Abstract
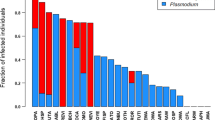
Avian haemosporidians make up one of the most widely distributed and diverse vector borne parasite systems, found nearly worldwide in tropical and temperate areas. Despite the clear relationship between avian host fitness measures and infection, few studies have addressed the importance of source material selection when assessing these relationships. We show that source material, here blood and pectoral muscle, do not yield equivalent results when assessing prevalence and genetic diversity of haemosporidian genera. We find higher prevalence and genetic diversity are recovered from blood versus pectoral muscle for Haemoproteus. Contrastingly, we find that a higher prevalence of Plasmodium is detected from pectoral muscle, while higher genetic diversity is recovered from blood. Our results indicate that source material may bias parasite detection and be an important factor in study design, which is not only related to parasite infection, but by extension to the ecology and fitness of avian hosts.


Similar content being viewed by others
References
Adler PH, Crosskey RW (2015) World blackflies (Diptera: Simuliidae): a comprehensive revision of the taxonomic and geographical inventory. 123 pp. Available from: http://www.clemson.edu/cafls/biomia/pdfs/blackflyinventory.pdf
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Applegate JE, Beaudoin RL (1970) Mechanism of spring relapse in avian malaria: effect of gonadotropin and corticosterone. J Wildl Dis 6:443–447
Asghar M, Hasselquist D, Bensch S (2011) Are chronic avian haemosporidian infections costly in wild birds? Avian Biol 42:530–537
Atkinson CT, Forrester DJ, Greiner EC (1988) Pathogenicity of Haemoproteus meleagridis (Haemosporina: Haemoproteidae) in experimentally infected domestic turkeys. J Parasitol 74:228–239
Atkinson CT, Greiner EC, Forrester DJ (1986) Pre-erythrocytic development and associated host responses to Haemoproteus meleagridis (Haemosporina: Haemoproteidae) in experimentally infected domestic Turkeys. J Protozool 33:375–381
Atkinson CT, Van Riper C (1991) Pathogenicity and epizootiology of avian haematozoa: Plasmodium, Haemoproteus, and Leucocytozoon. In: Loye JE, Zuk M (eds) Bird-parasite interactions: ecology, evolution, and behavior. Oxford University Press, Oxford, UK, pp 19–48
Bandelt H, Forster P, Röhl A (1999) Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol 16:37–48
Beadell JS, Covas R, Gebhard C, Ishtiaq F, Melo M, Schmidt BK, Perkins SL, Graves GR, Fleischer RC (2009) Host associations and evolutionary relationships of avian blood parasites from West Africa. Int J Parasitol 39:257–266
Bennett GF, Cameron M (1974) Seasonal prevalence of avian hematozoa in passeriform birds of Atlantic Canada. Can J Zoo 52:1259–1264
Bennett GF, Whiteway M, Woodworth-Lynas C (1982) A host-parasite catalogue of the avian haematozoa. Memorial University of Newfoundland Occasional Papers in Biology, no. 5, St. John's, Newfoundland
Bensch S, Hellgren O, Pérez-Tris J (2009) MalAvi: a public database of malaria parasites and related haemosporidians in avian hosts based on mitochondrial cytochrome b lineages. Mol Ecol Resour 9:1353–1358
Cannell BL, Krasnec KV, Campbell K, Jones HI, Miller RD, Stephens N (2013) The pathology and pathogenicity of a novel Haemoproteus spp. infection in wild Little Penguins (Eudyptula minor). Vet Parasitol 197:74–84
Clark NJ, Clegg SM, Lima MR (2014) A review of global diversity in avian Haemosporidians (Plasmodium and Haemoproteus: Haemosporida): new insights from molecular data. Int J Parasitol 44:329–338
Coon CAC, García-Longoria L, Martin LB, Magallanes S, de Lope F, Marzal A (2016) Malaria infection negatively affects feather growth rate in the house sparrow Passer domesticus. J Avian Biol 47:779–787
Cornelius JM, Zylberberg M, Breuner CW, Gleiss AC, Hahn TP (2014) Assessing the role of reproduction and stress in the spring emergence of haematozoan parasites in birds. J Exp Biol 217:841–849
Desser SS, Fallis AM, Garnham PCC (1968) Relapses in ducks chronically infected with Leucocytozoon simondi and Parahaemoproteus nettionis. Can J Zool 46:281–285
Deviche P, Greiner AC, Manteca X (2001) Seasonal and age-related changes in blood parasite prevalence in dark- eyed juncos (Junco hyemalis, Aves, Passeriformes). J Exp Zool 289:456–466
Donovan TA, Schrenzel M, Tucker TA, Pessier AP, Stalis IH (2008) Hepatic hemorrhage, hemocoelom, and sudden death due to Haemoproteus infection in passerine birds: eleven cases. J Vet Diagn Investig 20:304–313
Drovetski SV, Aghayan SA, Mata VA, Lopes RJ, Mode NA, Harvey JA, Voelker G (2014) Does the niche breadth or trade-off hypothesis explain the abundance-occupancy relationship in avian Haemosporidia? Mol Ecol 23:3322–3329
Dunn JC, Stockdale JE, Bradford EL, Mccubbin A, Morris AJ, Grice PV, Goodman J, Hamer KC (2016) High rates of infection by blood parasites during the nestling phase in UK Columbids with notes on ecological associations. Parasitology:1–7
Fair J, Paul E, Jones J (eds) (2010) Guidelines to the use of wild birds in research. Ornithological Council, Washington, D.C. URL www.nmnh.si.edu/BIRDNET/guide
Fallis AM, Bennett GF (1960) Description of Haemoproteus canachites n. sp. (Sporozoa: Haemoproteidae) and sporogony in Culicoides (Diptera: Ceraptogonidae). Can J Zool 38:455–464
Fay MP (2010) Two-sided exact tests and matching confidence intervals for discrete data. R Journal 2:53–58
García-Longoria L, Garamszegi LZ, Møller AP (2014) Host escape behavior and blood parasite infections in birds. Behav Ecol 00:1–11
Greiner EC, Mundy PJ (1979) Hematozoa from Southern African vultures, with a description of Haemoproteus janovyi sp. n. J Parasitol 65:147–153
Hamilton WD, Zuk M (1982) Heritable true fitness and bright birds: a role for parasites? Science 218:384–387
Hasselquist D, Östman Ö, Waldenström J, Bensch S (2007) Temporal patterns of occurrence and transmission of the blood parasite Haemoproteus payevskyi in the great reed warbler Acrocephalus arundinaceus. J Ornithol 148:401–409
Hau M, Perfito N, Moore IT (2008) Timing of breeding in tropical birds: mechanisms and evolutionary implications. Ornitol Neotrop 19:39–59
Hepler PK, Huff CG, Helmuth S (1966) The fine structure of the exoerythrocytic stages of Plasmodium fallax. J Cell Biol 30:333–358
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Climatol 25:1965–1978
Holmstad PR, Anwar A, Iezhova T, Skorping A (2003) Standard sampling techniques underestimate prevalence of avian hematozoa in willow ptarmigan (Lagopus lagopus). J Wildl Dis 39:354–358
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C, Thierer T, Ashton B, Mentjies P, Drummond A (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28:1647–1649
Khan RA, Fallis AM (1970) Relapses in birds infected with species of Leucocytozoon. Can J Zool 48:451–455
Knowles SCL, Palinauskas V, Sheldon BC (2010) Chronic malaria infections increase family inequalities and reduce parental fitness: experimental evidence from a wild bird population. J Evol Biol 23:557–569
Librado P, Rozas J (2009) DnaSP v5: a software for comprehensive analysis of DNA polymorphism data. Bioinformatics 25:1451–1452
Linder HP, de Klerk HM, Born J, Burgess ND, Fjeldså J, Rahbek C (2012) The partitioning of Africa: statistically defined biogeographical regions in sub-Saharan Africa. J Biogeogr 39:1189–1205
Lutz HL, Hochachka WM, Engel JI, Bell JA, Tkach VV, Bates JM, Hackett SJ, Weckstein JD (2015) Parasite prevalence corresponds to host life history in a diverse assemblage of Afrotropical birds and haemosporidian parasites. PLoS One 10:e0121254
Markus MB (2011) The hypnozoite concept, with particular reference to malaria. Parasitol Res 108:247–252
Marzal A, de Lope F, Navarro C, Møller AP (2005) Malarial parasites decrease reproductive success: an experimental study in a passerine bird. Oecologia 142:541–545
Merino S, Moreno J, Sanz JJ, Arriero E (2000) Are avian blood parasites pathogenic in the wild? A medication experiment in blue tits (Parus caeruleus). Proc Biol Sci 267:2507–2510
Mukhin A, Palinauskas V, Platonova E, Kobylkov D, Vakoliuk I, Valkiunas G (2016) The strategy to survive primary malaria infection: an experimental study on behavioural changes in parasitized birds. PLoS One 11:1–15
Olias P, Wegelin M, Zenker W, Freter S, Gruber AD, Klopfleisch R (2011) Avian malaria deaths in parrots, Europe. Emerg Infect Dis 17:950–952
Outlaw DC, Harvey JA, Drovetski SV, Voelker G (2017) Diversity and distribution of avian haemosporidians in sub-Saharan Africa: an inter-regional biogeographic overview. Parasitology 144:394–402
Peel MC, Finlayson BC, Mcmahon TA (2007) Updated world map of the Köppen-Geiger climate classification. Hydrol Earth Syst Sci 4:439–473
Pérez-Rodríguez A, de la Hera I, Bensch S, Pérez-Tris J (2015) Evolution of seasonal transmission patterns in avian blood-borne parasites. Int J Parasitol 45:605–611
R Core Team (2016) R: a language and environment for statistical computing. R foundation for statistical computing, Vienna, Austria. URL https://www.R-project.org/
Ramey AM, Fleskes JP, Schmutz JA, Yabsley MJ (2013) Evaluation of blood and wing pectoral muscle tissues for molecular detection and characterization of hematozoa infections in northern pintails (Anas acuta) wintering in California. Int J Parasitol Parasites Wildl 2:102–109
Sorensen MC, Asghar M, Bensch S, Fairhurst GD, Jenni-Eiermann S, Spottiswoode CN (2016) A rare study from the wintering grounds provides insight into the costs of malaria infection for migratory birds. J Avian Biol 47:1–8
Stephens M, Smith N, Donnelly P (2001) A new statistical method for haplotype reconstruction from population data. Am J Hum Genet 68:978–989
Sutcliffe JF (1986) Black fly host location: a review. Can J Zool 64:1041–1053
Svensson-coelho M, Silva GT, Santos SS, Miranda LS, Ricklefs RE, Miyaki CY, Maldonado-Coelho M (2016) Lower detection probability of avian plasmodium in blood compared to other tissues. J Parasit Dis 102:559–561
Szymanski MM, Lovette IJ (2005) High lineage diversity and host sharing of malarial parasites in a local avian assemblage. J Parasitol 91:768–774
Valkiūnas G (2005) Avian malaria parasites and other haemosporidia. CRC Press. Boca Raton, FL, Boca Raton
Valkiūnas G, Bairlein F, Iezhova TA, Dolnik OV (2004) Factors affecting the relapse of Haemoproteus belopolskyi infections and the parasitaemia of Trypanosoma spp. in a naturally infected European songbird, the blackcap, Sylvia atricapilla. Parasitol Res 93:218–222
Valkiūnas G, Iezhova TA, Palinauskas V, Ilgūnas M, Bernotienė R (2015) The evidence for rapid gametocyte viability changes in the course of parasitemia in Haemoproteus parasites. Parasitol Res 114:2903–2909
van Riper IIIC, van Riper SG, Goff ML, Laird M (1986) The Epizootiology and ecological significance of malaria in Hawaiian land birds. Ecol Monogr 56:327
Ya'cob Z, Takaoka H, Pramual P, Low VL, Sofian-Azirun M (2016) Breeding habitat preference of preimaginal black flies (Diptera: Simuliidae) in peninsular Malaysia. Acta Trop 153:57–63
Young BE, Garvin MC, McDonald DB (1993) Blood parasites in birds from Monteverde, Costa Rica. J Wildl Dis 29:555–560
Zehtindjiev P, Ilieva M, Westerdahl H, Hansson B, Valkiūnas G, Bensch S (2008) Dynamics of parasitemia of malaria parasites in a naturally and experimentally infected migratory songbird, the great reed warbler Acrocephalus arundinaceus. Exp Parasitol 119:99–110
Acknowledgements
We are grateful to the country of Benin for the permission to perform this research. We would like to thank Alphonse Adite for the invaluable assistance in obtaining permits and field collecting and Robert Adite for his driving prowess. We thank Dr. Toby J. Hibbitts and Jerry W. Huntley for the field collections of samples. We are grateful to Carter Atkinson for proposing valuable comments and suggestions that improved the manuscript. We would also like to thank Jerry Huntley and Jonathan Puritz for the much need insightful comments on an earlier draft of the paper. We also thank Danielle Walkup for the helpful discussions on the material along the way. This is publication number 1552 of the Biodiversity, Research, and Teaching Collections at Texas A&M University. This research received no specific grant from any funding agency, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Online Resources 1
Host species taxonomic information including, locality sampled (Chutes de Koudou (C), Point Triplo (P), Dogo Forest (D) Lama Forest (L), Lake Toho (T), Abomey Calavi (A), number of individuals sampled (N), samples greater then 1 per locality listed, and frequency of detection across Haemoproteus (H), Leucocytozoon (L), Plasmodium (P), bioregion sampled in, novel lineages recovered, MalAvi lineages recovered (XLSX 44 kb)
Rights and permissions
About this article
Cite this article
Harvey, J.A., Voelker, G. Avian haemosporidian detection across source materials: prevalence and genetic diversity. Parasitol Res 116, 3361–3371 (2017). https://doi.org/10.1007/s00436-017-5654-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-017-5654-0