mattei M de, Pellati A, Pasello M, Terlizzi F de, Massari L, Gemmati D, Caruso A (2002) High doses of glucosamine-HCl have detrimental effects on bovine articular cartilage explants cultured in vitro. Osteoarthritis Cartilage 10:816–825
Moussavi-Harami F, Duwayri Y, Martin JA, Moussavi-Harami F, Buckwalter JA (2004) Oxygen effects on senescence in chondrocytes and mesenchymal stem cells: consequences for tissue engineering. Iowa Orthop J 24:15–20
Mroz PJ, Silbert JE (2003) Effects of [3H]glucosamine concentration on [3H]chondroitin sulphate formation by cultured chondrocytes. Biochem J 376:511–515
Mroz PJ, Silbert JE (2004) Use of 3H-glucosamine and 35S-sulfate with cultured human chondrocytes to determine the effect of glucosamine concentration on formation of chondroitin sulfate. Arthritis Rheum 50:3574–3579
Murphy CL, Polak JM (2004) Control of human articular chondrocyte differentiation by reduced oxygen tension. J Cell Physiol 199:451–459
Najafipour H, Ferrell WR (1995) Comparison of synovial PO2 and sympathetic vasoconstrictor responses in normal and acutely inflamed rabbit knee joints. Exp Physiol 80:209–220
Neil KM, Orth MW, Coussens PM, Chan PS, Caron JP (2005) Effect of glucosamine and chondroitin sulfate on mediators of osteoarthritis in cultured equine chondrocytes stimulated by use of recombinant equine interleukin-1beta. Am J Vet Res 66:1861–1869
Parkkinen JJ, Lammi MJ, Helminen HJ, Tammi M (1992) Local stimulation of proteoglycan synthesis in articular cartilage explants by dynamic compression in vitro. J Orthop Res 10:610–620
Pavelka K, Gatterova J, Olejarova M, Machacek S, Giacovelli G, Rovati LC (2002) Glucosamine sulfate use and delay of progression of knee osteoarthritis: a 3-year, randomized, placebo-controlled, double-blind study. Arch Intern Med 162:2113–2123
Persiani S, Roda E, Rovati LC, Locatelli M, Giacovelli G, Roda A (2005) Glucosamine oral bioavailability and plasma pharmacokinetics after increasing doses of crystalline glucosamine sulfate in man. Osteoarthritis Cartilage 13:1041–1049
Piconi L, Quagliaro L, Assaloni R, Da Ros R, Maier A, Zuodar G, Ceriello A (2006) Constant and intermittent high glucose enhances endothelial cell apoptosis through mitochondrial superoxide overproduction. Diabetes Metab Res Rev 22:198–203
Qu CJ, Karjalainen HM, Helminen HJ, Lammi MJ (2006) The lack of effect of glucosamine sulphate on aggrecan mRNA expression and 35S-sulphate incorporation in bovine primary chondrocytes. Biochim Biophys Acta 1762:453–459
Qu CJ, Jauhiainen M, Auriola S, Helminen HJ, Lammi MJ (2007a) Effects of glucosamine sulfate on intracellular UDP-hexosamine and UDP-glucuronic acid levels in bovine primary chondrocytes. Osteoarthritis Cartilage 15:773–779
Qu CJ, Rieppo J, Hyttinen MM, Lammi MJ, Kiviranta I, Kurkijarvi J, Jurvelin JS, Töyräs J (2007b) Human articular cartilage proteoglycans are not undersulfated in osteoarthritis. Connect Tissue Res 48:27–33
Rajpurohit R, Koch CJ, Tao Z, Teixeira CM, Shapiro IM (1996) Adaptation of chondrocytes to low oxygen tension: relationship between hypoxia and cellular metabolism. J Cell Physiol 168:424–432
Reginster JY, Deroisy R, Rovati LC, Lee RL, Lejeune E, Bruyere O, Giacovelli G, Henrotin Y, Dacre JE, Gossett C (2001) Long-term effects of glucosamine sulphate on osteoarthritis progression: a randomised, placebo-controlled clinical trial. Lancet 357:251–256
Rozendaal RM, Koes BW, Osch GJ van, Uitterlinden EJ, Garling EH, Willemsen SP, Ginai AZ, Verhaar JA, Weinans H, Bierma-Zeinstra SM (2008) Effect of glucosamine sulphate on hip osteoarthritis: a randomized trial. Ann Intern Med 148:268–277
Saini S, Wick TM (2004) Effect of low oxygen tension on tissue-engineered cartilage construct development in the concentric cylinder bioreactor. Tissue Eng 10:825–832
Sandy JD, Gamett D, Thompson V, Verscharen C (1998) Chondrocyte-mediated catabolism of aggrecan: aggrecanase-dependent cleavage induced by interleukin-1 or retinoic acid can be inhibited by glucosamine. Biochem J 335:59–66
Scherer K, Schunke M, Sellckau R, Hassenpflug J, Kurz B (2004) The influence of oxygen and hydrostatic pressure on articular chondrocytes and adherent bone marrow cells in vitro. Biorheology 41:323–333
Suzuki N, Svensson K, Eriksson UJ (1996) High glucose concentration inhibits migration of rat cranial neural crest cells in vitro. Diabetologia 39:401–411
Terry DE, Rees-Milton K, Pruss C, Hopwood J, Carran J, Anastassiades TP (2007) Modulation of articular chondrocyte proliferation and anionic glycoconjugate synthesis by glucosamine (GlcN), N-acetyl GlcN (GlcNAc) GlcN sulfate salt (GlcN.S) and covalent glucosamine sulfates (GlcN-SO4). Osteoarthritis Cartilage 15:946–956
Treuhaft PS, McCarty DJ (1971) Synovial fluid pH, lactate, oxygen and carbon dioxide partial pressure in various joint diseases. Arthritis Rheum 14:475–484
Tsai TT, Danielson KG, Guttapalli A, Oguz E, Albert TJ, Shapiro IM, Risbud MV (2006) TonEBP/OREBP is a regulator of nucleus pulposus cell function and survival in the intervertebral disc. J Biol Chem 281:25416–25424
Uitterlinden EJ, Jahr H, Koevoet JL, Jenniskens YM, Bierma-Zeinstra SM, Degroot J, Verhaar JA, Weinans H, Osch GJ van (2006) Glucosamine decreases expression of anabolic and catabolic genes in human osteoarthritic cartilage explants. Osteoarthritis Cartilage 14:250–257
Varghese S, Theprungsirikul P, Sahani S, Hwang N, Yarema KJ, Elisseef JH (2007) Glucosamine modulates chondrocyte proliferation, matrix synthesis, and gene expression. Osteoarthritis Cartilage 15:59–68
Vlad SC, LaValley MP, McAlindon TE, Felson DT (2007) Glucosamine for pain in osteoarthritis: why do trial results differ? Arthritis Rheum 56:2267–2277
Wang DW, Fermor B, Gimble JM, Awad HA, Guilak F (2005) Influence of oxygen on the proliferation and metabolism of adipose derived adult stem cells. J Cell Physiol 204:184–191
Wernike E, Li Z, Alini M, Grad S (2008) Effect of reduced oxygen tension and long-term mechanical stimulation on chondrocyte-polymer constructs. Cell Tissue Res 331:473–483
Ye H, Zheng Y, Ma W, Ke D, Jin X, Liu S, Wang D (2005) Hypoxia down-regulates secretion of MMP-2, MMP-9 in porcine pulmonary artery endothelial and smooth muscle cells and the role of HIF-1. J Huazhong Univ Sci Technolog Med Sci 25:407
Zhou S, Cui Z, Urban JP (2004) Factors influencing the oxygen concentration gradient from the synovial surface of articular cartilage to the cartilage-bone interface: a modeling study. Arthritis Rheum 50:3915–3924
Acknowledgement
We are grateful to Dr. Hannu Karjalainen for discussions and tests involving reporter gene analysis, to Dr. Kari Törrönen for help with the computer and to Mr. Kari Kotikumpu for his assistance with oxygen tension measurements during this study. We express our special thanks to statistician Mrs. Marja-Leena Hannila for checking the statistical analyses of this study.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported by the Sigrid Juselius Foundation.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
441_2009_797_MOESM1_ESM.xls
Ct-values from nine animals (Normal O2 20% O2 tension, Low O2 5% O2 tension, GAPDH D-glyceraldehyde-3-phosphate dehydrogenase, GS glucosamine sulphate, S sulphate) (XLS 24 kb)
Rights and permissions
About this article
Cite this article
Qu, CJ., Pöytäkangas, T., Jauhiainen, M. et al. Glucosamine sulphate does not increase extracellular matrix production at low oxygen tension. Cell Tissue Res 337, 103–111 (2009). https://doi.org/10.1007/s00441-009-0797-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-009-0797-7