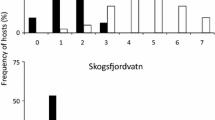
Abstract
Co-introduction and colonization of parasites with the introduction of new host species into aquatic habitats may depend on the host specificity and dispersal capabilities of the parasites. We compared the metazoan parasite community of an introduced three-spined stickleback (Gasterosteus aculeatus) population with that of the nearby source population in subarctic Norway. As expected from a small spatial scale (5 km), the parasite component communities in the two lakes were highly similar. All identifiable allogenic parasite taxa (Diphyllobothrium dendriticum, Diphyllobothrium ditremum, Diphyllobothrium spp., Schistocephalus solidus, Apatemon sp. and Diplostomum spp.) were also observed in both lakes while inter-lake differences were driven by autogenic parasite taxa (Eubothrium spp., Crepidostomum spp., Nematoda spp., Proteocephalus sp. and Gyrodactylus arcuatus). Contrary to expectation, the total number of parasite taxa was higher in the introduced stickleback population (12) compared to that found in the source population (9) with three parasite taxa (Eubothrium spp., Crepidostomum spp., Nematoda spp.) only occurring in the introduced population. These parasites were uncommon however and normally restricted to salmonids. Sticklebacks from both populations were heavily infected, particularly with eye-infecting metacercariae. Sequences from the DNA barcode region of cytochrome oxidase 1 indicated that these include Diplostomum lineage 6, a member of the Diplostomum baeri complex and a member of the Strigeinae. Despite high similarity between the two component communities, quantitative inter-lake differences were found at the infracommunity level. At this scale, parasite intensity was significantly higher in the source population for the two autogenic stickleback specialists: G. arcuatus and Proteocephalus sp., assumed to be the autogenic stickleback specialist Proteocephalus filicollis. Parasite infracommunities within each lake also resembled each other significantly more than infracommunities between lakes, primarily driven by the allogenic cestode D. ditremum, as well as G. arcuatus and Proteocephalus sp. Overall, quantitative dissimilarities between the two parasite communities were possibly explained by inter-lake differences in the density of sticklebacks and intermediate hosts.





Similar content being viewed by others
References
Amundsen PA, Klemetsen A, Grotnes PE (1993) Rehabilitation of a stunted population of Arctic char by intensive fishing. North Am J Fish Manage 13:483–491
Amundsen PA, Knudsen R, Klemetsen A (2007) Intraspecific competition and density dependence of food consumption and growth in Arctic charr. J Anim Ecol 76:149–158
Amundsen PA, Lafferty KD, Knudsen R, Primicerio R, Klemetsen A, Kuris AM (2009) Food web topology and parasites in the pelagic zone of a subarctic lake. J Anim Ecol 78:563–572
Amundsen PA, Lafferty KD, Knudsen R, Primicerio R, Kristoffersen R, Klemetsen A, Kuris AM (2013) New parasites and predators follow the introduction of two fish species to a subarctic lake: implications for food-web structure and functioning. Oecologia 171:993–1002
Anderson RM, May RM (1978) Regulation and stability of host-parasite population interactions: 1. Regulatory processes. J Anim Ecol 47:219–247
Arneberg P, Skorping A, Grenfell B, Read AF (1998) Host densities as determinants of abundance in parasite communities. Proc R Soc B Biol Sci 265:1283–1289
Barber I (2013) Sticklebacks as model hosts in ecological and evolutionary parasitology. Trends Parasitol 29:556–566
Behrmann-Godel J (2013) Parasite identification, succession and infection pathways in perch fry (Perca fluviatilis): new insights through a combined morphological and genetic approach. Parasitology 140:509–520
Blanar CA, Munkittrick KR, Houlahan J, Maclatchy DL, Marcogliese DJ (2009) Pollution and parasitism in aquatic animals: a meta-analysis of effect size. Aquat Toxicol 93:18–28
Blasco-Costa I, Faltýnková A, Georgieva S, Skírnisson K, Scholz T, Kostadinova A (2014) Fish pathogens near the Arctic Circle: molecular, morphological and ecological evidence for unexpected diversity of Diplostomum (Digenea: Diplostomidae) in Iceland. Int J Parasitol 44(10):703–715
Bush AO, Kennedy CR (1994) Host fragmentation and helminth parasites: hedging your bets against extinction. Int J Parasitol 24:1333–1343
Bush AO, Lafferty KD, Lotz JM, Shostak AW (1997) Parasitology meets ecology on its own terms: Margolis et al revisited. J Parasitol 83:575–583
Chappell LH (1969) The parasites of the three-spined stickleback Gasterosteus aculeatus L. from a Yorkshire pond. J Fish Biol 1:137–152
Chibwana FD, Blasco-Costa I, Georgieva S, Hosea KM, Nkvvengulila G, Scholz T, Kostadinova A (2013) A first insight into the barcodes for African diplostomids (Digenea: Diplostomidae): brain parasites in Clarias gariepinus (Siluriformes: Clariidae). Infect Genet Evol 17:62–70
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Aust J Ecol 18:117–143
Cleaveland S, Hess GR, Dobson AP, Laurenson MK, McCallum HI, Roberts MG, Woodroffe R (2002) The role of pathogens in biological conservation. In: Hudson PJ, Rizzoli A, Grenfell BT, Heesterbeek H, Dobson AP (eds) The ecology of wildlife diseases. Oxford University Press, Oxford, pp 139–150
Daszak P, Cunningham AA, Hyatt AD (2000) Emerging infectious diseases of wildlife—threats to biodiversity and human health. Science 287:443–449
Désilets HD, Locke SA, McLaughlin JD, Marcogliese DJ (2013) Community structure of Diplostomum spp. (Digenea: Diplostomidae) in eyes of fish: main determinants and potential interspecific interactions. Int J Parasitol 43:929–939
Dobson AP (1990) Models for multi-species parasite-host communities. In: Esch G, Bush A, Aho J (eds) Parasite communities: patterns and processes. Springer, Netherlands, pp 261–288
Esch GW (1971) Impact of ecological succession on parasite fauna in centrarchids from oligotrophic and eutrophic ecosystems. Am Midl Nat 86:160–168
Esch GW, Kennedy CR, Bush AO, Aho JM (1988) Patterns in helminth communities in freshwater fish in Great Britain: alternative strategies for colonization. Parasitology 96:519–532
Fellis KJ, Esch GW (2005) Variation in life cycle affects the distance decay of similarity among bluegill sunfish parasite communities. J Parasitol 91:1484–1486
Francová K, Ondracková M, Polacik M, Jurajda P (2011) Parasite fauna of native and non-native populations of Neogobius melanostomus (Pallas, 1814) (Gobiidae) in the longitudinal profile of the Danube River. J Appl Ichthyol 27:879–886
Garcia-Berthou E, Alcaraz C, Pou-Rovira Q, Zamora L, Coenders G, Feo C (2005) Introduction pathways and establishment rates of invasive aquatic species in Europe. Can J Fish Aquat Sci 62:453–463
Gendron A, Marcogliese DJ, Thomas M (2012) Invasive species are less parasitized than native competitors, but for how long? The case of the round goby in the Great Lakes-St. Lawrence Basin. Biol Invasions 14:367–384
Georgieva S, Soldánová M, Pérez-del-Olmo A, Dangel DR, Sitko J, Sures B, Kostadinova A (2013) Molecular prospecting for European Diplostomum (Digenea: Diplostomidae) reveals cryptic diversity. Int J Parasitol 43:57–72
Hoffman GL (1999) Parasites of North American freshwater fishes, 2nd edn. Cornell University Press, Ithaca
Jokela J, Lively CM (1995) Spatial variation in infection by digenetic trematodes in a population of freshwater snails (Potamopyrgus antipodarum). Oecologia 103:509–517
Jørgensen L, Klemetsen A (1995) Food resource partitioning of Arctic charr, Salvelinus alpinus (L.) and three-spined stickleback, Gasterosteus aculeatus L., in the littoral zone of lake Takvatn in northern Norway. Ecol Freshw Fish 4:77–84
Kalbe M, Wegner KM, Reusch TBH (2002) Dispersion patterns of parasites in 0+ year three-spined sticklebacks: a cross population comparison. J Fish Biol 60:1529–1542
Karvonen A, Kristjánsson BK, Skúlason S, Lanki M, Rellstab C, Jokela J (2013) Water temperature, not fish morph, determines parasite infections of sympatric Icelandic threespine sticklebacks (Gasterosteus aculeatus). Ecol Evol 3:1507–1517
Kelly DW, Paterson RA, Townsend CR, Poulin R, Tompkins DM (2009a) Parasite spillback: a neglected concept in invasion ecology? Ecology 90:2047–2056
Kelly DW, Paterson RA, Townsend CR, Poulin R, Tompkins DM (2009b) Has the introduction of brown trout altered disease patterns in native New Zealand fish? Freshw Biol 54:1805–1818
Kennedy CR (2009) The ecology of parasites of freshwater fishes: the search for patterns. Parasitology 136:1653–1662
Klemetsen A, Amundsen P-A, Grotnes PE, Knudsen R, Kristoffersen R, Svenning M-A (2002) Takvatn through 20 years: long-term effects of an experimental mass removal of Arctic charr, Salvelinus alpinus, from a subarctic lake. Environ Biol Fish 64:39–47
Klimpel S, Seehagen A, Palm HW (2003) Metazoan parasites and feeding behavior of four small-sized fish species from the central North Sea. Parasitol Res 91:290–297
Knudsen R, Klemetsen A, Staldvik F (1996) Parasites as indicators of individual feeding specialization in Arctic charr during winter in northern Norway. J Fish Biol 48:1256–1265
Knudsen R, Amundsen PA, Klemetsen A (2002) Parasite-induced host mortality: indirect evidence from a long-term study. Environ Biol Fish 64:257–265
Knudsen R, Curtis MA, Kristoffersen R (2004) Aggregation of helminths: the role of feeding behavior of fish hosts. J Parasitol 90:1–7
Kolar CS, Lodge DM (2002) Ecological predictions and risk assessment for alien fishes in North America. Science 298:1233–1236
Krebs CJ (1999) Ecological methodology, 2nd edn. Benjamin Cummings, Menlo Park
Levsen A (1992) Parasitter hos trepigget stingsild i Norge - med hovedvekt på trichodinide ciliater. Fauna 45:40–48 (in Norwegian)
Locke SA, McLaughlin JD, Dayanandan S, Marcogliese DJ (2010a) Diversity and specificity in Diplostomum spp. metacercariae in freshwater fishes revealed by cytochrome c oxidase I and internal transcribed spacer sequences. Int J Parasitol 40:333–343
Locke SA, McLaughlin JD, Marcogliese DJ (2010b) DNA barcodes show cryptic diversity and a potential physiological basis for host specificity among Diplostomoidea (Platyhelminthes: Digenea) parasitizing freshwater fishes in the St. Lawrence River, Canada. Mol Ecol 19:2813–2827
Locke SA, McLaughlin JD, Lapierre AR, Johnson PT, Marcogliese DJ (2011) Linking larvae and adults of Apharyngostrigea cornu, Hysteromorpha triloba, and Alaria mustelae (Diplostomoidea: Digenea) using molecular data. J Parasitol 97:846–851
Locke SA, Levy MS, Marcogliese DJ, Ackerman S, McLaughlin JD (2012) The decay of parasite community similarity in ring-billed gulls Larus delawarensis and other hosts. Ecography 35:530–538
Locke SA, McLaughlin JD, Marcogliese DJ (2013) Predicting the similarity of parasite communities in freshwater fishes using the phylogeny, ecology and proximity of hosts. Oikos 122:73–83
Locke SA, Marcogliese DJ, Valtonen ET (2014) Vulnerability and diet breadth predict larval and adult parasite diversity in fish of the Bothnian Bay. Oecologia 174:253–262
Lymbery AJ, Hassan M, Morgan DL, Beatty SJ, Doupé RG (2010) Parasites of native and exotic freshwater fishes in south-western Australia. J Fish Biol 76:1770–1785
MacLeod CJ, Paterson AM, Tompkins DM, Duncan RP (2010) Parasites lost—do invaders miss the boat or drown on arrival? Ecol Lett 13:516–527
Marcogliese DJ, Cone DK (1991) Do brook charr (Salvelinus fontinalis) from insular Newfoundland have different parasites than their mainland counterparts. Can J Zool 69:809–811
Marcogliese DJ, Cone DK (1996) On the distribution and abundance of eel parasites in Nova Scotia: influence of pH. J Parasitol 82:389–399
Moszczynska A, Locke SA, McLaughlin JD, Marcogliese DJ, Crease TJ (2009) Development of primers for the mitochondrial cytochrome c oxidase I gene in digenetic trematodes (Platyhelminthes) illustrates the challenge of barcoding parasitic helminths. Mol Ecol Resour 9:75–82
Ondrackova M, Francová K, Dávidová M, Polacik M, Jurajda P (2010) Condition status and parasite infection of Neogobius kessleri and N. melanostomus (Gobiidae) in their native and non-native area of distribution of the Danube River. Ecol Res 25:857–866
Pennycuick L (1971) Seasonal variations in the parasite infections in a population of three-spined sticklebacks, Gasterosteus aculeatus L. Parasitology 63:373–388
Pérez-del-Olmo A, Fernández M, Raga JA, Kostadinova A, Morand S (2009) Not everything is everywhere: the distance decay of similarity in a marine host-parasite system. J Biogeogr 36:200–209
Persson L, Amundsen PA, De Roos AM, Klemetsen A, Knudsen R, Primicerio R (2007) Culling prey promotes predator recovery—alternative states in a whole-lake experiment. Science 316:1743–1746
Poulin R (2003) The decay of similarity with geographical distance in parasite communities of vertebrate hosts. J Biogeogr 30:1609–1615
Poulin R (2007) Are there general laws in parasite ecology? Parasitology 134:763–776
Poulin R, Morand S (1999) Geographical distances and the similarity among parasite communities of conspecific host populations. Parasitology 119:369–374
Poulin R, Morand S (2004) Parasite biodiversity. Smithsonian Books, Washington
Poulin R, Mouillot D (2003) Host introductions and the geography of parasite taxonomic diversity. J Biogeogr 30:837–845
Poulin R, Mouillot D, George-Nascimento M (2003) The relationship between species richness and productivity in metazoan parasite communities. Oecologia 137:277–285
Poulin R, Blanar CA, Thieltges DW, Marcogliese DJ (2011) The biogeography of parasitism in sticklebacks: distance, habitat differences and the similarity in parasite occurrence and abundance. Ecography 34:540–551
R Core Team (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, http://www.R-project.Org/
Ratnasingham S, Hebert PDN (2013) A DNA-based registry for all animal species: the Barcode Index Number (BIN) system. PLoS One 8:e66213
Rellstab C, Louhi K-R, Karvonen A, Jokela J (2011) Analysis of trematode parasite communities in fish eye lenses by pyrosequencing of naturally pooled DNA. Infect Genet Evol 11:1276–1286
Roche DG, Leung B, Franco EFM, Torchin ME (2010) Higher parasite richness, abundance and impact in native versus introduced cichlid fishes. Int J Parasitol 40:1525–1530
Ruxton GD (2006) The unequal variance t-test is an underused alternative to Student's t-test and the Mann–Whitney U test. Behav Ecol 17:688–690
Scholz T, Hanzelová V, Skeriková A, Shimazu T, Rolbiecki L (2007) An annotated list of species of the Proteocephalus Weinland, 1858 aggregate sensu de Chambrier et al. (2004) (Cestoda: Proteocephalidea), parasites of fishes in the Palaearctic Region, their phylogenetic relationships and a key to their identification. Syst Parasitol 67:139–156
Skoglund S, Knudsen R, Amundsen P-A (2013) Selective predation on zooplankton by pelagic Arctic charr, Salvelinus alpinus, in six subarctic lakes. J Ichthyol 53:849–855
Sterud E (1999) Parasitter hos norske ferskvannsfisk. Norsk Zoologisk Foreining: rapport 7, Oslo (in Norwegian)
Takemoto RM, Pavanelli GC, Lizama MAP, Luque JL, Poulin R (2005) Host population density as the major determinant of endoparasite species richness in floodplain fishes of the upper Paraná River, Brazil. J Helminthol 79:75–84
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) Mega5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Torchin ME, Lafferty KD, Kuris AM (2002) Parasites and marine invasions. Parasitology 124:137–151
Torchin ME, Lafferty KD, Dobson AP, McKenzie VJ, Kuris AM (2003) Introduced species and their missing parasites. Nature 421:628–630
Valtonen ET, Marcogliese DJ, Julkunen M (2010) Vertebrate diets derived from trophically transmitted fish parasites in the Bothnian Bay. Oecologia 162:139–152
Van der Gucht K, Vandekerckhove T, Vloemans N, Cousin S, Muylaert K, Sabbe K, Gillis M, Declerk S, Meester LD, Vyverman W (2005) Characterization of bacterial communities in four freshwater lakes differing in nutrient load and food web structure. FEMS Microbiol Ecol 53:205–220
Vignon M, Sasal P, Galzin R (2009) Host introduction and parasites: a case study on the parasite community of the peacock grouper Cephalopholis argus (Serranidae) in the Hawaiian Islands. Parasitol Res 104:775–782
Visser M (1982) Prey selection by the three-spined stickleback (Gasterosteus aculeatus L.). Oecologia 55:395–402
Williamson MH (1996) Biological invasions. Chapman & Hall, London
Zander CD (2007) Parasite diversity of sticklebacks from the Baltic Sea. Parasitol Res 100:287–297
Acknowledgments
Thanks go to Professor Daniel McLaughlin at the Department of Biology, Concordia University, Canada for inviting the first author for a scientific stay which aided in the completion of this manuscript. Also, thanks go to UiT The Arctic University of Norway and the Norwegian Research Council for the financial support (NFR 213610/F20). Molecular work performed in connection to this study was supported by funding from NSERC and from the government of Canada through Genome Canada and the Ontario Genomics Institute in support of the international Barcode of Life project and by the Canadian federal government’s GRDI initiative.
Author information
Authors and Affiliations
Corresponding author
Appendix
Appendix
Photographs of selected parasites infecting three-spined sticklebacks in the present study. a Nematoda sp. found in the body cavity. b Diphyllobothrium spp. found in the body cavity and liver. c Cyathocephalus truncatus found in the intestine. d Schistocephalus solidus found in the body cavity. e Apatemon sp. found in the viscous humour of the eye. f Proteocephalus sp. found in the intestine. g Gyrodactylus arcuatus found on the skin and fins. h Diplostomum lineage 6 found in the viscous humour of the eye. i Strigeinae gen. sp. found in the viscous humour of the eye. Pictures are not to scale

Rights and permissions
About this article
Cite this article
Kuhn, J.A., Kristoffersen, R., Knudsen, R. et al. Parasite communities of two three-spined stickleback populations in subarctic Norway—effects of a small spatial-scale host introduction. Parasitol Res 114, 1327–1339 (2015). https://doi.org/10.1007/s00436-015-4309-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-015-4309-2