Abstract
Purpose
Stathmin (Oncoprotein18), a ubiquitous and highly conserved 19-kDa cytosolic phosphoprotein, has been reported to play a critical role in mitosis and possibly other cellular processes, which is associated with tumor carcinogenesis and development. The purpose of this study was to examine the involvement of stathmin in human cervical carcinogenesis and to evaluate its prognostic significance in human cervical carcinoma.
Methods
Using semiquantitative RT-PCR and Western blotting, we detected the expression of stathmin in human normal cervical epithelial cell line, immortalized cervical epithelial cell lines, and cervical carcinoma cell lines. Additionally, we also detected the expression of stathmin protein in 15 cases of cervical carcinoma tissues and adjacent non-carcinomous margin tissues. Furthermore, specimens from 148 patients with different grade and stage cervical carcinoma were investigated by immunohistochemistry for stathmin expression. Correlations between the expression of stathmin and various clinicopathological factors were studied, while statistical analyses were performed to evaluate prognostic and diagnostic associations.
Results
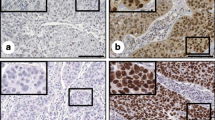
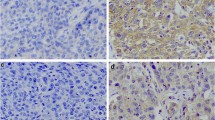
The levels of stathmin mRNA and protein expression were significantly higher in cervical carcinoma cells and immortalized cervical epithelial cells than in normal cervical epithelial cells (P < 0.05). Moreover, Western blotting revealed high stathmin protein expression in 73.3% (11/15) cervical carcinoma tissues, while stathmin were overexpressed in tumor tissues as compared with adjacent non-carcinomous margin samples (P = 0.017). In addition, immunohistochemical staining revealed stathmin immunoreactivity in 81.1% (120/148) of cervical carcinoma tissues and high stathmin expression was significantly correlated with clinical stage (P = 0.006), T classification (P = 0.012), regional lymph node metastasis (P = 0.005) and hematogenous metastasis (P = 0.021). Kaplan–Meier analysis showed that high stathmin positivity was significantly associated with a shorter survival time (P < 0.001). Clinical stage (P = 0.0022), T classification (P = 0.0035), regional lymph node (P = 0.0008) or hematogenous metastasis (P = 0.0015) were also associated with survival time. Furthermore, by Cox multivariate analysis, only lymph node (P = 0.0052) or hematogenous metastasis (P = 0.0046) maintained their significance as independent prognostic factors, although stathmin was not an independent prognostic factor (risk ratio: 1.45; P = 0.0624).
Conclusions
Stathmin expression correlates with cervical carcinogenesis and tumor progression. This molecule is a valuable prognostic marker in patients with cervical carcinoma.





Similar content being viewed by others
References
Arends MJ, Buckley CH, Wells M (1998) Aetiology, pathogenesis, and pathology of cervical neoplasia. J Clin Pathol 51:96–103
Belletti B, Nicoloso MS, Schiappacassi M, Berton S, Lovat F, Wolf K, Canzonieri V, D’Andrea S, Zucchetto A, Friedl P, Colombatti A, Baldassarre G (2008) Stathmin activity influences sarcoma cell shape, motility, and metastatic potential. Mol Biol Cell 19:2003–2013. doi:10.1091/mbc.E07-09-0894
Belmont LD, Mitchison TJ (1996) Identification of a protein that interacts with tubulin dimers and increases the catastrophe rate of microtubules. Cell 84:623–631. doi:10.1016/S0092-8674(00)81037-5
Bièche I, Lachkar S, Becette V, Cifuentes-Diaz C, Sobel A, Lidereau R, Curmi PA (1998) Overexpression of the stathmin gene in a subset of human breast cancer. Br J Cancer 78:701–719
Brattsand G (2000) Correlation of oncoprotein 18/stathmin expression in human breast cancer with established prognostic factors. Br J Cancer 83:311–318. doi:10.1054/bjoc.2000.1264
Chen G, Wang H, Miller CT, Thomas DG, Gharib TG, Misek DE, Giordano TJ, Orringer MB, Hanash SM, Beer DG (2003) Overexpression of oncoprotein 18 correlates with poor differentiation in lung adenocarcinomas. Mol Cell Proteomics 2:107–116. doi:10.1074/mcp.M200055-MCP200
Duggan MA (2002) A review of the natural history of cervical intraepithelial neoplasia. Gan To Kagaku Ryoho 29:176–193
Duk JM, Groenier KH, de Bruijn HW, Hollema H, ten Hoor KA, van der Zee AG, Aalders JG (1996) Pretreatment serum squamous cell carcinoma antigen: a newly identified prognostic factor in early-stage cervical carcinoma. J Clin Oncol 14:111–118
Friedrich B, Grönberg H, Landström M, Gullberg M, Bergh A (1995) Differentiation stage specific expression of oncoprotein 18 in human and rat prostatic adenocarcinoma. Prostate 27:102–109. doi:10.1002/pros.2990270207
Gasinska A, Urbanski K, Adamczyk A, Pudelek J, Lind BK, Brahme A (2002) Prognostic significance of intratumour microvessel density and haemoglobin level in carcinoma of the uterine cervix. Acta Oncol 41:437–443. doi:10.1080/028418602320405023
Ghosh PK, Anderson J, Cohen N, Takeshita K, Atweh GF, Lebowitz P (1993) Expression of the leukemia-associated gene, p18, in normal and malignant tissues: inactivation of expression in a patient with cleaved B cell lymphoma/leukemia. Oncogene 8:2869–2872
Hanash SM, Strahler JR, Kuick R, Chu EH, Nichols D (1988) Identification of a polypeptide associated with the malignant phenotype in acute leukemia. J Biol Chem 263:12813–12815
Huang FY, Kwok YK, Lau ET, Tang MH, Ng TY, Ngan HY (2005) Genetic abnormalities and HPV status in cervical and vulvar squamous cell carcinomas. Cancer Genet Cytogenet 157:42–48. doi:10.1016/j.cancergencyto.2004.06.002
Jemal A, Murray T, Samuels A, Ghafoor A, Ward E, Thun MJ (2003) Cancer statistics, 2003. CA cancer. J Clin 53:5–26
Kawagoe T, Kashimura M, Matsuura Y, Sugihara K, Toki N, Aoki T (1999) Clinical significance of tumor size in stage IB and II carcinoma of the uterine cervix. Int J Gynecol Cancer 9:421–426. doi:10.1046/j.1525-1438.1999.99059.x
Koike H, Uzawa K, Nakashima D, Shimada K, Kato Y, Higo M, Kouzu Y, Endo Y, Kasamatsu A, Tanzawa H (2005) Identification of differentially expressed proteins in oral squamous-cell carcinoma using a global proteomic approach. Int J Oncol 27:59–67
Kouzu Y, Uzawa K, Koike H, Saito K, Nakashima D, Higo M, Endo Y, Kasamatsu A, Shiiba M, Bukawa H, Yokoe H, Tanzawa H, Kouzu Y, Uzawa K, Koike H (2006) Overexpression of stathmin in oral squamous-cell carcinoma: correlation with tumour progression and poor prognosis. Br J Cancer 94:717–723
Laird AD, Shalloway D (1997) Oncoprotein signalling and mitosis. Cell Signal 9:249–255. doi:10.1016/S0898-6568(96)00176-3
Luo XN, Mookerjee B, Ferrari A, Mistry S, Atweh GF (1994) Regulation of phosphoprotein p18 in leukemic cells. Cell cycle regulated phosphorylation by p34cdc2 kinase. J Biol Chem 269:10312–10318
Marklund U, Osterman O, Melander H, Bergh A, Gullberg M (1994) The phenotype of a “Cdc2 kinase target site-deficient” mutant of oncoprotein18 reveals a role of this protein in cell cycle control. J Biol Chem 269:30626–30635
Martin-Loeches M, Ortí RM, Cazorla E, Asins E, Llixiona J (2002) Multivariate analysis of the morphometric characteristics of tumours as prognostic factors in the survival of patients with uterine cervix cancer treated with radical surgery. Eur J Obstet Gynecol Reprod Biol 105:170–176
McGrogan BT, Gilmartin B, Carney DN, McCann A (2008) Taxanes, microtubules and chemoresistant breast cancer. Biochim Biophys Acta 1785:96–132
Melhem R, Hailat N, Kuick R, Hanash SM (1997) Quantitative analysis of Op18 phosphorylation in childhood acute leukemia. Leukemia 11:1690–1695. doi:10.1038/sj.leu.2400792
Parkin DM, Bray F, Ferlay J, Pisani P (2005) Global cancer statistics, 2002. CA Cancer J Clin 55:74–108
Price DK, Ball JR, Bahrani-Mostafavi Z, Vachris JC, Kaufman JS, Naumann RW, Higgins RV, Hall JB (2000) The phosphoprotein Op18/stathmin is differentially expressed in ovarian cancer. Cancer Invest 18:722–730. doi:10.3109/07357900009012204
Rubin CI, Atweh GF (2004) The role of stathmin in the regulation of the cell cycle. J Cell Biochem 93:242–250. doi:10.1002/jcb.20187
Scambia G, Benedetti Panici P, Foti E, Amoroso M, Salerno G, Ferrandina G, Battaglia F, Greggi S, De Gaetano A, Puglia G (1994) Squamous cell carcinoma antigen: prognostic significance and role in the monitoring of neoadjuvant chemotherapy response in cervical cancer. J Clin Oncol 12:2309–2316
Takahashi M, Yang XJ, Lavery TT, Furge KA, Williams BO, Tretiakova M, Montag A, Vogelzang NJ, Re GG, Garvin AJ, Söderhäll S, Kagawa S, Hazel-Martin D, Nordenskjold A, Teh BT (2002) Gene expression profiling of favorable histology Wilms tumors and its correlation with clinical features. Cancer Res 62:6598–6605
Wang R, Dong K, Lin F, Wang X, Gao P, Wei SH, Cheng SY, Zhang HZ (2007) Inhibiting proliferation and enhancing chemosensitivity to taxanes in osteosarcoma cells by RNA interference-mediated downregulation of stathmin expression. Mol Med 13:567–575. doi:10.2119/2007-00046.Wang
Wei SH, Lin F, Wang X, Gao P, Zhang HZ (2008) Prognostic significance of stathmin expression in correlation with metastasis and clinicopathological characteristics in human ovarian carcinoma. Acta Histochem 110:59–65. doi:10.1016/j.acthis.2007.06.002
Zhang HZ, Gao P, Yan L, Lin F (2004) Significance of stathmin gene overexpression in osteosarcoma cells. Ai Zheng 23:493–496
Zhang HZ, Wang Y, Gao P, Lin F, Liu L, Yu B, Ren JH, Zhao H, Wang R (2006) Silencing stathmin gene expression by survivin promoter-driven siRNA vector to reverse malignant phenotype of tumor cells. Cancer Biol Ther 5:1457–1461
Acknowledgment
Thanks to every one of the Department of Clinical Laboratory for their sincere help and excellent technical assitance.
Author information
Authors and Affiliations
Corresponding author
Additional information
W. Xi and W. Rui contributed equally to this study, should be regarded as joint First authors.
Rights and permissions
About this article
Cite this article
Xi, W., Rui, W., Fang, L. et al. Expression of stathmin/op18 as a significant prognostic factor for cervical carcinoma patients. J Cancer Res Clin Oncol 135, 837–846 (2009). https://doi.org/10.1007/s00432-008-0520-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-008-0520-1