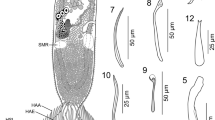
Abstract
Many methods have been proposed for collecting and fixing parasites, but most were written before the molecular age, and were intended to be practised by experienced parasitologists in well-equipped laboratories. We describe here a very simple method, illustrated by photographs, for collecting helminths from the digestive tract of vertebrates. It only requires a few plastic vials, some ethanol and a means to heat water. Basically, the method consists of: (a) the extraction of all organs from the abdominal cavity; (b) opening the digestive system longitudinally; (c) agitate gut and contents in a saline solution (i.e. ca. 9% NaCl or 1/4 sea water in tap water); (d) decant in saline as many times as needed to clean contents; (e) immediately fix parasites in near-boiling saline; (f) discard saline and keep specimens in 95% ethanol. Additional information is given for collecting parasites from fish gills with a similar process. The method will collect most helminths (digeneans, larval cestodes, nematodes, acanthocephalans) from the digestive tract, and monogeneans and isopod and copepod crustaceans from fish gills. The specimens will be suitable for both morphological study and DNA sequencing. The method is simple, fast, inexpensive and can be used by untrained personnel, even in the field without electricity and without a binocular microscope. It can also be used by trained parasitologists who need to expedite treatment of abundant samples.


Similar content being viewed by others
References
Albuquerque MC, Ederli NB, Monteiro CM, Rodrigues MLA (2009) Técnica de montagem permanente e morfometria de Ancylostoma caninum (Ercolani, 1859), Oxyspirura mansoni (Cobbold, 1879) Ransom, 1904 e Oesophagostomum columbianum Curtice, 1890. Rev Bras Med Vet 31:80–84
Bakke TA (1988) Morphology of adult Phyllodistomum umblae (Fabricius) (Platyhelminthes, Gorgoderidae): the effect of preparation, killing and fixation procedures. Zool Scr 17:1–13
Baylis HA (1915) Instructions for collectors: no. 12.—worms. British Museum (Natural History), London
Berland B (1961a) Nematodes from some Norwegian marine fishes. Sarsia 2:1–50
Berland B (1961b) Use of glacial acetic acid for killing parasitic Nematodes for collection purposes. Nature 191(4795):1320–1321
Berland B (1984) Basic techniques involved in helminth preservation. Syst Parasitol 6(4):242–245. doi:10.1007/bf00012195
Bouchet P (2006) The magnitude of marine biodiversity. In: Duarte CM (ed) The exploration of marine biodiversity. Scientific and technological challenges. Fundacion BBVA, Bilbao, pp 31–64
Braun M, Lühe M (1910) A handbook of practical parasitology. John Bale, Sons and Danielsson, London
Bray RA (1984) The curation of helminths at the British Museum (Natural History). Syst Parasitol 6:251–253
Bray R, Waeschenbach A, Cribb T, Weedall G, Dyal P, Littlewood D (2009) The phylogeny of the Lepocreadioidea (Platyhelminthes, Digenea) inferred from nuclear and mitochondrial genes: implications for their systematics and evolution. Acta Parasitol 54(4):310–329. doi:10.2478/s11686-009-0045-z
Brooks DR, Hoberg EP (2000) Triage for the biosphere: the need and rationale for taxonomic inventories and phylogenetic studies of parasites. Comp Parasitol 67:1–5
Brooks DR, Hoberg EP (2001) Parasite systematics in the 21st century: opportunities and obstacles. Trends Parasitol 17(6):273–275
Bruno DW, Nowak B, Elliott DG (2006) Guide to the identification of fish protozoan and metazoan parasites in stained tissue sections. Dis Aquat Org 70:1–36
Bunkley-Williams L, Williams EH (1994) Parasites of Puerto Rican freshwater sport fishes. Puerto Rico Department of Natural and Environmental Resources, San Juan, and Department of Marine Sciences, University of Puerto Rico, Mayaguez
Burt MDB (1984) Problems with tense tapeworms. Syst Parasitol 6:249
Campbell RA, Beveridge I (1994) Order Trypanorhyncha Diesing, 1863. In: Khalil LF, Jones A, Bray RA (eds) Keys to the cestode parasites of vertebrates. CAB International, Wellingford, pp 51–148
Černotíková E, Horák A, Moravec F (2011) Phylogenetic relationships of some spirurine nematodes (Nematoda: Chromadorea: Rhabditida: Spirurina) parasitic in fishes inferred from SSU rRNA gene sequences. Folia Parasitol 58:135–148
Chakraborty A, Sakai M, Iwatsuki Y (2006) Museum fish specimens and molecular taxonomy: a comparative study on DNA extraction protocols and preservation techniques. J Appl Ichthyol 22(2):160–166. doi:10.1111/j.1439-0426.2006.00718.x
Chubb JC (1962) Acetic acid as a diluent and dehydrant in the preparation of whole, stained helminths. Biotech Histochem 37:179–182
Cooper DW (1988) The preparation of serial sections of platyhelminth parasites, with details of the materials and facilities required. Syst Parasitol 12:211–229
Cribb TH (2004) Living on the edge: parasite taxonomy in Australia. Int J Parasitol 34:117–123
Cribb TH, Bray RA (1998) Trematodes of fishes: a test case for predictions of parasite biodiversity on the Great Barrier Reef. In: Greenwood JG, Hall NJ (eds) Australian Coral Reef Society 75th Anniversary Conference. School of Marine Science, The University of Queensland, Brisbane, Heron Island October 1997, pp 43–56
Cribb TH, Bray RA (2010) Gut wash, body soak, blender, and heat-fixation: approaches to the effective collection, fixation and preservation of trematodes of fishes. Syst Parasitol 76:1–7
Cribb B, Armstrong W, Whittington I (2004) Simultaneous fixation using glutaraldehyde and osmium tetroxide or potassium ferricyanide-reduced osmium for the preservation of monogenean flatworms: an assessment for Merizocotyle icopae. Microsc Res Tech 63(2):102–110. doi:10.1002/jemt.20015
Criscione CD, Font WF (2001) Artifactual and natural variation of Oochoristica javaensis: statistical evaluation of in situ fixation. Comp Parasitol 68(2):156–163
Deveney M, Whittington ID (2001) A technique for preserving pigmentation in some capsalid monogeneans for taxonomic purposes. Syst Parasitol 48(1):31–35. doi:10.1023/a:1026558405617
Dobson A, Lafferty KD, Kuris AM, Hechinger RF, Jetz W (2008) Homage to Linnaeus: how many parasites? How many hosts? Proc Natl Acad Sci U S A 105:11482–11489. doi:10.1073/pnas.0803232105
Dujardin F (1845) Histoire naturelle des helminthes ou vers intestinaux. Librairie encyclopédique de Roret, Paris
Durette-Desset M-C (1984) Techniques de récolte, de fixation et de conservation des Nématodes parasites de Vertébrés. Syst Parasitol 6:248
Fagerholm HP (1979) Nematode length and preservatives, with a method for determining the length of live specimens. J Parasitol 65(2):334–335
Fagerholm H-P, Lövdahl M (1984) Induced morphometric variation in the preparation of nematode parasites for the LM and SEM. Syst Parasitol 6(4):245–247. doi:10.1007/bf00012196
Frampton M, Droege S, Conrad T, Prager S, Richards MH (2008) Evaluation of specimen preservatives for DNA analyses of bees. J Hymenopt Res 17:195–200
Fukatsu T (1999) Acetone preservation: a practical technique for molecular analysis. Mol Ecol 8(11):1935–1945. doi:10.1046/j.1365-294x.1999.00795.x
Galli P, Strona G, Villa AM, Benzoni F, Stefani F, Doglia SM, Kritsky DC (2006) Three-dimensional imaging of monogenoidean sclerites by laser scanning confocal fluorescence microscopy. J Parasitol 92:395–399
Galli P, Strona G, Villa AM, Benzoni F, Stefani F, Doglia SM, Kritsky DC (2007) Two-dimensional versus three-dimensional morphometry of monogenoidean sclerites. Int J Parasitol 37(3–4):449–456
García-Vásquez A, Shinn A, Bron J (2011) Development of a light microscopy stain for the sclerites of Gyrodactylus von Nordmann, 1832 (Monogenea) and related genera. Parasitol Res. doi:10.1007/s00436-011-2675-y
Garvin MC, Bates JM, Kinsella JM (1997) Field techniques for collecting and preserving helminth parasites from birds, with new geographic and host records of parasitic nematodes from Bolivia. Ornithol Monogr 48:261–266
Gibson DI (1979) Materials and methods in helminth alpha-taxonomy. Parasitology 79:r36
Gibson DI (1984) Technology as applied to museum collections: the collection, fixation and conservation of helminths. Syst Parasitol 6:241
Gläser H-J (1965) Zur Kenntnis der Gattung Dactylogyrus Diesing 1850 (Monogenoidea). Z ParasitenKde 25(5):459–484
Grutter AS (1995) Comparison of methods for sampling ectoparasites from coral reef fishes. Mar Freshw Res 46:897–903
Gusev AV (1983) Methods for collection and preparation of monogeneans parasitizing fish. Nauka, Leningrad
Hajibabaei M, Smith MA, Janzen DH, Rodriguez JJ, Whitfield JB, Hebert PDN (2006) A minimalist barcode can identify a specimen whose DNA is degraded. Mol Ecol Notes 6(4):959–964. doi:10.1111/j.1471-8286.2006.01470.x
Hanson Pritchard M, Kruse G (1984) Making the best of things: reclaiming specimens. Syst Parasitol 6(4):253–255. doi:10.1007/bf00012201
Hargis WJ Jr (1953) Chloretone as a Trematode relaxer, and its use in mass-collecting techniques. J Parasitol 39(2):224–225
Harris PD, Cable J, Tinsley RC, Lazarus CM (1999) Combined ribosomal DNA and morphological analysis of individual gyrodactylid monogeneans. J Parasitol 85(2):188–191
Hendrix SS (1994) Marine flora and fauna of the eastern United States. Platyhelminthes: Monogenea. NOAA Technical Report NMFS 121. US Department of Commerce, Seattle
Herniou EA, Pearce AC, Littlewood DTJ (1998) Vintage helminths yield valuable molecules. Parasitol Today 14(7):289–292
Hoberg EP, Pilitt PA, Galbreath KE (2009) Why museums matter: a tale of pinworms (Oxyuroidea: Heteroxynematidae) among pikas (Ochotona princeps and O. collaris) in the American West. J Parasitol 95:490–501
Huber JT (1998) The importance of voucher specimens, with practical guidelines for preserving specimens of the major invertebrate phyla for identification. J Nat Hist 32(3):367–385
Jeon H-K, Kim K-H, Eom KS (2011) Molecular identification of Taenia specimens after long-term preservation in formalin. Parasitol Int 60(2):203–205
Justine J-L (2005) Species of Pseudorhabdosynochus Yamaguti, 1958 (Monogenea: Diplectanidae) from Epinephelus fasciatus and E. merra (Perciformes: Serranidae) off New Caledonia and other parts of the Indo-Pacific Ocean, with a comparison of measurements of specimens prepared using different methods, and a description of P. caledonicus n. sp. Syst Parasitol 62(1):1–37. doi:10.1007/s11230-005-5480-0
Justine J-L (2007) Huffmanela spp. (Nematoda, Trichosomoididae) parasites in coral reef fishes off New Caledonia, with descriptions of H. balista n. sp. and H. longa n. sp. Zootaxa 1628:23–41
Justine J-L (2010) Parasites of coral reef fish: how much do we know? With a bibliography of fish parasites in New Caledonia. Belg J Zool 140(Suppl):155–190
Justine J-L, Grugeaud A (2010) Does the number of sclerotised structures used for the systematics of monogeneans change with age? A study of the monocotylid Dendromonocotyle pipinna. Parasitol Res 107:1509–1514
Justine J-L, Beveridge I, Boxshall GA, Bray RA, Moravec F, Trilles J-P, Whittington ID (2010a) An annotated list of parasites (Isopoda, Copepoda, Monogenea, Digenea, Cestoda and Nematoda) collected in groupers (Serranidae, Epinephelinae) in New Caledonia emphasizes parasite biodiversity in coral reef fish. Folia Parasitol 57:237–262
Justine J-L, Beveridge I, Boxshall GA, Bray RA, Moravec F, Whittington ID (2010b) An annotated list of fish parasites (Copepoda, Monogenea, Digenea, Cestoda and Nematoda) collected from Emperors and Emperor Bream (Lethrinidae) in New Caledonia further highlights parasite biodiversity estimates on coral reef fish. Zootaxa 2691:1–40
Kennedy MJ (1979) Basic methods of specimen preparation in parasitology. International Development Research Centre, Ottawa
King JR, Porter SD (2004) Recommendations on the use of alcohols for preservation of ant specimens (Hymenoptera, Formicidae). Insect Soc 51(2):197–202. doi:10.1007/s00040-003-0709-x
Klopfleisch R, Weiss ATA, Gruber AD (2011) Excavation of a buried treasure—DNA, mRNA, miRNA and protein analysis in formalin fixed, paraffin embedded tissues. Histol Histopathol 26:797–810
Košková E, Matějusová I, Civáňová K, Koubková B (2011) Ethanol-fixed material used for both classical and molecular identification purposes: Eudiplozoon nipponicum (Monogenea: Diplozoidae) as a case parasite species. Parasitol Res 107(4):909–914. doi:10.1007/s00436-010-1949-0
Kritsky D, Bakenhaster M (2011) Monogenoidean parasites of the gill lamellae of the sheepshead Archosargus probatocephalus (Walbaum) (Perciformes: Sparidae) from the Indian River Lagoon, Florida, with descriptions of four new species of Euryhaliotrema Kritsky & Boeger, 2002 (Dactylogyridae). Syst Parasitol 78(1):57–68. doi:10.1007/s11230-010-9282-7
Kritsky DC, Fennessy CJ (1999) Calicobenedenia polyprioni n. gen., n. sp. (Monogenoidea: Capsalidae) from the external surfaces of wreckfish, Polyprion americanus (Teleostei: Polyprionidae), in the North Atlantic. J Parasitol 85:192–195
Kritsky DC, Leiby PD, Kayton RJ (1978) A rapid stain technique for the haptoral bars of Gyrodactylus species (Monogenea). J Parasitol 64:172–174
Kuris AM, Hechinger RF, Shaw JC, Whitney KL, Aguirre-Macedo L, Boch CA, Dobson AP, Dunham EJ, Fredensborg BL, Huspeni TC, Lorda J, Mababa L, Mancini FT, Mora AB, Pickering M, Talhouk NL, Torchin ME, Lafferty KD (2008) Ecosystem energetic implications of parasite and free-living biomass in three estuaries. Nature 454(7203):515–518. doi:10.1038/nature06970
Lachenmeier DW, Rehm J, Gmel G (2007) Surrogate alcohol: what do we know and where do we go? Alcohol Clin Exp Res 31(10):1613–1624. doi:10.1111/j.1530-0277.2007.00474.x
Lafferty KD, Dobson AP, Kuris AM (2006) Parasites dominate food web links. Proc Natl Acad Sci U S A 103(30):11211–11216. doi:10.1073/pnas.0604755103
Lichtenfels J (1984) Methods for conserving, storing, and studying helminths in the U.S. National Parasite Collection. Syst Parasitol 6(4):250–251. doi:10.1007/bf00012199
Lim LHS (1991) Preparation of Museum specimens—Monogenea. Fish Health Sect Newsl 2:10–11
Maillard C, Gonzalez J, Noisy D (1982) A scanning electron microscope study of the male copulatory sclerite of the monogenean Diplectanum aequans. Parasitology 84:63–64
Malmberg G (1957) Om förekomsten av Gyrodactylus på svenska fiskar (In Swedish.). Skrifter Utgivna av Södra Sveriges Fiskeriförening. Årsskrift 1956:19–76
Manter HW (1926) Some North American fish Trematodes. Illinois Biological Monographs, 10. University of Illinois, Urbana
McCue JF, Thorson RE (1963) A rapid method for collecting large numbers of intestinal helminths. J Parasitol 49(6):997
McDougal HD, Mizelle JD (1969) Studies on Monogenetic Trematodes. XLIV. A new method for collection of Monogenea. Trans Am Microsc Soc 88(3):445–447
Miller TL, Bray RA, Goiran C, Justine J-L, Cribb TH (2009) Adlardia novaecaledoniae n.g., n. sp. (Digenea: Cryptogonimidae) from the fork-tailed threadfin bream Nemipterus furcosus (Val.) (Perciformes: Nemipteridae) off New Caledonia. Syst Parasitol 73:151–160
Miller TL, Adlard RD, Bray RA, Justine J-L, Cribb TH (2010a) Cryptic species of Euryakaina n. g. (Digenea: Cryptogonimidae) from sympatric lutjanids in the Indo-West Pacific. Syst Parasitol 77:185–204
Miller TL, Bray RA, Justine J-L, Cribb TH (2010b) Varialvus gen. nov. (Digenea, Cryptogonimidae), from species of Lutjanidae (Perciformes) off the Great Barrier Reef, New Caledonia and the Maldives. Acta Parasitol 55(4):327–339. doi:10.2478/s11686-010-0045-z
Milne SJ, Avenant-Oldewage A (2006) The fluorescent detection of Paradiplozoon sp. (Monogenea: Diplozoidae) attachment clamps' sclerites and integumental proteins: research communication. Onderstepoort J Vet Res 73:149–152
Mizelle JD (1936) New species of Trematodes from the gills of Illinois fishes. Am Midl Nat 17(5):785–806
Mizelle JD (1938) Comparative studies on Trematodes (Gyrodactyloidea) from the gills of North-American fresh-water fishes. Illinois Biological Monographs, 17(1). The University of Illinois Press, Urbana
Mo TA, Appleby C (1990) A special technique for studying haptoral sclerites of monogeneans. Syst Parasitol 17:103–108
Monteiro C, Kritsky DC, Brasil-Sato M (2010) Neotropical Monogenoidea. 55. Dactylogyrids parasitising the pintado-amarelo Pimelodus maculatus Lacépède (Actinopterygii: Pimelodidae) from the Rio São Francisco, Brazil. Syst Parasitol 76(3):179–190. doi:10.1007/s11230-010-9250-2
Moravec F (1994) Parasitic nematodes of freshwater fishes of Europe. Academia, Praha
Moravec F (2001) Trichinelloid nematodes parasitic in cold-blooded vertebrates. Academia, Praha
Moravec F, Justine J-L (2005) Two species of Philometra (Nematoda, Philometridae) from serranid fishes off New Caledonia. Acta Parasitol 50(4):323–331
Moravec F, Justine J-L (2010a) Some trichinelloid nematodes from marine fishes off New Caledonia, including description of Pseudocapillaria novaecaledoniensis sp. nov. (Capillariidae). Acta Parasitol 55:71–80
Moravec F, Justine J-L (2010b) Two new genera and species of cystidicolids (Nematoda, Cystidicolidae) from marine fishes off New Caledonia. Parasitol Int 59:198–205. doi:10.1016/j.parint.2010.01.005
Nadler S (1999) Nucleotide sequences from vintage helminths: fine wine or vinegar? Parasitol Today 15(3):122
Naem S, Pagan C, Nadler SA (2010) Structural restoration of nematodes and acanthocephalans fixed in high percentage alcohol using DESS solution and rehydration. J Parasitol 96:809–811
Nagy Z (2010) A hands-on overview of tissue preservation methods for molecular genetic analyses. Org Divers Evol 10(1):91–105. doi:10.1007/s13127-010-0012-4
Newman LJ, Cannon LRG (1995) The importance of the fixation of color, pattern and form in tropical Pseudocerotidae (Platyhelminthes, Polycladida). Hydrobiologia 305(1–3):141–143
Nichols E, Gomez A (2011) Conservation education needs more parasites. Biol Conserv 144(2):937–941. doi:10.1016/j.biocon.2010.10.025
Olson PD, Caira JN, Jensen K, Overstreet RM, Palm HW, Beveridge I (2010) Evolution of the trypanorhynch tapeworms: parasite phylogeny supports independent lineages of sharks and rays. Int J Parasitol 40:223–242
Pariselle A, Lambert A, Euzet L (1991) A new type of haptor in mesoparasitic monogeneans of the genus Enterogyrus Paperna, 1963, with a description of Enterogyrus foratus n. sp. and E. coronatus n. sp, stomach parasites of cichlids in West Africa. Syst Parasitol 20(3):211–220. doi:10.1007/bf00009785
Parker JH, Curran SS, Overstreet RM, Tkach VV (2010) Examination of Homalometron elongatum Manter, 1947 and description of a new congener from Eucinostomus currani Zahuranec, 1980 in the Pacific Ocean off Costa Rica. Comp Parasitol 77(2):154–163
Perkins EM, Donnellan SC, Bertozzi T, Chisholm LA, Whittington ID (2009) Looks can deceive: molecular phylogeny of a family of flatworm ectoparasites (Monogenea: Capsalidae) does not reflect current morphological classification. Mol Phylogenet Evol 52:705–714. doi:10.1016/j.ympev.2009.05.008
Post RJ, Flook PK, Millest AL (1993) Methods for the preservation of insects for DNA studies. Biochem Syst Ecol 21(1):85–92
Poulin R (2004) Parasite species richness in New Zealand fishes: a grossly underestimated component of biodiversity? Divers Distrib 10:31–37
Poulin R, Leung TLF (2010) Taxonomic resolution in parasite community studies: are things getting worse? Parasitology 137:1967–1973. doi:10.1017/S0031182010000910
Poulin R, Morand S (2000) The diversity of parasites. Q Rev Biol 75(3):277–293
Poulin R, Morand S (2004) Parasite biodiversity. Smithsonian Books, Washington
Pritchard MH, Kruse GOW (1982) The collection and preservation of animal parasites. University of Nebraska Press, Lincoln
Quicke DLJ, Lopez-Vaamonde C, Belshaw R (1999) Preservation of hymenopteran specimens for subsequent molecular and morphological study. Zool Scr 28(1–2):261–267. doi:10.1046/j.1463-6409.1999.00004.x
Richards GR, Chubb JC (1995) Trichrome staining of Gyrodactylus sclerites and soft tissues following fixation in ammonium picrate-glycerin, including an improved rendition of the haptoral bars of G. tumbulli. J Helminthol 69:149–154
Rogers WA (1966) Three new species of Pseudomurraytrema (Trematoda: Monogenea) from gills of catostomid fishes. J Parasitol 52(3):462–465
Rohde K (1987) Different populations of Scomber australasicus in New Zealand and south-eastern Australia, demonstrated by a simple method using monogenean sclerites. J Fish Biol 30(6):651–657. doi:10.1111/j.1095-8649.1987.tb05794.x
Rohde K (2001) Marine parasite diversity and environmental gradients. In: Levin S (ed) Encyclopedia of Biodiversity, vol 1. Academic, New York, pp 73–88
Rohde K, Watson N (1985) Morphology, microhabitats and geographical variation of Kuhnia spp. (Monogenea: Polyopisthocotylea). Int J Parasitol 15(5):569–586
Schmidt GD (1986) CRC handbook of tapeworm identification. CRC Press, Boca Raton, Florida
Shinn AP, Gibson DI, Sommerville C (1993) An SEM study of the haptoral sclerites of the genus Gyrodactylus Nordmann, 1832 (Monogenea) following extraction by digestion and sonication techniques. Syst Parasitol 25(2):135–144. doi:10.1007/bf00009983
Shinn AP, Collins C, Garcia-Vasquez A, Snow M, Matejusova I, Paladini G, Longshaw M, Lindenstrøm T, Stone DM, Turnbull JF, Picon-Camacho SM, Rivera CV, Duguid RA, Mo TA, Hansen H, Olstad K, Cable J, Harris PD, Kerr R, Graham D, Monaghan SJ, Yoon GH, Buchmann K, Taylor NGH, Bakke TA, Raynard R, Irving S, Bron JE (2010) Multi-centre testing and validation of current protocols for the identification of Gyrodactylus salaris (Monogenea). Int J Parasitol 40(12):1455–1467
Simsek S, Kaplan M, Ozercan I (2011) A comprehensive molecular survey of Echinococcus granulosus in formalin-fixed paraffin-embedded tissues in human isolates in Turkey. Parasitol Int 109(2):411–416. doi:10.1007/s00436-011-2269-8
Snyder SD, Clopton RE (2005) New methods for the collection and preservation of spirorchiid trematodes and polystomatid monogeneans from turtles. Comp Parasitol 72:102–107
Srinivasan M, Sedmak D, Jewell S (2002) Effect of fixatives and tissue processing on the content and integrity of nucleic acids. Am J Pathol 161(6):1961–1971
Strona G, Stefani F, Galli P (2009) Field preservation of monogenean parasites for molecular and morphological analyses. Parasitol Int 58(1):51–54
Thatcher VE (2006) Amazon fish parasites. ABLA Series, Vol. 1. Aquatic Biodiversity of Latin America (ABLA Series). Pensoft, Sofia, Bulgaria
Toe L, Back C, Adjami AG, Tang JM, Unnasch TR (1997) Onchocerca volvulus: comparison of field collection methods for the preservation of parasite and vector samples for PCR analysis. Bull World Health Organ 75(5):443–447
Unnithan RV (1957) On the functional morphology of a new fauna of Monogenea on fishes drom Trivandrum and environs. Part I, Axinidae Fam. nov. Bull Cent Res Inst Univ Kerala Ser C 5:27–122
Van Beneden P-J (1878) Les commensaux et les parasites dans le règne animal. Librairie Germer Baillière et Cie, Paris
Van Beneden P-J (1887) Animal parasites and messmates. Appleton, New York
Vannier-Santos MA, Lenzi HL (2011) Parasites or cohabitants: cruel omnipresent usurpers or creative “éminences grises”? J Parasitol Res 2011:19. doi:10.1155/2011/214174, Article ID 214174
Vickerman K (2009) Not a very nice subject. Changing views of parasites and parasitology in the twentieth century. Parasitology 136(12):1395–1402. doi:10.1017/s0031182009990825
Whittington ID, Chisholm LA (2003) Biodiversity of marine parasites in Australia: more than just a list of largely invisible creatures. Rec S Aust Mus Monogr Ser 7:51–60
Williams EHJ, Bunkley-Williams L, Dowgiallo MJ, Dyer WG (1991) Influence of collection methods on the occurrence of alimentary canal helminth parasites in fish. J Parasitol 77:1019–1022
Windsor DA (1995) Equal rights for parasites. Conserv Biol 9(1):1–2. doi:10.1046/j.1523-1739.1995.09010001.x
Windsor DA (1998) Most of species on Earth are parasites. Int J Parasitol 28:1939–1941
Wong WL, Tan WB, Lim LHS (2006) Sodium dodecyl sulphate as a rapid clearing agent for studying the hard parts of monogeneans and nematodes. J Helminthol 80:87–90. doi:10.1079/JOH2005320
Yamaguti S (1965a) New digenetic trematodes from Hawaiian fishes, I. Pac Sci 19:458–481
Yamaguti S (1965b) Preparation of stained whole mounts of flatworms. Trans Am Microsc Soc 84:602–603
Yamaguti S (1968) Monogenetic trematodes of Hawaiian Fishes. University of Hawaii Press, Honolulu
Yoder M, De Ley IT, King IW, Mundo-Ocampo M, Mann J, Blaxter M, Poiras L, De Ley P (2006) DESS: a versatile solution for preserving morphology and extractable DNA of nematodes. Nematology 8:367–376
Acknowledgements
David Gibson (NHM, London) provided literature; Sarah Samadi and Regis Debruyne (MNHN, Paris) discussed DNA conservation. Students (Cyndie Dupoux, Adeline Grugeaud, Isabelle Mary, Sophie Olivier, Charlotte Schoelinck, Aude Sigura) helped in testing various methods; Eva Řehulkova (Masaryk University, Czech Republic) helped in the field.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Justine, JL., Briand, M.J. & Bray, R.A. A quick and simple method, usable in the field, for collecting parasites in suitable condition for both morphological and molecular studies. Parasitol Res 111, 341–351 (2012). https://doi.org/10.1007/s00436-012-2845-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-012-2845-6