Abstract
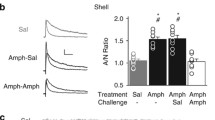
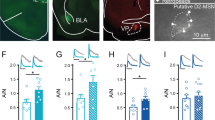
Despite evidence that morphine-related pathologies reflect adaptations in NAc glutamate signaling, substantial gaps in basic information remain. The current study examines the impact of non-contingent acute, repeated, and withdrawal-inducing morphine dosing regimens on glutamate transmission in D1- or D2-MSNs in the nucleus accumbens shell (NAcSh) and core (NAcC) sub-regions in hopes of identifying excitatory plasticity that may contribute to unique facets of opioid addiction-related behavior. Following an acute morphine injection (10 mg/kg), average miniature excitatory postsynaptic current (mEPSC) amplitude mediated by AMPA-type glutamate receptors was increased at D1-MSNs in the both the NAcShl and NAcC, whereas only the frequency of events was elevated at D2-MSNs in the NAcSh. In contrast, spontaneous somatic withdrawal induced by escalating dose of repeated morphine twice per day (20, 40, 60, 80, 100 mg/kg) enhanced mEPSC frequency specifically at D2-MSNs in the NAcSh. Similar to previous findings, excitatory drive was elevated at NAcSh D1-MSNs after 10–14 days home cage abstinence. Following abstinence, an acute drug re-exposure produced a rapid and enduring endocytosis of GluA2-containing AMPARs at D1-MSNs in the shell, that when blocked by an intra-NAc shell infusion of the Tat-GluA23Y peptide, increased reinstatement of morphine place preference—a phenomenon distinctly different than effects previously found with cocaine. The present study is the first to directly identify unique circuit specific adaptations in NAc glutamate synaptic transmission associated with morphine-related acute reward and somatic withdrawal as well as post-abstinence short-term plasticity. Moreover, while differing classes of abused drugs (i.e., psychostimulants and opioids) produce seemingly similar bidirectional plasticity in the NAc following drug re-exposure, our findings indicate this plasticity has distinct behavioral consequences.




Similar content being viewed by others
References
Ahmadian G et al (2004) Tyrosine phosphorylation of GluR2 is required for insulin-stimulated AMPA receptor endocytosis and LTD. EMBO J 23:1040–1050. https://doi.org/10.1038/sj.emboj.7600126
Al-Hasani R et al (2015) Distinct subpopulations of nucleus accumbens dynorphin neurons drive aversion and reward. Neuron 87:1063–1077. https://doi.org/10.1016/j.neuron.2015.08.019
Badiani A, Belin D, Epstein D, Calu D, Shaham Y (2011) Opiate versus psychostimulant addiction: the differences do matter. Nat Rev Neurosci 12:685–700. https://doi.org/10.1038/nrn3104
Baharlouei N, Sarihi A, Komaki A, Shahidi S, Haghparast A (2015) Blockage of acquisition and expression of morphine-induced conditioned place preference in rats due to activation of glutamate receptors type II/III in nucleus accumbens. Pharmacol Biochem Behav 135:192–198. https://doi.org/10.1016/j.pbb.2015.06.004
Benneyworth MA et al (2019) Synaptic depotentiation and mGluR5 activity in the nucleus accumbens drives cocaine-primed reinstatement of place preference. J Neurosci. https://doi.org/10.1523/JNEUROSCI.3020-17.2019
Bossert JM, Busch RF, Gray SM (2005) The novel mGluR2/3 agonist LY379268 attenuates cue-induced reinstatement of heroin seeking. NeuroReport 16:1013–1016
Bossert JM, Gray SM, Lu L, Shaham Y (2006) Activation of group II metabotropic glutamate receptors in the nucleus accumbens shell attenuates context-induced relapse to heroin seeking. Neuropsychopharmacology 31:2197–2209. https://doi.org/10.1038/sj.npp.1300977
Brebner K et al (2005) Nucleus accumbens long-term depression and the expression of behavioral sensitization. Science 310:1340–1343. https://doi.org/10.1126/science.1116894
Chartoff EH, Connery HS (2014) It’s MORe exciting than mu: crosstalk between mu opioid receptors and glutamatergic transmission in the mesolimbic dopamine system. Front Pharmacol 5:116. https://doi.org/10.3389/fphar.2014.00116
Cruz HG, Berton F, Sollini M, Blanchet C, Pravetoni M, Wickman K, Luscher C (2008) Absence and rescue of morphine withdrawal in GIRK/Kir3 knock-out mice. J Neurosci 28:4069–4077. https://doi.org/10.1523/jneurosci.0267-08.2008
Desole MS et al (1996) Effects of morphine treatment and withdrawal on striatal and limbic monoaminergic activity and ascorbic acid oxidation in the rat. Brain Res 723:154–161
Dobi A, Seabold GK, Christensen CH, Bock R, Alvarez VA (2011) Cocaine-induced plasticity in the nucleus accumbens is cell specific and develops without prolonged withdrawal. J Neurosci 31:1895–1904. https://doi.org/10.1523/JNEUROSCI.5375-10.2011
Dong Z et al (2015) Long-term potentiation decay and memory loss are mediated by AMPAR endocytosis. J Clin Investig 125:234–247. https://doi.org/10.1172/JCI77888
Dworkin SI, Guerin GF, Goeders NE, Smith JE (1988) Kainic acid lesions of the nucleus accumbens selectively attenuate morphine self-administration. Pharmacol Biochem Behav 29:175–181
Ebner SR, Larson EB, Hearing MC, Ingebretson AE, Thomas MJ (2018) Extinction and reinstatement of cocaine-seeking in self-administering mice is associated with bidirectional AMPAR-mediated plasticity in the nucleus accumbens shell. Neuroscience 384:340–349. https://doi.org/10.1016/j.neuroscience.2018.05.043
Enrico P et al (1998) Effect of naloxone on morphine-induced changes in striatal dopamine metabolism and glutamate, ascorbic acid and uric acid release in freely moving rats. Brain Res 797:94–102
Everitt BJ, Parkinson JA, Olmstead MC, Arroyo M, Robledo P, Robbins TW (1999) Associative processes in addiction and reward the role of amygdala-ventral striatal subsystems. Ann N Y Acad Sci 877:412–438
Famous KR, Kumaresan V, Sadri-Vakili G, Schmidt HD, Mierke DF, Cha JH, Pierce RC (2008) Phosphorylation-dependent trafficking of GluR2-containing AMPA receptors in the nucleus accumbens plays a critical role in the reinstatement of cocaine seeking. J Neurosci 28:11061–11070. https://doi.org/10.1523/jneurosci.1221-08.2008
Fujio M et al (2005) Gene transfer of GLT-1, a glutamate transporter, into the nucleus accumbens shell attenuates methamphetamine- and morphine-induced conditioned place preference in rats. Eur J Neurosci 22:2744–2754. https://doi.org/10.1111/j.1460-9568.2005.04467.x
Gipson CD, Kupchik YM, Shen H, Reissner KJ, Thomas CA, Kalivas PW (2013a) Relapse induced by cues predicting cocaine depends on rapid, transient synaptic potentiation. Neuron 77:867–872. https://doi.org/10.1016/j.neuron.2013.01.005
Gipson CD, Reissner KJ, Kupchik YM, Smith AC, Stankeviciute N, Hensley-Simon ME, Kalivas PW (2013b) Reinstatement of nicotine seeking is mediated by glutamatergic plasticity. Proc Natl Acad Sci USA 110:9124–9129. https://doi.org/10.1073/pnas.1220591110
Gracy KN, Dankiewicz LA, Koob GF (2001) Opiate withdrawal-induced fos immunoreactivity in the rat extended amygdala parallels the development of conditioned place aversion. Neuropsychopharmacology 24:152–160. https://doi.org/10.1016/S0893-133X(00)00186-X
Graziane NM et al (2016) Opposing mechanisms mediate morphine- and cocaine-induced generation of silent synapses. Nat Neurosci 19:915–925. https://doi.org/10.1038/nn.4313
Harris GC, Aston-Jones G (1994) Involvement of D2 dopamine receptors in the nucleus accumbens in the opiate withdrawal syndrome. Nature 371:155–157. https://doi.org/10.1038/371155a0
Hearing M (2019) Prefrontal-accumbens opioid plasticity: implications for relapse and dependence. Pharmacol Res 139:158–165. https://doi.org/10.1016/j.phrs.2018.11.012
Hearing M, Kotecki L, de Velasco EMF, Fajardo-Serrano A, Chung HJ, Lujan R, Wickman K (2013) Repeated cocaine weakens GABA(B)-Girk signaling in layer 5/6 pyramidal neurons in the prelimbic cortex. Neuron 80:159–170. https://doi.org/10.1016/j.neuron.2013.07.019
Hearing MC et al (2016) Reversal of morphine-induced cell-type-specific synaptic plasticity in the nucleus accumbens shell blocks reinstatement. Proc Natl Acad Sci USA 113:757–762. https://doi.org/10.1073/pnas.1519248113
Hearing M, Graziane N, Dong Y, Thomas MJ (2018) Opioid and psychostimulant plasticity: targeting overlap in nucleus accumbens glutamate signaling. Trends Pharmacol Sci 39:276–294. https://doi.org/10.1016/j.tips.2017.12.004
Heimer L, Zahm DS, Churchill L, Kalivas PW, Wohltmann C (1991) Specificity in the projection patterns of accumbal core and shell in the rat. Neuroscience 41:89–125
Heimer L, Alheid GF, de Olmos JS, Groenewegen HJ, Haber SN, Harlan RE, Zahm DS (1997) The accumbens: beyond the core-shell dichotomy. J Neuropsychiatry Clin Neurosci 9:354–381. https://doi.org/10.1176/jnp.9.3.354
Herrold AA, Persons AL, Napier TC (2013) Cellular distribution of AMPA receptor subunits and mGlu5 following acute and repeated administration of morphine or methamphetamine. J Neurochem 126:503–517. https://doi.org/10.1111/jnc.12323
Hipps PP, Eveland MR, Meyer ER, Sherman WR, Cicero TJ (1976) Mass fragmentography of morphine: relationship between brain levels and analgesic activity. J Pharmacol Exp Ther 196:642–648
Ingebretson AE, Hearing MC, Huffington ED, Thomas MJ (2018) Endogenous dopamine and endocannabinoid signaling mediate cocaine-induced reversal of AMPAR synaptic potentiation in the nucleus accumbens shell. Neuropharmacology 131:154–165. https://doi.org/10.1016/j.neuropharm.2017.12.011
Jacobs EH, Wardeh G, Smit AB, Schoffelmeer AN (2005) Morphine causes a delayed increase in glutamate receptor functioning in the nucleus accumbens core. Eur J Pharmacol 511:27–30. https://doi.org/10.1016/j.ejphar.2005.02.009
Jedynak J et al (2016) Cocaine and amphetamine induce overlapping but distinct patterns of AMPAR plasticity in nucleus accumbens medium spiny neurons. Neuropsychopharmacology 41:464–476. https://doi.org/10.1038/npp.2015.168
Kalivas PW (2009) The glutamate homeostasis hypothesis of addiction. Nat Rev Neurosci 10:561–572. https://doi.org/10.1038/nrn2515
Kalivas PW, Hu XT (2006) Exciting inhibition in psychostimulant addiction. Trends Neurosci 29:610–616. https://doi.org/10.1016/j.tins.2006.08.008
Kerchner GA, Nicoll RA (2008) Silent synapses and the emergence of a postsynaptic mechanism for LTP. Nat Rev Neurosci 9:813–825. https://doi.org/10.1038/nrn2501
Kim J, Park BH, Lee JH, Park SK, Kim JH (2011) Cell type-specific alterations in the nucleus accumbens by repeated exposures to cocaine. Biol Psychiatry 69:1026–1034. https://doi.org/10.1016/j.biopsych.2011.01.013
Koob GF, Wall TL, Bloom FE (1989) Nucleus accumbens as a substrate for the aversive stimulus effects of opiate withdrawal. Psychopharmacology 98:530–534
Kourrich S, Rothwell PE, Klug JR, Thomas MJ (2007) Cocaine experience controls bidirectional synaptic plasticity in the nucleus accumbens. J Neurosci 27:7921–7928. https://doi.org/10.1523/JNEUROSCI.1859-07.2007
Le Moine C, Bloch B (1995) D1 and D2 dopamine receptor gene expression in the rat striatum: sensitive cRNA probes demonstrate prominent segregation of D1 and D2 mRNAs in distinct neuronal populations of the dorsal and ventral striatum. J Comp Neurol 355:418–426. https://doi.org/10.1002/cne.903550308
Lee SH, Liu L, Wang YT, Sheng M (2002) Clathrin adaptor AP2 and NSF interact with overlapping sites of GluR2 and play distinct roles in AMPA receptor trafficking and hippocampal LTD. Neuron 36:661–674
Lobo DS, Kennedy JL (2006) The genetics of gambling and behavioral addictions. CNS Spectr 11:931–939
Lobo MK et al (2010) Cell type-specific loss of BDNF signaling mimics optogenetic control of cocaine reward. Science 330:385–390. https://doi.org/10.1126/science.1188472
Martin G, Nie Z, Siggins GR (1997) μ-Opioid receptors modulate NMDA receptor-mediated responses in nucleus accumbens neurons. J Neurosci 17:11–22
Olds ME (1982) Reinforcing effects of morphine in the nucleus accumbens. Brain Res 237:429–440
Ortinski PI, Vassoler FM, Carlson GC, Pierce RC (2012) Temporally dependent changes in cocaine-induced synaptic plasticity in the nucleus accumbens shell are reversed by D1-like dopamine receptor stimulation. Neuropsychopharmacology 37:1671–1682. https://doi.org/10.1038/npp.2012.12
Ortinski PI, Briand LA, Pierce RC, Schmidt HD (2015) Cocaine-seeking is associated with PKC-dependent reduction of excitatory signaling in accumbens shell D2 dopamine receptor-expressing neurons. Neuropharmacology 92:80–89. https://doi.org/10.1016/j.neuropharm.2015.01.002
Papaleo F, Contarino A (2006) Gender- and morphine dose-linked expression of spontaneous somatic opiate withdrawal in mice. Behav Brain Res 170:110–118. https://doi.org/10.1016/j.bbr.2006.02.009
Pascoli V, Turiault M, Luscher C (2011) Reversal of cocaine-evoked synaptic potentiation resets drug-induced adaptive behaviour. Nature 481:71–75. https://doi.org/10.1038/nature10709
Pascoli V, Terrier J, Espallergues J, Valjent E, O’Connor EC, Luscher C (2014) Contrasting forms of cocaine-evoked plasticity control components of relapse. Nature 509:459–464. https://doi.org/10.1038/nature13257
Reynolds SM, Berridge KC (2003) Glutamate motivational ensembles in nucleus accumbens: rostrocaudal shell gradients of fear and feeding. Eur J Neurosci 17:2187–2200
Rothwell PE, Kourrich S, Thomas MJ (2011) Synaptic adaptations in the nucleus accumbens caused by experiences linked to relapse. Biol Psychiatry 69:1124–1126. https://doi.org/10.1016/j.biopsych.2010.12.028
Rothwell PE, Thomas MJ, Gewirtz JC (2012) Protracted manifestations of acute dependence after a single morphine exposure. Psychopharmacology 219:991–998. https://doi.org/10.1007/s00213-011-2425-y
Russell SE, Puttick DJ, Sawyer AM, Potter DN, Mague S, Carlezon WA Jr, Chartoff EH (2016) Nucleus accumbens AMPA receptors are necessary for morphine-withdrawal-induced negative-affective states in rats. J Neurosci 36:5748–5762. https://doi.org/10.1523/jneurosci.2875-12.2016
Schmidt HD, Schassburger RL, Guercio LA, Pierce RC (2013) Stimulation of mGluR5 in the accumbens shell promotes cocaine seeking by activating PKC gamma. J Neurosci 33:14160–14169. https://doi.org/10.1523/jneurosci.2284-13.2013
Schmidt HD, Kimmey BA, Arreola AC, Pierce RC (2015) Group I metabotropic glutamate receptor-mediated activation of PKC gamma in the nucleus accumbens core promotes the reinstatement of cocaine seeking. Addict Biol 20:285–296. https://doi.org/10.1111/adb.12122
Scholz R, Berberich S, Rathgeber L, Kolleker A, Kohr G, Kornau HC (2010) AMPA receptor signaling through BRAG2 and Arf6 critical for long-term synaptic depression. Neuron 66:768–780. https://doi.org/10.1016/j.neuron.2010.05.003
Schulteis G, Markou A, Gold LH, Stinus L, Koob GF (1994) Relative sensitivity to naloxone of multiple indices of opiate withdrawal: a quantitative dose–response analysis. J Pharmacol Exp Ther 271:1391–1398
Sepulveda MJ, Hernandez L, Rada P, Tucci S, Contreras E (1998) Effect of precipitated withdrawal on extracellular glutamate and aspartate in the nucleus accumbens of chronically morphine-treated rats: an in vivo microdialysis study. Pharmacol Biochem Behav 60:255–262
Sepulveda J, Oliva P, Contreras E (2004) Neurochemical changes of the extracellular concentrations of glutamate and aspartate in the nucleus accumbens of rats after chronic administration of morphine. Eur J Pharmacol 483:249–258
Sesack SR, Grace AA (2010) Cortico-basal ganglia reward network: microcircuitry. Neuropsychopharmacology 35:27–47. https://doi.org/10.1038/npp.2009.93
Shen H, Moussawi K, Zhou W, Toda S, Kalivas PW (2011) Heroin relapse requires long-term potentiation-like plasticity mediated by NMDA2b-containing receptors. Proc Natl Acad Sci USA 108:19407–19412. https://doi.org/10.1073/pnas.1112052108
Shen HW, Scofield MD, Boger H, Hensley M, Kalivas PW (2014) Synaptic glutamate spillover due to impaired glutamate uptake mediates heroin relapse. J Neurosci 34:5649–5657. https://doi.org/10.1523/JNEUROSCI.4564-13.2014
Shiflett MW, Balleine BW (2011) Molecular substrates of action control in cortico-striatal circuits. Prog Neurobiol 95:1–13. https://doi.org/10.1016/j.pneurobio.2011.05.007
Siahposht-Khachaki A, Fatahi Z, Yans A, Khodagholi F, Haghparast A (2017) Involvement of AMPA/kainate glutamate receptor in the extinction and reinstatement of morphine-induced conditioned place preference: a behavioral and molecular study. Cell Mol Neurobiol 37:315–328. https://doi.org/10.1007/s10571-016-0371-2
Smith RJ, Lobo MK, Spencer S, Kalivas PW (2013) Cocaine-induced adaptations in D1 and D2 accumbens projection neurons (a dichotomy not necessarily synonymous with direct and indirect pathways). Curr Opin Neurobiol 23:546–552. https://doi.org/10.1016/j.conb.2013.01.026
Suska A, Lee BR, Huang YH, Dong Y, Schluter OM (2013) Selective presynaptic enhancement of the prefrontal cortex to nucleus accumbens pathway by cocaine. Proc Natl Acad Sci USA 110:713–718. https://doi.org/10.1073/pnas.1206287110
Svingos AL, Moriwaki A, Wang JB, Uhl GR, Pickel VM (1997) μ-Opioid receptors are localized to extrasynaptic plasma membranes of GABAergic neurons and their targets in the rat nucleus accumbens. J Neurosci 17:2585–2594
Terrier J, Luscher C, Pascoli V (2016) Cell-type specific insertion of GluA2-lacking AMPARs with cocaine exposure leading to sensitization cue-induced seeking, and incubation of craving. Neuropsychopharmacology 41:1779–1789. https://doi.org/10.1038/npp.2015.345
Thomas MJ, Beurrier C, Bonci A, Malenka RC (2001) Long-term depression in the nucleus accumbens: a neural correlate of behavioral sensitization to cocaine. Nat Neurosci 4:1217–1223. https://doi.org/10.1038/nn757
Trantham-Davidson H, LaLumiere RT, Reissner KJ, Kalivas PW, Knackstedt LA (2012) Ceftriaxone normalizes nucleus accumbens synaptic transmission, glutamate transport, and export following cocaine self-administration and extinction training. J Neurosci 32:12406–12410. https://doi.org/10.1523/jneurosci.1976-12.2012
Turrigiano GG (2008) The self-tuning neuron: synaptic scaling of excitatory synapses. Cell 135:422–435. https://doi.org/10.1016/j.cell.2008.10.008
van der Laan JW, de Groot G (1988) Changes in locomotor-activity patterns as a measure of spontaneous morphine withdrawal: no effect of clonidine. Drug Alcohol Depend 22:133–140
van der Laan JW, van’t Land CJ, Loeber JG, de Groot GV (1991) Validation of spontaneous morphine withdrawal symptoms in rats. Arch Int Pharmacodyn Ther 311:32–45
van Ree JM, Gerrits MA, Vanderschuren LJ (1999) Opioids, reward and addiction: an encounter of biology, psychology, and medicine. Pharmacol Rev 51:341–396
Vekovischeva OY et al (2001) Morphine-induced dependence and sensitization are altered in mice deficient in AMPA-type glutamate receptor-A subunits. J Neurosci 21:4451–4459
Voorn P, Vanderschuren LJ, Groenewegen HJ, Robbins TW, Pennartz CM (2004) Putting a spin on the dorsal-ventral divide of the striatum. Trends Neurosci 27:468–474. https://doi.org/10.1016/j.tins.2004.06.006
Wang Q, Li D, Bubula N, Campioni MR, McGehee DS, Vezina P (2017) Sensitizing exposure to amphetamine increases AMPA receptor phosphorylation without increasing cell surface expression in the rat nucleus accumbens. Neuropharmacology 117:328–337. https://doi.org/10.1016/j.neuropharm.2017.02.018
Wise RA (1989) Opiate reward: sites and substrates. Neurosci Biobehav Rev 13:129–133
Wydeven N et al (2014) Mechanisms underlying the activation of G-protein-gated inwardly rectifying K+ (GIRK) channels by the novel anxiolytic drug, ML297. Proc Natl Acad Sci USA 111:10755–10760. https://doi.org/10.1073/pnas.1405190111
Yoon BJ, Smith GB, Heynen AJ, Neve RL, Bear MF (2009) Essential role for a long-term depression mechanism in ocular dominance plasticity. Proc Natl Acad Sci USA 106:9860–9865. https://doi.org/10.1073/pnas.0901305106
Zahm DS, Brog JS (1992) On the significance of subterritories in the “accumbens” part of the rat ventral striatum. Neuroscience 50:751–767
Zhu Y, Wienecke CF, Nachtrab G, Chen X (2016) A thalamic input to the nucleus accumbens mediates opiate dependence. Nature 530:219–222. https://doi.org/10.1038/nature16954
Acknowledgements
The behavioral work is done in part with the support by the Mouse Behavior Core at the University of Minnesota, which received funding from the National Institute for Neurological Disorders and Stroke (P30 NS062158). These studies were also supported by funding from the National Institute on Drug Abuse Grant K99 DA038706 (to M.H.), R00DA038706 (M.H.), R00DA038706-04S1 (A.C.M), R01DA019666 (M.J.T.), K02DA035459 (M.J.T.) and T32 DA007234 (A.E.I.).
Funding
The following funding sources made the study possible: National Institute for Neurological Disorders and Stroke (P30 NS062158); National Institute on Drug Abuse Grant K99 DA038706 (to M.H.), R00DA038706 (M.H.), R00DA038706-04S1 (A.C.M), R01DA019666 (M.J.T.), K02DA035459 (M.J.T.) and T32 DA007234 (A.E.I.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose.
Ethical approval
The research in the current study used mice single or group housed on a 12-h light/dark cycle with food and water available ad libitum with experiments run during the light portion. All experiments were approved by the University of Minnesota and Marquette University Institutional Animal Care and Use Committee.
Informed consent
All authors have given their consent for manuscript submission.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Madayag, A.C., Gomez, D., Anderson, E.M. et al. Cell-type and region-specific nucleus accumbens AMPAR plasticity associated with morphine reward, reinstatement, and spontaneous withdrawal. Brain Struct Funct 224, 2311–2324 (2019). https://doi.org/10.1007/s00429-019-01903-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-019-01903-y