Abstract
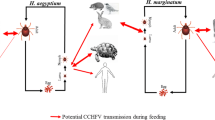
Crimean-Congo hemorrhagic fever virus (CCHFV) is transmitted to humans mainly through the bite of infected ticks. In Greece, only one clinical case has been observed, in 2008, but the seroprevalence in humans is relatively high (4.2%). To have a first insight into the circulation of CCHFV in Greece, 2000 ticks collected from livestock during 2012–2014 were tested. CCHFV was detected in 36 of the 1290 (2.8%) tick pools (1–5 ticks per pool). Two genetic lineages were identified: Europe 1 and Europe 2. Most Europe 1 sequences were obtained from Rhipicephalus sanguineus sensu lato ticks, while most Europe 2 sequences were recovered from Rhipicephalus bursa ticks. The number of collected Hyalomma marginatum ticks (the principal vector of CCHFV) was low (0.5% of ticks) and all were CCHFV negative. Since it is not known how efficient ticks of the Rhipicephalus genus are as vectors of the virus, laboratory studies will be required to explore the role of Rhipicephalus spp. ticks in CCHFV maintenance and transmission.


Similar content being viewed by others
References
Antoniadis A, Casals J (1982) Serological evidence of human infection with Congo-Crimean hemorrhagic fever virus in Greece. Am J Trop Med Hyg 31:1066–1067
Bente DA, Forrester NL, Watts DM, McAuley AJ, Whitehouse CA, Bray M (2013) Crimean-Congo hemorrhagic fever: history, epidemiology, pathogenesis, clinical syndrome and genetic diversity. Antivir Res 100:159–189
Elevli M, Ozkul AA, Civilibal M, Midilli K, Gargili A, Duru NS (2009) A newly identified Crimean-Congo hemorrhagic fever virus strain in Turkey. Int J Infect Dis 14(Suppl 3):e213–e216
Estrada-Peña A, Bouattour A, Camicas JL, Walker AR (2004) Ticks of domestic animals in the Mediterranean region. A Guide to Identification of Species. University of Zaragoza Press, Zaragoza
Gargili A, Estrada-Pena A, Spengler JR, Lukashev A, Nuttall PA, Bente DA (2017) The role of ticks in the maintenance and transmission of Crimean-Congo hemorrhagic fever virus: a review of published field and laboratory studies. Antivir Res 144:93–119
Honig JE, Osborne JC, Nichol ST (2004) The high genetic variation of viruses of the genus Nairovirus reflects the diversity of their predominant tick hosts. Virology 318:10–16
Kumar S, Stecher G, Tamura K (2016) MEGA7: Molecular Evolutionary Genetics Analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Midilli K, Gargili A, Ergonul O, Elevli M, Ergin S, Turan N, Sengoz G, Ozturk R, Bakar M (2009) The first clinical case due to AP92 like strain of Crimean-Congo hemorrhagic fever virus and a field survey. BMC Infect Dis 9:90
OIE Crimean-Congo Haemorrhagic Fever OIE Terrestrial Manual 2014
Ozkaya E, Dincer E, Carhan A, Uyar Y, Ertek M, Whitehouse CA, Ozkul A (2010) Molecular epidemiology of Crimean-Congo hemorrhagic fever virus in Turkey: occurrence of local topotype. Virus Res 149:64–70
Panayotova E, Papa A, Trifonova I, Christova I (2016) Crimean-Congo hemorrhagic fever virus lineages Europe 1 and Europe 2 in Bulgarian ticks. Ticks Tick Borne Dis 7:1024–1028
Papa A, Maltezou HC, Tsiodras S, Dalla VG, Papadimitriou T, Pierroutsakos I, Kartalis GN, Antoniadis A (2008) A case of Crimean-Congo haemorrhagic fever in Greece, June 2008. Euro Surveill 13
Papa A, Sidira P, Kallia S, Ntouska M, Zotos N, Doumbali E, Maltezou H, Demiris N, Tsatsaris A (2013) Factors associated with IgG positivity to Crimean-Congo hemorrhagic fever virus in the area with the highest seroprevalence in Greece. Ticks Tick Borne Dis 4:417–420
Papa A, Chaligiannis I, Kontana N, Sourba T, Tsioka K, Tsatsaris A, Sotiraki S (2014) A novel AP92-like Crimean-Congo hemorrhagic fever virus strain, Greece. Ticks Tick Borne Dis 5:590–593
Papa A, Mirazimi A, Koksal I, Estrada-Pena A, Feldmann H (2015) Recent advances in research on Crimean-Congo hemorrhagic fever. J Clin Virol 64:137–143
Papa A, Sidira P, Tsatsaris A (2016) Spatial cluster analysis of Crimean-Congo hemorrhagic fever virus seroprevalence in humans, Greece. Parasit Epidemiol Control 1:211–218
Papadopoulos O, Koptopoulos G (1978) Isolation of Crimean-Congo hemorrhagic fever (C-CHF) virus from Rhipicephalus bursa ticks in Greece. Acta Hell Microbiol 23:20–28
Reeves WC (1957) Arthropods as vectors and reservoirs of animal pathogenic viruses. In: Hallauer C, Meyer KF (eds) Handbuch der Virusforschung. Springer-Verlag, Vienna, pp 177–202
Rodriguez LL, Maupin GO, Ksiazek TG, Rollin PE, Khan AS, Schwarz TF, Lofts RS, Smith JF, Noor AM, Peters CJ, Nichol ST (1997) Molecular investigation of a multisource outbreak of Crimean-Congo hemorrhagic fever in the United Arab Emirates. Am J Trop Med Hyg 57:512–518
Salehi-Vaziri M, Baniasadi V, Jalali T, Mirghiasi SM, Azad-Manjiri S, Zarandi R, Mohammadi T, Khakifirouz S, Fazlalipour M (2016) The first fatal case of Crimean-Congo hemorrhagic fever caused by the AP92-like strain of the Crimean-Congo hemorrhagic fever virus. Jpn J Infect Dis 69:344–346
Schuster I, Mertens M, Mrenoshki S, Staubach C, Mertens C, Bruning F, Wernike K, Hechinger S, Berxholi K, Mitrov D, Groschup MH (2016) Sheep and goats as indicator animals for the circulation of CCHFV in the environment. Exp Appl Acarol 68:337–346
Shepherd AJ, Swanepoel R, Shepherd SP, Leman PA, Mathee O (1991) Viraemic transmission of Crimean-Congo haemorrhagic fever virus to ticks. Epidemiol Infect 106:373–382
Sherifi K, Cadar D, Muji S, Robaj A, Ahmeti S, Jakupi X, Emmerich P, Kruger A (2014) Crimean-Congo hemorrhagic fever virus clades V and VI (Europe 1 and 2) in ticks in Kosovo, 2012. PLoS Negl Trop Dis 8:e3168
Sidira P, Maltezou HC, Haidich AB, Papa A (2012) Seroepidemiological study of Crimean-Congo haemorrhagic fever in Greece, 2009-2010. Clin Microbiol Infect 18:E16–E19
Tekin S, Bursali A, Mutluay N, Keskin A, Dundar E (2011) Crimean-Congo hemorrhagic fever virus in various ixodid tick species from a highly endemic area. Vet Parasitol 186:546–552
Tonbak S, Aktas M, Altay K, Azkur AK, Kalkan A, Bolat Y, Dumanli N, Ozdarendeli A (2006) Crimean-Congo hemorrhagic fever virus: genetic analysis and tick survey in Turkey. J Clin Microbiol 44:4120–4124
Turell MJ (2007) Role of ticks in the transmission of Crimean-Congo Hemorrhagic fever virus. In: Ergonul O, Whitehouse CA (eds) Crimean-Congo Hemorrhagic Fever. Springer, Dordrecht, pp 143–154
Wolfel R, Paweska JT, Petersen N, Grobbelaar AA, Leman PA, Hewson R, Georges-Courbot MC, Papa A, Gunther S, Drosten C (2007) Virus detection and monitoring of viral load in Crimean-Congo hemorrhagic fever virus patients. Emerg Infect Dis 13:1097–1100
World Health Organization (2013) Crimean-Congo Haemorrhagic Fever. Fact sheet No. 208 - January 2013. http://www.who.int/mediacentre/factsheets/fs208/en/
Acknowledgements
The work was supported by ANTIGONE and COMPARE EU projects (grant agreement numbers 278976 and 643476, respectively).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Papa, A., Kontana, A., Tsioka, K. et al. Molecular detection of Crimean-Congo hemorrhagic fever virus in ticks, Greece, 2012–2014. Parasitol Res 116, 3057–3063 (2017). https://doi.org/10.1007/s00436-017-5616-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-017-5616-6