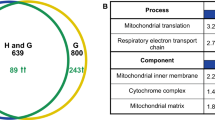
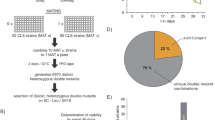
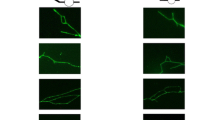
Abstract
Chromosome translocation is a major genomic event for a cell, affecting almost every of its life aspects ranging from metabolism, organelle maintenance and homeostasis to gene maintenance and expression. By using the bridge-induced translocation system, we defined the effects of induced chromosome translocation on the chronological life span (CLS) of yeast with particular interest to the oxidative stress condition. The results demonstrate that every translocant strain has a different CLS, but all have a high increase in reactive oxygen species and in lipid peroxides levels at the end of the life span. This could be due to the very unique and strong deregulation of the oxidative stress network. Furthermore, the loss of the translocated chromosome occurs at the end of the life span and is locus dependent. Additionally, the RDH54 gene may play a role in the correct segregation of the translocant chromosome, since in its absence there is an increase in loss of the bridge-induced translocated chromosome.





Similar content being viewed by others
References
Aust SD (1994) Thiobarbituric acid assay reactants. In: Tyson CA, Frazier JM (eds) In vitro toxicity indicators. Academic Press, San Diego, pp 367–375
Bakhoum SF, Thompson SL, Manning AL, Compton DA (2009) Genome stability is ensured by temporal control of kinetochore-microtubule dynamics. Nat Cell Biol 11:27–35
Belancio VP, Blask DE, Deininger P, Hill SM, Jazwinski SM (2015) The aging clock and circadian control of metabolism and genome stability. Front Genet 5:455
Benov L, Sztejnberg L, Fridovich I (1998) Critical evaluation of the use of hydroethidine as a measure of superoxide anion radical. Free Radic Biol Med 25:826–831
Bruschi CV, Howe GA (1988) High frequency FLP-independent homologous DNA recombination of 2-micron plasmid in the yeast Saccharomyces cerevisiae. Curr Genet 14:191–199
Duesberg P, Li R, Fabarius A, Hehlmann R (2005) The chromosomal basis of cancer. Cell Oncol 27:293–318
Fabrizio P, Longo VD (2003) The chronological life span of Saccharomyces cerevisiae. Aging Cell 2(2):73–81
Fabrizio P, Longo VD (2007) The chronological life span of Saccharomyces cerevisiae. Methods Mol Biol 371:89–95
Fabrizio P, Hoon S, Shamalnasab M, Galbani A, Wei M, Giaever G, Nislow C, Longo VD (2010) Genome-wide screen in Saccharomyces cerevisiae identifies vacuolar protein sorting, autophagy, biosynthetic, and tRNA methylation genes involved in life span regulation. PLoS Genet 6:e1001024. doi:10.1371/journal.pgen.1001024
Hu J, Wei M, Mirisola MG, Longo VD (2013) Assessing chronological aging in Saccharomyces cerevisiae. Methods Mol Biol 965:463–472. doi:10.1007/978-1-62703-239-130
Kaiser C, Michaelis S, Mitchell A (1994) Preparation of chromosome-sized yeast DNA molecules in solid agarose. Methods in yeast genetics. Cold Spring Harbor Laboratory Press, New York, pp 179–180
Klein HL (1997) Rdh54, a Rad54 homologue in Saccharomyces cerevisiae, is required for mitotic diploid-specific recombination and repair and for meiosis. Genetics 147:1533–1543
Klinger H, Rinnerthaler M, Lam YT, Laun P, Heeren G, Klocker A, Simon-Nobbe B, Dickinson JR, Dawes IW, Breitenbach M (2010) Quantitation of (a)symmetric inheritance of functional and of oxidatively damaged mitochondrial aconitase in the cell division of old yeast mother cells. Exp Gerontol 45:533–542
Kumaran R, Yang SY, Leu JY (2013) Characterization of chromosome stability in diploid, polyploid and hybrid yeast cells. PLoS One 8:e68094. doi:10.1371/journal.pone.0068094
Madeo F, Frohlich E, Frohlich KU (1997) A yeast mutant showing diagnostic markers of early and late apoptosis. J Cell Biol 139:729–734
Madeo F, Fröhlich E, Ligr M, Grey M, Sigrist SJ, Wolf DH, Fröhlich KU (1999) Oxygen stress: a regulator of apoptosis in yeast. J Cell Biol 145(4):757–767
Madia F, Gattazzo C, Fabrizio P, Longo VD (2007) A simple model system for age-dependent DNA damage and cancer. Mech Ageing Dev 128(1):45–49
Madia F, Wei M, Yuan V, Hu J, Gattazzo C, Pham P, Goodman MF, Longo VD (2009) Oncogene homologue Sch9 promotes age-dependent mutations by a superoxide and Rev1/Polzeta-dependent mechanism. J Cell Biol 186:509–523
Martins D, English AM (2014) Catalase activity is stimulated by H(2)O(2) in rich culture medium and is required for H(2)O(2) resistance and adaptation in yeast. Redox Biol 2:308–313
Maxwell PH, Burhans WC, Curcio MJ (2011) Retrotransposition is associated with genome instability during chronological aging. Proc Natl Acad Sci USA 108:20376–20381
Mazurik VK, Mikhaĭlov VF, Ushenkova LN, Raeva NF (2004) Sexual differences in a state systems of DNA structure maintenance and generation of reactive oxygen species in somatic cells of mice 101/H reparation-defective strain and manifestation of these differences after exposure to ionizing radiation. Radiats Biol Radioecol 44:516–523
Moraitis C, Curran BP (2007) Can the different heat shock response thresholds found in fermenting and respiring yeast cells be attributed to their differential redox states? Yeast 24:653–666
Mostafavi S, Ray D, Warde-Farley D, Grouios C, Morris Q (2008) GeneMANIA: a real-time multiple association network integration algorithm for predicting gene function. Genome Biol 9(Suppl 1):S4. doi:10.1186/gb-2008-9-s1-s4
Nikitin D, Tosato V, Zavec AB, Bruschi CV (2008) Cellular and molecular effects of nonreciprocal chromosome translocations in Saccharomyces cerevisiae. Proc Natl Acad Sci USA 105:9703–9708
Nolan T, Hands RE, Bustin SA (2006) Quantification of mRNA using real-time RT-PCR. Nat Protoc 1:1559–1582
Powers RW 3rd, Kaeberlein M, Caldwell SD, Kennedy BK, Fields S (2006) Extension of chronological life span in yeast by decreased TOR pathway signaling. Genes Dev 20:174–184
Rinnerthaler M, Büttner S, Laun P, Heeren G, Felder TK, Klinger H, Weinberger M, Stolze K, Grousl T, Hasek J, Benada O, Frydlova I, Klocker A, Simon-Nobbe B, Jansko B, Breitenbach-Koller H, Eisenberg T, Gourlay CW, Madeo F, Burhans WC, Breitenbach M (2012) Yno1p/Aim14p, a NADPH-oxidase ortholog, controls extramitochondrial reactive oxygen species generation, apoptosis, and actin cable formation in yeast. Proc Natl Acad Sci USA 109:8658–8663
Rossi B, Noel P, Bruschi CV (2010) Different aneuploidies arise from the same bridge-induced chromosomal translocation event in Saccharomyces cerevisiae. Genetics 186:775–790
Rowe LA, Degtyareva N, Doetsch PW (2008) DNA damage-induced reactive oxygen species (ROS) stress response in Saccharomyces cerevisiae. Free Radic Biol Med 45:1167–1177
Sambrook J, Fritsch EF, Maniatis T (1989) Analysis of genomic DNA by southern hybridization. Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, New York, p 9.31
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C (T) method. Nat Protoc 3:1101–1108
Teste MA, Duquenne M, François JM, Parrou JL (2009) Validation of reference genes for quantitative expression analysis by real-time RT-PCR in Saccharomyces cerevisiae. BMC Mol Biol 10:99
Thompson SL, Compton DA (2008) Examining the link between chromosomal instability and aneuploidy in human cells. JCB 180:665–672
Torres EM, Sokolsky T, Tucker CM, Chan LY, Boselli M, Dunham MJ, Amon A (2007) Effects of aneuploidy on cellular physiology and cell division in haploid yeast. Science 317:916–924
Tosato V, Waghmare SK, Bruschi CV (2005) Non-reciprocal chromosomal bridge-induced translocation (BIT) by targeted DNA integration in yeast. Chromosoma 114:15–27
Tosato V, Nicolini C, Bruschi CV (2009) DNA bridging of yeast chromosomes VIII leads to near-reciprocal translocation and loss of heterozygosity with minor cellular defects. Chromosoma 118:179–191
Tosato V, Grüning NM, Breitenbach M, Arnak R, Ralser M, Bruschi CV (2013a) Warburg effect and translocation-induced genomic instability: two yeast models for cancer cells. Front Oncol 2:212
Tosato V, Sidari S, Bruschi CV (2013b) Bridge-induced chromosome translocation in yeast relies upon a Rad54/Rdh54-dependent, Pol32-independent pathway. PLoS One 8:e60926. doi:10.1371/journal.pone.0060926
Acknowledgments
The authors wish to thank Dmitri Nikitin and Beatrice Rossi for technical help and suggestions and Frank Madeo for making available the facilities for the apoptosis experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
VT was supported by Crescendo Biologics Ltd., Cambridge, UK.
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by S. Hohmann.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sims, J., Bruschi, C.V., Bertin, C. et al. High reactive oxygen species levels are detected at the end of the chronological life span of translocant yeast cells. Mol Genet Genomics 291, 423–435 (2016). https://doi.org/10.1007/s00438-015-1120-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00438-015-1120-9