Abstract
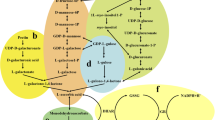
Ascorbic acid (AsA) is an important nutrient in the human body and performs various healthy functions. With considerable medicinal properties, celery (Apium graveolens L.) could be a good source of AsA for human health. However, the biosynthetic, recycling, and degradation pathways of AsA in celery have yet to be characterized. To study the metabolic pathways involved in AsA, the genes involved in AsA biosynthesis, recycling, and degradation were isolated from celery, and their expression profiles and AsA levels were analyzed in the leaf blades and petioles of two celery varieties at three different growth stages. AsA levels were higher in ‘Ventura’ compared with ‘Liuhehuangxinqin’ in both tissues possibly because of different transcription levels of genes, such as l-galactose dehydrogenase (GalDH), l-galactono-1,4-lactone dehydrogenase (GalLDH), and glutathione reductase (GR). Results revealed that the d-mannose/l-galactose pathway may be the predominant pathway in celery, and the d-galacturonic acid pathway appeared to contribute largely to AsA accumulation in petioles than in leaf blades in ‘Liuhehuangxinqin.’ AsA contents are regulated by complex regulatory mechanisms and vary at different growth stages, tissues, and varieties in celery. The results provide novel insights into AsA metabolic pathways in leaf during celery growth and development.







Similar content being viewed by others
Abbreviations
- APX:
-
Ascorbate peroxidase
- AsA:
-
Ascorbic acid
- AO:
-
Ascorbate oxidase
- DHAR:
-
Dehydroascorbate reductase
- d-Man:
-
d-Mannose
- EtOH:
-
Ethyl alcohol
- GalDH:
-
l-Galactose dehydrogenase
- GalLDH:
-
l-Galactono-1,4-lactone dehydrogenase
- GalUR:
-
d-Galacturonate reductase
- GGP:
-
GDP-l-galactose phosphorylase
- GME:
-
GDP-d-mannose-3′,5′-epimerase
- GMP:
-
GDP-d-mannose pyrophosphorylase
- GPP:
-
l-Galactose-1-P phosphatase
- GR:
-
Glutathione reductase
- l-Gal:
-
l-Galactose
- MDHAR:
-
Monodehydroascorbate reductase
- MIOX:
-
Myo-inositol oxygenase
- PGI:
-
Phosphoglucose isomerase
- PMI:
-
Phosphomannose isomerase
- PMM:
-
Phosphomannose mutase
- qRT-PCR:
-
Quantitative real-time polymerase chain reaction
- TCA:
-
Trichloroacetic acid
- T-AsA:
-
Total ascorbic acid
References
Agius F, Gonzalez-Lamothe R, Caballero JL, Munoz-Blanco J, Botella MA, Valpuesta V (2003) Engineering increased vitamin C levels in plants by overexpression of a d-galacturonic acid reductase. Nat Biotechnol 21:177–181
Alhagdow M, Mounet F, Gilbert L, Nunes-Nesi A, Garcia V, Just D, Petit J, Beauvoit B, Fernie AR, Rothan C, Baldet P (2007) Silencing of the mitochondrial ascorbate synthesizing enzyme l-galactono-1,4-lactone dehydrogenase affects plant and fruit development in tomato. Plant Physiol 145:1408–1422
Alos E, Rodrigo MJ, Zacarias L (2013) Transcriptomic analysis of genes involved in the biosynthesis, recycling and degradation of l-ascorbic acid in pepper fruits (Capsicum annuum L.). Plant Sci 207:2–11
Alos E, Rodrigo MJ, Zacarias L (2014) Differential transcriptional regulation of l-ascorbic acid content in peel and pulp of citrus fruits during development and maturation. Planta 239:1113–1128
Badejo AA, Eltelib HA, Fukunaga K, Fujikawa Y, Esaka M (2009a) Increase in ascorbate content of transgenic tobacco plants overexpressing the acerola (Malpighia glabra) phosphomannomutase gene. Plant Cell Physiol 50:423–428
Badejo AA, Fujikawa Y, Esaka M (2009b) Gene expression of ascorbic acid biosynthesis related enzymes of the Smirnoff–Wheeler pathway in acerola (Malpighia glabra). J Plant Physiol 166:652–660
Bulley SM, Rassam M, Hoser D, Otto W, Schünemann N, Wright M, MacRae E, Gleave A, Laing W (2009) Gene expression studies in kiwifruit and gene over-expression in Arabidopsis indicates that GDP-l-galactose guanyltransferase is a major control point of vitamin C biosynthesis. J Exp Bot 60:765–778
Chatterjee IB (1973) Evolution and the biosynthesis of ascorbic acid. Science 182:1271–1272
Cocetta G, Karppinen K, Suokas M, Hohtola A, Häggman H, Spinardi A, Mignani I, Jaakola L (2012) Ascorbic acid metabolism during bilberry (Vaccinium myrtillus L.) fruit development. J Plant Physiol 169:1059–1065
Davey MW, Montagu MV, Inze D, Sanmartin M, Kanellis A, Smirnoff N, Benzie IJJ, Strain JJ, Favell D, Fletcher J (2000) Plant l-ascorbic acid: chemistry, function, metabolism, bioavailability and effects of processing. J Sci Food Agric 80:825–860
Dianat M, Veisi A, Ahangarpour A, Fathi Moghaddam H (2015) The effect of hydro-alcoholic celery (Apiumgraveolens) leaf extract on cardiovascular parameters and lipid profile in animal model of hypertension induced by fructose. Avicenna J Phytomed 5:203–209
Eltayeb AE, Kawano N, Badawi GH, Kaminaka H, Sanekata T, Shibahara T, Inanaga S, Tanaka K (2007) Overexpression of monodehydroascorbate reductase in transgenic tobacco confers enhanced tolerance to ozone, salt and polyethylene glycol stresses. Planta 225:1255–1264
Frei B, Lawson S (2008) Vitamin C and cancer revisited. Proc Natl Acad Sci USA 105:11037–11038
Gallie DR (2013a) l-ascorbic Acid: a multifunctional molecule supporting plant growth and development. Scientifica. doi:10.1155/2013/795964
Gallie DR (2013b) The role of l-ascorbic acid recycling in responding to environmental stress and in promoting plant growth. J Exp Bot 64:433–443
Gatzek S, Wheeler GL, Smirnoff N (2002) Antisense suppression of l-galactose dehydrogenase in Arabidopsis thaliana provides evidence for its role in ascorbate synthesis and reveals light modulated l-galactose synthesis. Plant J 30:541–553
Gest N, Garchery C, Gautier H, Jimenez A, Stevens R (2013) Light-dependent regulation of ascorbate in tomato by a monodehydroascorbate reductase localized in peroxisomes and the cytosol. Plant Biotechnol J 11:344–354
Gilbert L, Alhagdow M, Nunes-Nesi A, Quemener B, Guillon F, Bouchet B, Faurobert M, Gouble B, Page D, Garcia V, Petit J, Stevens R, Causse M, Fernie AR, Lahaye M, Rothan C, Baldet P (2009) GDP-d-mannose 3,5-epimerase (GME) plays a key role at the intersection of ascorbate and non-cellulosic cell-wall biosynthesis in tomato. Plant J 60:499–508
Hancock RD, Viola R (2005) Improving the nutritional value of crops through enhancement of l-ascorbic acid (vitamin C) content: rationale and biotechnological opportunities. J Agric Food Chem 53:5248–5257
Hemavathi Upadhyaya CP, Young KE, Akula N, Kim HS, Heung JJ, Oh OM, Aswath CR, Chun SC, Kim DH, Park SW (2009) Over-expression of strawberry d-galacturonic acid reductase in potato leads to accumulation of vitamin C with enhanced abiotic stress tolerance. Plant Sci 177:659–667
Holler S, Ueda Y, Wu L, Wang Y, Hajirezaei MR, Ghaffari MR, von Wirén N, Frei M (2015) Ascorbate biosynthesis and its involvement in stress tolerance and plant development in rice (Oryza sativa L.). Plant Mol Biol 88:545–560
Imai T, Niwa M, Ban Y, Hirai M, Oba K, Moriguchi T (2009) Importance of the l-galactonolactone pool for enhancing the ascorbate content revealed by l-galactonolactone dehydrogenase-overexpressing tobacco plants. Plant Cell Tissue Org 96:105–112
Ioannidi E, Kalamaki MS, Engineer C, Pateraki I, Alexandrou D, Mellidou I, Giovannonni J, Kanellis AK (2009) Expression profiling of ascorbic acid-related genes during tomato fruit development and ripening and in response to stress conditions. J Exp Bot 60:663–678
Kampfenkel K, Vanmontagu M, Inze D (1995) Extraction and determination of ascorbate and dehydroascorbate from plant tissue. Anal Biochem 225:165–167
Kojo S (2004) Vitamin C: basic metabolism and its function as an index of oxidative stress. Curr Med Chem 11:1041–1064
Laing WA, Wright MA, Cooney J, Bulley SM (2007) The missing step of the l-galactose pathway of ascorbate biosynthesis in plants, an l-galactose guanyltransferase, increases leaf ascorbate content. Proc Natl Acad Sci USA 104:9534–9539
Li Y, Schellhorn HE (2007) New developments and novel therapeutic perspectives for vitamin C. J Nutr 137:2171–2184
Li MJ, Ma FW, Zhang M, Pu F (2008) Distribution and metabolism of ascorbic acid in apple fruits (Malus domestica Borkh cv. Gala). Plant Sci 174:606–612
Li M, Ma F, Shang P, Zhang M, Hou C, Liang D (2009) Influence of light on ascorbate formation and metabolism in apple fruits. Planta 230:39–51
Li M, Ma F, Guo C, Liu J (2010) Ascorbic acid formation and profiling of genes expressed in its synthesis and recycling in apple leaves of different ages. Plant Physiol Biochem 48:216–224
Li MJ, Chen XS, Wang PP, Ma FW (2011) Ascorbic acid accumulation and expression of genes involved in its biosynthesis and recycling in developing apple fruit. J Am Soc Hortic Sci 136:231–238
Li MY, Wang F, Jiang Q, Ma J, Xiong AS (2014) Identification of SSRs and differentially expressed genes in two cultivars of celery (Apium graveolens L.) by deep transcriptome sequencing. Hortic Res 1:10. doi:10.1038/hortres.2014.10
Li MY, Wang F, Jiang Q, Wang GL, Tian C, Xiong AS (2016) Validation and comparison of reference genes for qPCR normalization of celery (Apium graveolens) at different development stages. Front Plant Sci 7:313. doi:10.3389/fpls.2016.00313
Linster CL, Clarke SG (2008) l-Ascorbate biosynthesis in higher plants: the role of VTC2. Trends Plant Sci 13:567–573
Lorence A, Chevone BI, Mendes P, Nessler CL (2004) Myo-inositol oxygenase offers a possible entry point into plant ascorbate biosynthesis. Plant Physiol 134:1200–1205
Marin A, Ferreres F, Tomas-Barberan FA, Gil MI (2004) Characterization and quantitation of antioxidant constituents of sweet pepper (Capsicum annuum L.). J Agric Food Chem 52:3861–3869
Massot C, Stevens R, Genard M, Longuenesse JJ, Gautier H (2012) Light affects ascorbate content and ascorbate-related gene expression in tomato leaves more than in fruits. Planta 235:153–163
Noctor G, Foyer CH (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49:249–279
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Qin AG, Shi QH, Yu XC (2011) Ascorbic acid contents in transgenic potato plants overexpressing two dehydroascorbate reductase genes. Mol Biol Rep 38:1557–1566
Ren J, Chen ZW, Duan WK, Song XM, Liu TK, Wang JJ, Hou XL, Li Y (2013) Comparison of ascorbic acid biosynthesis in different tissues of three non-heading Chinese cabbage cultivars. Plant Physiol Biochem 73:229–236
Smirnoff N (2011) Vitamin C: the metabolism and functions of ascorbic acid in plants. Adv Bot Res 59:107–177
Smirnoff N, Wheeler GL (2000) Ascorbic acid in plants: biosynthesis and function. Crit Rev Biochem Mol Biol 35:291–314
Wang ZN, Xiao Y, Chen WS, Tang KX, Zhang L (2010) Increased vitamin C content accompanied by an enhanced recycling pathway confers oxidative stress tolerance in Arabidopsis. J Integr Plant Biol 52:400–409
Wang GL, Sun S, Xing GM, Wu XJ, Wang F, Xiong AS (2015a) Morphological characteristics, anatomical structure, and gene expression: novel insights into cytokinin accumulation during carrot growth and development. PLoS One 10:e0134166
Wang GL, Xu ZS, Wang F, Li MY, Tan GF, Xiong AS (2015b) Regulation of ascorbic acid biosynthesis and recycling during root development in carrot (Daucus carota L.). Plant Physiol Biochem 94:10–18
Wheeler GL, Jones MA, Smirnoff N (1998) The biosynthetic pathway of vitamin C in higher plants. Nature 393:365–369
Wieczorek J, Sienkiewicz S, Pietrzak M, Wieczorek Z (2015) Uptake and phytotoxicity of anthracene and benzo[k]fluoranthene applied to the leaves of celery plants (Apium graveolens var. secalinum L.). Ecotoxicol Environ Saf 115:19–25
Wolucka BA, Goossens A, Inze D (2005) Methyl jasmonate stimulates the de novo biosynthesis of vitamin C in plant cell suspensions. J Exp Bot 56:2527–2538
Xu Y, Zhu XW, Chen YL, Gong YQ, Liu LW (2013) Expression profiling of genes involved in ascorbate biosynthesis and recycling during fleshy root development in radish. Plant Physiol Biochem 70:269–277
Zhang YY, Li HX, Shu WB, Zhang CJ, Zhang W, Ye ZB (2011) Suppressed expression of ascorbate oxidase gene promotes ascorbic acid accumulation in tomato fruit. Plant Mol Biol Rep 29:638–645
Acknowledgments
The research was supported by Jiangsu Natural Science Foundation (BK20130027), Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), and Shanxi Province Coal Based Key Scientific and Technological Project (FT201402-07).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This article does not contain any studies with human participants or animal performed by any of the authors.
Conflict of interest
The authors declare that there are no competing interests.
Additional information
Communicated by S. Hohmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Huang, W., Wang, GL., Li, H. et al. Transcriptional profiling of genes involved in ascorbic acid biosynthesis, recycling, and degradation during three leaf developmental stages in celery. Mol Genet Genomics 291, 2131–2143 (2016). https://doi.org/10.1007/s00438-016-1247-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-016-1247-3