Abstract
Purpose
The Glasgow Prognostic Score (GPS), an inflammation-based prognostic score systems composed of C-reactive protein and albumin, has been reported to be predictive of survival in several types of malignancies. The prognostic significance of GPS in epithelial ovarian cancer (EOC) remains unclear. We conducted this study to assess the prognostic value of GPS in a cohort of patients with advanced EOC receiving neoadjuvant chemotherapy (NAC) followed by debulking surgery.
Methods
Six hundred and seventy-two patients newly diagnosed with advanced EOC were retrospectively analyzed.
Results
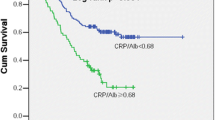
High GPS was significantly related to Eastern Cooperative Group performance status, histological type, histological grade and the size of residual tumor after the debulking surgery. In addition, patients with higher GPS at diagnosis achieved lower complete remission rates after NAC (P < 0.05) and had shorter progression-free survival (PFS; P < 0.001) and overall survival (OS; P < 0.001). Multivariate analysis showed high GPS was independent adverse predictors of PFS and OS.
Conclusions
Our data demonstrated that GPS at diagnosis is a powerful independent prognostic factor for advanced epithelial ovarian cancer. However, further studies are needed to prospectively validate this prognostic model and investigate the mechanisms underlying the correlation between high GPS and poor prognosis in advanced epithelial ovarian cancer.

Similar content being viewed by others
Abbreviations
- GPS:
-
Glasgow Prognostic Score
- EOC:
-
Epithelial ovarian cancer
- NAC:
-
Neoadjuvant chemotherapy
- CRP:
-
C-reactive protein
- PFS:
-
Progression-free survival
- OS:
-
Overall survival
- ECOG PS:
-
Eastern Cooperative Oncology Group performance status
- FIGO:
-
International Federation of Gynecology and Obstetrics
- HR:
-
Hazard ratio
- CI:
-
Confidence intervals
References
Candido J, Hagemann T (2013) Cancer-related inflammation. J Clin Immunol 33(Suppl 1):S79–S84. doi:10.1007/s10875-012-9847-0
Cannistra SA (2004) Cancer of the ovary. N Engl J Med 351:2519–2529. doi:10.1056/NEJMra041842
Casadei GA et al (2016) Prognostic role of serum concentrations of high-sensitivity C-reactive protein in patients with metastatic colorectal cancer: results from the ITACa trial. Oncotarget 7:10193–10202. doi:10.18632/oncotarget.7166
Elinav E, Nowarski R, Thaiss CA, Hu B, Jin C, Flavell RA (2013) Inflammation-induced cancer: crosstalk between tumours, immune cells and microorganisms. Nat Rev Cancer 13:759–771. doi:10.1038/nrc3611
Eren T et al (2016) Clinical significance of the Glasgow Prognostic Score for survival after colorectal cancer surgery. J Gastrointest Surg 20:1231–1238. doi:10.1007/s11605-016-3114-2
Ferlay J, Parkin DM, Steliarova-Foucher E (2010) Estimates of cancer incidence and mortality in Europe in 2008. Eur J Cancer (Oxford England) 46:765–781. doi:10.1016/j.ejca.2009.12.014
Forrest LM, McMillan DC, McArdle CS, Angerson WJ, Dunlop DJ (2003) Evaluation of cumulative prognostic scores based on the systemic inflammatory response in patients with inoperable non-small-cell lung cancer. Br J Cancer 89:1028–1030. doi:10.1038/sj.bjc.6601242
Forrest LM, McMillan DC, McArdle CS, Angerson WJ, Dunlop DJ (2004) Comparison of an inflammation-based prognostic score (GPS) with performance status (ECOG) in patients receiving platinum-based chemotherapy for inoperable non-small-cell lung cancer. Br J Cancer 90:1704–1706. doi:10.1038/sj.bjc.6601789
Grivennikov SI, Greten FR, Karin M (2010) Immunity, inflammation, and cancer. Cell 140:883–899. doi:10.1016/j.cell.2010.01.025
Gupta D, Lis CG (2010) Pretreatment serum albumin as a predictor of cancer survival: a systematic review of the epidemiological literature. Nutr J 9:69. doi:10.1186/1475-2891-9-69
Hefler LA et al (2008) Serum C-reactive protein as independent prognostic variable in patients with ovarian cancer. Clin Cancer Res 14:710–714. doi:10.1158/1078-0432.ccr-07-1044
Hosford-Donovan A, Nilsson A, Wahlin-Larsson B, Kadi F (2016) Observational and mechanistic links between C-reactive protein and blood pressure in elderly women. Maturitas 89:52–57. doi:10.1016/j.maturitas.2016.04.016
Jing X et al (2015) Association between serum C-reactive protein value and prognosis of patients with non-small cell lung cancer: a meta-analysis. Int J Clin Exp Med 8:10633–10639
King TA, Morrow M (2015) Surgical issues in patients with breast cancer receiving neoadjuvant chemotherapy. Nat Rev Clin Oncol 12:335–343. doi:10.1038/nrclinonc.2015.63
Kinoshita A et al (2012) Comparison of the prognostic value of inflammation-based prognostic scores in patients with hepatocellular carcinoma. Br J Cancer 107:988–993. doi:10.1038/bjc.2012.354
Koukourakis MI, Kambouromiti G, Pitsiava D, Tsousou P, Tsiarkatsi M, Kartalis G (2009) Serum C-reactive protein (CRP) levels in cancer patients are linked with tumor burden and are reduced by anti-hypertensive medication. Inflammation 32:169–175. doi:10.1007/s10753-009-9116-4
Li MX et al (2015) Prognostic role of Glasgow Prognostic Score in patients with hepatocellular carcinoma: a systematic review and meta-analysis. Medicine 94:e2133. doi:10.1097/md.0000000000002133
Lu Y, Huang S, Li P, Chen B, Liu W, Chen Z, Yin F (2015) Prognostic evaluation of preoperative serum C-reactive protein concentration in patients with epithelial ovarian cancer. Exp Ther Med 9:2003–2007. doi:10.3892/etm.2015.2350
Ma LN et al (2015) Serum high-sensitivity C-reactive protein are associated with HBV replication, liver damage and fibrosis in patients with chronic hepatitis B. Hepatogastroenterology 62:368–372
Marnell L, Mold C, Du Clos TW (2005) C-reactive protein: ligands, receptors and role in inflammation. Clini Immunol (Orlando, Fla) 117:104–111. doi:10.1016/j.clim.2005.08.004
McMillan DC (2009) Systemic inflammation, nutritional status and survival in patients with cancer. Curr Opin Clin Nutr Metab Care 12:223–226. doi:10.1097/MCO.0b013e32832a7902
Ogita M et al (2015) Impact of combined C-reactive protein and high-density lipoprotein cholesterol levels on long-term outcomes in patients with coronary artery disease after a first percutaneous coronary intervention. Am J Cardiol 116:999–1002. doi:10.1016/j.amjcard.2015.06.036
Ramsey S, Lamb GW, Aitchison M, Graham J, McMillan DC (2007) Evaluation of an inflammation-based prognostic score in patients with metastatic renal cancer. Cancer 109:205–212. doi:10.1002/cncr.22400
Ren G et al (2012) CCR2-dependent recruitment of macrophages by tumor-educated mesenchymal stromal cells promotes tumor development and is mimicked by TNFalpha. Cell Stem Cell 11:812–824. doi:10.1016/j.stem.2012.08.013
Schwartz PE, Zheng W (2003) Neoadjuvant chemotherapy for advanced ovarian cancer: the role of cytology in pretreatment diagnosis. Gynecol Oncol 90:644–650
Smith JJ, Garcia-Aguilar J (2015) Advances and challenges in treatment of locally advanced rectal cancer. J Clin Oncol 33:1797–1808. doi:10.1200/jco.2014.60.1054
Therasse P et al (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216
Thurner EM et al (2015) The elevated C-reactive protein level is associated with poor prognosis in prostate cancer patients treated with radiotherapy. Eur J Cancer (Oxford England: 1990) 51:610–619. doi:10.1016/j.ejca.2015.01.002
Yang J et al (2007) Human C-reactive protein binds activating Fcgamma receptors and protects myeloma tumor cells from apoptosis. Cancer Cell 12:252–265. doi:10.1016/j.ccr.2007.08.008
Zheng J, Wu Y, Li Z, Wang H, Xiao W, Shi Y, Wang T (2016) Low serum total bilirubin concentration was associated with increased high sensitive C reactive protein level in patients with impaired glucose tolerance and type 2 diabetes mellitus subjects. Clin Lab 62:901–907
Acknowledgments
We thank the patients and their families and all the investigators, including the physicians, nurses and laboratory technicians in this study.
Funding
Our work was supported by the following funds: Science and Technology Projects of Guangdong Province (contract/Grant Numbers: 2014A020212577), Young Teachers’ Cultivation Project of Sun Yat-Sen University (No. 16ykpy30) and Medical Research Foundation of Guangdong Province (contract/Grant Number: A2015008).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Jiayu Zhu, Hua Wang, Cheng-Cheng Liu, Yue Lu and Hailin Tang declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Jia-yu Zhu, Hua Wang and Cheng-Cheng Liu contributed equally to this article.
Rights and permissions
About this article
Cite this article
Zhu, J., Wang, H., Liu, CC. et al. The Glasgow Prognostic Score (GPS) is a novel prognostic indicator in advanced epithelial ovarian cancer: a multicenter retrospective study. J Cancer Res Clin Oncol 142, 2339–2345 (2016). https://doi.org/10.1007/s00432-016-2228-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-016-2228-y