Abstract
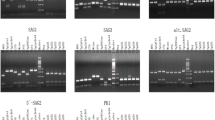
Toxoplasma gondii is one of the most prevalent protozoan parasites in Iran. This study was aimed to isolate T. gondii from a variety of hosts and to genetically analyze the parasite isolates. The prevalence of T. gondii in different animal hosts was assessed in two provinces of Iran, Tehran and Mazandaran in the central and northern parts, respectively. The latex agglutination (LA) test was carried out, and antibodies were found in 24 out of 105 sheep, 5 out of 35 goats, 23 out of 45 free-ranging chickens (Gallus domesticus), 2 out of 13 ducks (Anas spp.), and two of four stray cats (Felis domesticus). T. gondii was isolated by bioassay in mice from four sheep, six chickens, one duck, two cats, and three human samples. Genotyping of these 16 isolates was performed using Multiplex PCR for five microsatellite markers and GRA6 gene sequence analysis. The results indicated that the studied isolates consisted of only two genotypes, II and III, with no evidence of type 1 or mixed genotypes.
Similar content being viewed by others
References
Ajzenberg D, Banuls AL, Su C, Dumètre A, Demar M, Carme B, Dardé ML (2004) Genetic diversity, clonality and sexuality in Toxoplasma gondii. Int J Parasitol 34:1185–1196
Ajzenberg D, Dumètre A, Dardé ML (2005) Multiplex PCR for typing strains of Toxoplasma gondii. J Clin Microbiol 43:1940–1943
Assmar M, Amirkhani A, Piazak N, Hovanesian A, Kooloobandi A, Etessami R (1997) Toxoplasmosis in Iran, results of a seroepidemiological investigation. Bull Soc Pathol Exot 90:19–21
Bell AS, Ranford-cartwright LC (2002) Real-time quantitative PCR in parasitology. Trends Parasitol 114:89–95
Dardé ML (2004) Genetic analysis of the diversity in Toxoplasma gondii. Ann Ist Super Sanità 40:57–63
Dardé ML, Bouteille B, Perstre M (1992) Isoenzyme analysis of 35 Toxoplasma gondii isolates and the biological and epidemiologic implications. J Parasitol 78:909–912
Dubey JP (1998) Refinement of pepsin digestion method for isolation of Toxoplasma gondii from infected tissues. Vet Parasitol 74:75–77
Dubey JP, Beattie CP (1988) Toxoplasmosis of Animals and Man. CRC, Boca Raton, FL, pp 220
Dubey JP, Graham DH, Blackston CR, Lehmann T, Gennari SM, Ragozo AMA, Nishi SM, Shen SK, Kwok OC, Hill DE, Thulliez P (2002) Biological and genetic characterisation of Toxoplasma gondii isolated from chicken (Gallus domesticus) from Sao Paulo Brazil. Int J Parasitol 32:99–105
Dubey JP, Graham DH, Dahl E, Hilali M, El-Ghaysh A, Sreekumar C, Kwok OC, Shen SK, Lehmann T (2003) Isolation and molecular characterization of Toxoplasma gondii from chickens and ducks from Egypt. Vet Parasitol 114:89–95
Dubey JP, Morales ES, Lehmann T (2004) Isolation and genotyping of Toxoplasma gondii from free-ranging chickens from Mexico. J Parasitol 90:411–413
Fazaeli A, Carter PE, Dardé ML, Pennington TH (2000) Molecular typing of Toxoplasma gondii strains by GRA6 gene sequence analysis. Int J Parasitol 30:637–642
Ghorbani M, Samii AH (1973) Toxoplasmic lymphadenitis in Iran. J Trop Med Hyg 76:158–161
Ghorbani M, Hafizi A, Shegerfcar MT, Rezaian M, Nadim A, Anwar M, Afshar A (1983) Animal toxoplasmosis in Iran. J Trop Med Hyg 86:73–76
Hashemi-Fesharki R (1996) Seroprevalence of Toxoplasma gondii in cattle, sheep and goats in Iran. Vet Parasitol 61:1–3
Homan WL, Vercammen M, De Braekeleer J, Verschueren H (2000) Identification of a 200- to 300-fold repetitive 529 bp DNA fragment in Toxoplasma gondii, and its use for diagnostic and quantitative PCR. Int J Parasitol 30:69–75
Howe DK, Sibley LD (1995) Toxoplasma gondii comprises three clonal lineages: correlation of parasite genotype with human disease. J Infect Dis 172:1561–1566
Jacobs L, Remington JSL, Melton ML (1960) A survey of meat samples from swine, cattle and sheep for the presence of encysted Toxoplasma. J Parasitol 46:23–28
Keshavarz-Valian H, Ebrahimi A (1997) The prevalence of Toxoplasma gondii in birds by serology and parasitology methods in Kerman, Iran. Iranian J Publ Health 23:25–33
Mazumder P, Chuang HY, Wentz MW, Wiedbrauk DL (1988) Latex agglutination test for detection of antibodies to Toxoplasma gondii. J Clin Microbiol 26:2444–2446
Mondragon R, Howe DK, Dubey JP, Sibley LD (1998) Genotyping analysis of Toxoplasma gondii isolates from pigs. J Parasitol 84:639–641
Owen MR, Trees AJ (1999) Genotyping of Toxoplasma gondii associated with abortion in sheep. J Parasitol 85:382–384
Ruiz A, Frenkel JK (1980) Intermediate and transport hosts of Toxoplasma gondii in Costa Rica. Am J Trop Med Hyg 29:1161–1166
Samad MA, Rahman KB, Halder AK (1993) Seroprevalence ofT. gondii in domestic ruminants in Bangladesh. Vet Parasitol 47:157–159
Sedaghat A, Ardehali SM, Sedigh M, Buxton M (1978) The prevalence of Toxoplasma infection in southern Iran. J Trop Med Hyg 81:204–207
Sibley LD, Howe DK (1996) Genetic basis of pathogenicity in toxoplasmosis. Curr Top Microbiol Immunol 219:3–15
Tabaii J (1999) Seroprevalence of Toxoplasma gondii in stray cats of Tehran. MS thesis, Tarbiat Modarres University, Tehran, Iran
Acknowledgment
We are indebted to N. Mohajeri, Z. Garoosi, and E. Gholami for their technical assistance. Our thanks are due to the head and staff of the Babol Health Research Station affiliated to Tehran University of Medical Sciences for their kind cooperation. We are grateful to Dr. J.P. Dubey (USDA) for the critical review and helpful discussion.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zia-Ali, N., Fazaeli, A., Khoramizadeh, M. et al. Isolation and molecular characterization of Toxoplasma gondii strains from different hosts in Iran. Parasitol Res 101, 111–115 (2007). https://doi.org/10.1007/s00436-007-0461-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-007-0461-7