Abstract
Purpose
To better understand the clinicopathological characteristics and molecular alterations in different intratumoral components of colorectal cancer (CRC) with heterogeneity of mismatch repair (MMR) protein expression and microsatellite instability (MSI) status.
Methods
The histopathological features, MSI status, and other molecular alterations were analyzed in separately microdissected intratumoral regions and matched metastatic lymph nodes in four cases with intratumoral heterogenous MMR expression screened from 500 CRC patients, using PCR-based MSI testing, MLH1 promoter methylation, and targeted next-generation sequencing (NGS).
Results
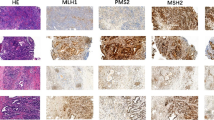
High microsatellite instability (MSI-H) was identified in MLH1/PMS2-deficient regions in Cases 1 to 3 and in MSH2/MSH6-deficient regions in Case 4, while microsatellite stability (MSS) was detected in all the intratumoral regions and metastatic lymph nodes with proficient MMR expression (pMMR). Intratumoral heterogeneity of MLH1 promoter methylation and/or other common driving gene mutations of CRC, such as KRAS and PIK3CA mutations, was identified in all four CRCs. Further, three cases (75%) showed heterogeneous histomorphological features in intratumoral components and metastatic lymph nodes (Cases 1, 2, and 4), and the corresponding metastatic lymph nodes showed moderate differentiation with MSS/pMMR (Cases 2 and 3).
Conclusions
Intratumoral heterogeneous MSI status is highly correlated with intratumoral histomorphological heterogeneity, which is also an important clue for the intratumoral heterogeneity of drive gene mutations in CRC. Thus, it is essential to detect MMR protein expression and other gene mutations in metastases before treatment, especially for CRCs with intratumoral heterogenous MMR protein expression or heterogenous histomorphological features.



Similar content being viewed by others
References
Ágoston EI, Baranyai Z, Dede K, Bodoky G, Kulka J, Bursics A et al (2015) Occurrence, intratumoral heterogeneity, prognostic and predictive potential of microsatellite instability following surgical resection of primary colorectal carcinomas and corresponding liver metastases. Orv Hetil 156(36):1460–1471. https://doi.org/10.1556/650.2015.30218
André T, de Gramont A, Vernerey D, Chibaudel B, Bonnetain F, Tijeras-Raballand A et al (2015) Adjuvant fluorouracil, leucovorin, and oxaliplatin in stage II to III colon cancer: updated 10-year survival and outcomes according to BRAF mutation and mismatch repair status of the MOSAIC study. J Clin Oncol 33(35):4176–4187. https://doi.org/10.1200/JCO.2015.63.4238
Bai Q, Zhang X, Zhu X, Wang L, Huang D, Cai X et al (2016) Pancreatic carcinosarcoma with the same KRAS gene mutation in both carcinomatous and sarcomatous components: molecular evidence for monoclonal origin of the tumour. Histopathology 69(3):393–405. https://doi.org/10.1111/his.12975
Bai W, Ma J, Liu Y, Liang J, Wu Y, Yang X et al (2019) Screening of MSI detection loci and their heterogeneity in East Asian colorectal cancer patients. Cancer Med 8(5):2157–2166. https://doi.org/10.1002/cam4.2111
Benson AB, Venook AP, Al-Hawary MM, Cederquist L, Chen YJ, Ciombor KK et al (2018) NCCN guidelines insights: colon cancer Version 2.2018. J Natl Compr Canc Netw 16(4):359–369. https://doi.org/10.6004/jnccn.2018.0021
Boutin AT, Liao WT, Wang M, Hwang SS, Karpinets TV, Cheung H et al (2017) Oncogenic Kras drives invasion and maintains metastases in colorectal cancer. Genes Dev 31(4):370–382. https://doi.org/10.1101/gad.293449.116
Brannon AR, Vakiani E, Sylvester BE, Scott SN, McDermott G, Shah RH et al (2014) Comparative sequencing analysis reveals high genomic concordance between matched primary and metastatic colorectal cancer lesions. Genome Biol 15(8):454. https://doi.org/10.1186/s13059-014-0454-7
Cai Z, Wang Z, Liu C, Shi D, Li D, Zheng M et al (2020) Detection of microsatellite instability from circulating tumor DNA by targeted deep sequencing. J Mol Diagn 22(7):860–870. https://doi.org/10.1016/j.jmoldx.2020.04.210
Chapusot C, Martin L, Bouvier AM, Bonithon-Kopp C, Ecarnot-Laubriet A, Rageot D et al (2002) Microsatellite instability and intratumoural heterogeneity in 100 right-sided sporadic colon carcinomas. Br J Cancer 87(4):400–404. https://doi.org/10.1038/sj.bjc.6600474
Cohen SA, Turner EH, Beightol MB, Jacobson A, Gooley TA, Salipante SJ et al (2016) Frequent PIK3CA mutations in colorectal and endometrial tumors with 2 or more somatic mutations in mismatch repair genes. Gastroenterology 151(3):440-447.e1. https://doi.org/10.1053/j.gastro.2016.06.004
Corti C, Sajjadi E, Fusco N (2019) Determination of mismatch repair status in human cancer and its clinical significance: does one size fit all. Adv Anat Pathol 26(4):270–279. https://doi.org/10.1097/PAP.0000000000000234
Dallol A, Buhmeida A, Al-Ahwal MS, Al-Maghrabi J, Bajouh O, Al-Khayyat S et al (2016) Clinical significance of frequent somatic mutations detected by high-throughput targeted sequencing in archived colorectal cancer samples. J Transl Med 14(1):118. https://doi.org/10.1186/s12967-016-0878-9
Diaz LA Jr, Le DT (2015) PD-1 blockade in tumors with mismatch-repair deficiency. N Engl J Med 373(20):1979. https://doi.org/10.1056/NEJMc1510353
Fadhil W, Field J, Cross G, Kaye P, Ilyas M (2012) Immunostaining in the context of loss mismatch repair function: interpretive confounders and cautionary tales. Histopathology 61(3):522–525. https://doi.org/10.1111/j.1365-2559.2012.04268.x
Germano G, Lamba S, Rospo G, Barault L, Magrì A, Maione F et al (2017) Inactivation of DNA repair triggers neoantigen generation and impairs tumour growth. Nature 552(7683):116–120. https://doi.org/10.1038/nature24673
Graham RP, Kerr SE, Butz ML, Thibodeau SN, Halling KC, Smyrk TC et al (2015) Heterogenous MSH6 loss is a result of microsatellite instability within MSH6 and occurs in sporadic and hereditary colorectal and endometrial carcinomas. Am J Surg Pathol 39(10):1370–1376. https://doi.org/10.1097/PAS.0000000000000459
Guerra J, Pinto C, Pinto D, Pinheiro M, Silva R, Peixoto A et al (2017) POLE somatic mutations in advanced colorectal cancer. Cancer Med 6(12):2966–2971. https://doi.org/10.1002/cam4.1245
Hackler L Jr, Masuda T, Oliver VF, Merbs SL, Zack DJ (2012) Use of laser capture microdissection for analysis of retinal mRNA/miRNA expression and DNA methylation. Methods Mol Biol 884:289–304. https://doi.org/10.1007/978-1-61779-848-1_21
Haddad R, Ogilvie RT, Croitoru M, Muniz V, Gryfe R, Pollet A et al (2004) Microsatellite instability as a prognostic factor in resected colorectal cancer liver metastases. Ann Surg Oncol 11(11):977–982. https://doi.org/10.1245/ASO.2004.03.585
Halvarsson B, Lindblom A, Rambech E, Lagerstedt K, Nilbert M (2004) Microsatellite instability analysis and/or immunostaining for the diagnosis of hereditary nonpolyposis colorectal cancer. Virchows Arch 444(2):135–141. https://doi.org/10.1007/s00428-003-0922-z
Hamzaoui N, Alarcon F, Leulliot N, Guimbaud R, Buecher B, Colas C et al (2020) Genetic, structural, and functional characterization of POLE polymerase proofreading variants allows cancer risk prediction. Genet Med 22(9):1533–1541. https://doi.org/10.1038/s41436-020-0828-z
Haraldsdottir S, Roth R, Pearlman R, Hampel H, Arnold CA, Frankel WL (2016) Mismatch repair deficiency concordance between primary colorectal cancer and corresponding metastasis. Fam Cancer 15(2):253–260. https://doi.org/10.1007/s10689-015-9856-2
He WZ, Hu WM, Wang F, Rong YM, Yang L, Xie QK et al (2019) Comparison of mismatch repair status between primary and matched metastatic sites in patients with colorectal cancer. J Natl Compr Canc Netw 17(10):1174–1183. https://doi.org/10.6004/jnccn.2019.7308
Hemminger JA, Pearlman R, Haraldsdottir S, Knight D, Jonasson JG, Pritchard CC et al (2018) Histology of colorectal adenocarcinoma with double somatic mismatch-repair mutations is indistinguishable from those caused by Lynch syndrome. Hum Pathol 78:125–130. https://doi.org/10.1016/j.humpath.2018.04.017
Hemminki A, Peltomäki P, Mecklin JP, Järvinen H, Salovaara R, Nyström-Lahti M et al (1994) Loss of the wild type MLH1 gene is a feature of hereditary nonpolyposis colorectal cancer. Nat Genet 8(4):405–410. https://doi.org/10.1038/ng1294-405
Hu H, Cai W, Wu D, Hu W, Dong Wang L, Mao J et al (2021) Ultra-mutated colorectal cancer patients with POLE driver mutations exhibit distinct clinical patterns. Cancer Med 10(1):135–142. https://doi.org/10.1002/cam4.3579
Hühns M, Krohn S, Murua Escobar H, Prall F (2018) Genomic heterogeneity in primary colorectal carcinomas and their metastases: born bad or brought up a villain. Hum Pathol 74:54–63. https://doi.org/10.1016/j.humpath.2017.12.015
Jeantet M, Tougeron D, Tachon G, Cortes U, Archambaut C, Fromont G et al (2016) High intra- and inter-tumoral heterogeneity of RAS mutations in colorectal cancer. Int J Mol Sci 17:12. https://doi.org/10.3390/ijms17122015
Joost P, Veurink N, Holck S, Klarskov L, Bojesen A, Harbo M et al (2014) Heterogenous mismatch-repair status in colorectal cancer. Diagn Pathol 9:126. https://doi.org/10.1186/1746-1596-9-126
Jung J, Kang Y, Lee YJ, Kim E, Ahn B, Lee E et al (2017) Comparison of the mismatch repair system between primary and metastatic colorectal cancers using immunohistochemistry. J Pathol Transl Med 51(2):129–136. https://doi.org/10.4132/jptm.2016.12.09
Kawai T, Nyuya A, Mori Y, Tanaka T, Tanioka H, Yasui K et al (2021) Clinical and epigenetic features of colorectal cancer patients with somatic POLE proofreading mutations. Clin Epigenetics 13(1):117. https://doi.org/10.1186/s13148-021-01104-7
Latham A, Srinivasan P, Kemel Y, Shia J, Bandlamudi C, Mandelker D et al (2019) Microsatellite instability Is associated with the presence of lynch syndrome pan-cancer. J Clin Oncol 37(4):286–295. https://doi.org/10.1200/JCO.18.00283
Lemery S, Keegan P, Pazdur R (2017) First FDA approval agnostic of cancer site - when a biomarker defines the indication. N Engl J Med 377(15):1409–1412. https://doi.org/10.1056/NEJMp1709968
León-Castillo A, Britton H, McConechy MK, McAlpine JN, Nout R, Kommoss S et al (2020) Interpretation of somatic POLE mutations in endometrial carcinoma. J Pathol 250(3):323–335. https://doi.org/10.1002/path.5372
Lindor NM, Burgart LJ, Leontovich O, Goldberg RM, Cunningham JM, Sargent DJ et al (2002) Immunohistochemistry versus microsatellite instability testing in phenotyping colorectal tumors. J Clin Oncol 20(4):1043–1048. https://doi.org/10.1200/JCO.2002.20.4.1043
McConechy MK, Talhouk A, Leung S, Chiu D, Yang W, Senz J et al (2016) Endometrial carcinomas with POLE exonuclease domain mutations have a favorable prognosis. Clin Cancer Res 22(12):2865–2873. https://doi.org/10.1158/1078-0432.CCR-15-2233
Pearlman R, Haraldsdottir S, de la Chapelle A, Jonasson JG, Liyanarachchi S, Frankel WL et al (2019) Clinical characteristics of patients with colorectal cancer with double somatic mismatch repair mutations compared with Lynch syndrome. J Med Genet 56(7):462–470. https://doi.org/10.1136/jmedgenet-2018-105698
Renkonen E, Zhang Y, Lohi H, Salovaara R, Abdel-Rahman WM, Nilbert M et al (2003) Altered expression of MLH1, MSH2, and MSH6 in predisposition to hereditary nonpolyposis colorectal cancer. J Clin Oncol 21(19):3629–3637. https://doi.org/10.1200/JCO.2003.03.181
Sinicrope FA, Foster NR, Thibodeau SN, Marsoni S, Monges G, Labianca R et al (2011) DNA mismatch repair status and colon cancer recurrence and survival in clinical trials of 5-fluorouracil-based adjuvant therapy. J Natl Cancer Inst 103(11):863–875. https://doi.org/10.1093/jnci/djr153
Tachon G, Frouin E, Karayan-Tapon L, Auriault ML, Godet J, Moulin V et al (2018) Heterogeneity of mismatch repair defect in colorectal cancer and its implications in clinical practice. Eur J Cancer 95:112–116. https://doi.org/10.1016/j.ejca.2018.01.087
Temko D, Van Gool IC, Rayner E, Glaire M, Makino S, Brown M et al (2018) Somatic POLE exonuclease domain mutations are early events in sporadic endometrial and colorectal carcinogenesis, determining driver mutational landscape, clonal neoantigen burden and immune response. J Pathol 245(3):283–296. https://doi.org/10.1002/path.5081
Tougeron D, Mouillet G, Trouilloud I, Lecomte T, Coriat R, Aparicio T et al (2016) Efficacy of adjuvant chemotherapy in colon cancer with microsatellite instability: a large multicenter AGEO study. J Natl Cancer Inst. https://doi.org/10.1093/jnci/djv438
Yaeger R, Chatila WK, Lipsyc MD, Hechtman JF, Cercek A, Sanchez-Vega F et al (2018) Clinical sequencing defines the genomic landscape of metastatic colorectal cancer. Cancer Cell 33(1):125-136.e3. https://doi.org/10.1016/j.ccell.2017.12.004
Yim KL (2012) Microsatellite instability in metastatic colorectal cancer: a review of pathology, response to chemotherapy and clinical outcome. Med Oncol 29(3):1796–1801. https://doi.org/10.1007/s12032-011-0050-6
Zhu L, Huang Y, Fang X, Liu C, Deng W, Zhong C et al (2018) A novel and reliable method to detect microsatellite instability in colorectal cancer by next-generation sequencing. J Mol Diagn 20(2):225–231. https://doi.org/10.1016/j.jmoldx.2017.11.007
Zwaenepoel K, Holmgaard Duelund J, De Winne K, Maes V, Weyn C, Lambin S et al (2020) Clinical performance of the idylla MSI test for a rapid assessment of the dna microsatellite status in human colorectal cancer. J Mol Diagn 22(3):386–395. https://doi.org/10.1016/j.jmoldx.2019.12.002
Acknowledgements
We acknowledge technical help from Xu Cai and Wenhua Jiang, and experimental assistance by the Clinical Molecular Pathology Laboratory of Fudan University Shanghai Cancer Center.
Funding
This research was financially supported by the National Key Research and Development Program of China (No.YFC20170110100), the Science and Technology Commission of Shanghai Municipality (No.19441904900), the Shanghai Science and Technology Development Foundation (19MC1911000), the Shanghai Municipal Key Clinical Specialty (shslczdzk01301), the Innovation Group Project of Shanghai Municipal Health Commission (No.2019CXJQ03), and the Integrated Diagnostic Pathological Study on Cancer of Unknown Primary (No.20Z11900300).
Author information
Authors and Affiliations
Contributions
JZ and XZ: designed and conducted the study, and wrote the manuscript. QW and YYX: collected and tested samples. QLY and DH: analyzed and interpreted of the experimental data. XLZ and WQS: performed pathology assessment and article revision. XYZ and QMB: designed and managed the study, and had final responsibility for the decision to submit for publication. All authors reviewed the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflict of interest
The authors declare no conflicts of interest.
Ethical approval
The study was approved by Fudan University Shanghai Cancer Center’s institutional ethics committee (approval number 1812195-3).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, J., Zhang, X., Wang, Q. et al. Histomorphological and molecular genetic characterization of different intratumoral regions and matched metastatic lymph nodes of colorectal cancer with heterogenous mismatch repair protein expression. J Cancer Res Clin Oncol 149, 3423–3434 (2023). https://doi.org/10.1007/s00432-022-04261-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-022-04261-1