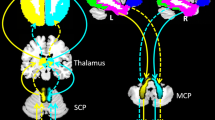
Abstract
The integrity of white matter architecture in the human brain is related to cognitive processing abilities. The corpus callosum is the largest white matter bundle interconnecting the two cerebral hemispheres. “Split-brain” patients in whom all cortical commissures have been severed to alleviate intractable epilepsy demonstrate remarkably intact cognitive abilities despite the lack of this important interhemispheric pathway. While it has often been speculated that there are compensatory alterations in the remaining interhemispheric fibers in split-brain patients several years post-commissurotomy, this has never been directly shown. Here we examined extra-callosal pathways for interhemispheric communication in the brain of a patient who underwent complete cerebral commissurotomy using diffusion weighted imaging tractography. We found that compared with a healthy age-matched comparison group, the split-brain patient exhibited increased fractional anisotropy (FA) of the dorsal and ventral pontine decussations of the cortico-cerebellar interhemispheric pathways. Few differences were observed between the patient and the comparison group with respect to FA of other long-range intrahemispheric fibers. These results point to specific cerebellar anatomical substrates that may account for the spared interhemispheric coordination and intact cognitive abilities that have been extensively documented in this unique patient.









Similar content being viewed by others
References
Adamaszek M, D’Agata F, Ferrucci R, Habas C, Keulen S, Kirkby KC, Leggio M, Marien P, Molinari M, Moulton E, Orsi L, Van Overwalle F, Papadelis C, Priori A, Sacchetti B, Schutter DJ, Styliadis C, Verhoeven J (2017) Consensus paper: cerebellum and emotion. Cerebellum 16(2):552–576. https://doi.org/10.1007/s12311-016-0815-8
Beaulieu C (2002) The basis of anisotropic water diffusion in the nervous system—a technical review. NMR Biomed 15(7–8):435–455. https://doi.org/10.1002/nbm.782
Bellebaum C, Daum I (2007) Cerebellar involvement in executive control. Cerebellum 6(3):184–192. https://doi.org/10.1080/14734220601169707
Bogen JE, Fisher ED, Vogel PJ (1965) Cerebral commissurotomy. A second case report. JAMA 194(12):1328–1329
Bogen JE, Schultz DH, Vogel PJ (1988) Completeness of callosotomy shown by magnetic resonance imaging in the long term. Arch Neurol 45(11):1203–1205
Campbell AL Jr, Bogen JE, Smith A (1981) Disorganization and reorganization of cognitive and sensorimotor functions in cerebral commissurotomy. Compensatory roles of the forebrain commissures and cerebral hemispheres in man. Brain 104(3):493–511
Cheng H, Wang Y, Sheng J, Sporns O, Kronenberger WG, Mathews VP, Hummer TA, Saykin AJ (2012) Optimization of seed density in DTI tractography for structural networks. J Neurosci Methods 203(1):264–272. https://doi.org/10.1016/j.jneumeth.2011.09.021
Clarke JM, Zaidel E (1989) Simple reaction times to lateralized light flashes. Varieties of interhemispheric communication routes. Brain 112(Pt 4):849–870
Clarke JM, Zaidel E (1994) Anatomical-behavioral relationships: corpus callosum morphometry and hemispheric specialization. Behav Brain Res 64(1–2):185–202
Eviatar Z, Zaidel E (1994) Letter matching within and between the disconnected hemispheres. Brain Cogn 25(1):128–137
Geschwind N (1965) Disconnexion syndromes in animals and man. I. Brain 88(2):237–294
Honey CJ, Sporns O, Cammoun L, Gigandet X, Thiran JP, Meuli R, Hagmann P (2009) Predicting human resting-state functional connectivity from structural connectivity. Proc Natl Acad Sci USA 106(6):2035–2040
Huebner EA, Strittmatter SM (2009) Axon regeneration in the peripheral and central nervous systems. Results Probl Cell Differ 48:339–351. https://doi.org/10.1007/400_2009_19
Jenkinson M, Pechaud M, Smith S (2005) BET2: MR-based estimation of brain, skull and scalp surfaces. In: 11th annual meeting of the organization for human brain mapping, Toronto
Jeurissen B, Leemans A, Tournier JD, Jones DK, Sijbers J (2013) Investigating the prevalence of complex fiber configurations in white matter tissue with diffusion magnetic resonance imaging. Hum Brain Mapp 34(11):2747–2766. https://doi.org/10.1002/hbm.22099
Johnston JM, Vaishnavi SN, Smyth MD, Zhang D, He BJ, Zempel JM, Shimony JS, Snyder AZ, Raichle ME (2008) Loss of resting interhemispheric functional connectivity after complete section of the corpus callosum. J Neurosci 28(25):6453–6458. https://doi.org/10.1523/jneurosci.0573-08.2008
Jones DK, Christiansen KF, Chapman RJ, Aggleton JP (2013) Distinct subdivisions of the cingulum bundle revealed by diffusion MRI fibre tracking: implications for neuropsychological investigations. Neuropsychologia 51(1):67–78. https://doi.org/10.1016/j.neuropsychologia.2012.11.018
Keser Z, Hasan KM, Mwangi BI, Kamali A, Ucisik-Keser FE, Riascos RF, Yozbatiran N, Francisco GE, Narayana PA (2015) Diffusion tensor imaging of the human cerebellar pathways and their interplay with cerebral macrostructure. Front Neuroanat 9:41. https://doi.org/10.3389/fnana.2015.00041
Leitner Y, Travis KE, Ben-Shachar M, Yeom KW, Feldman HM (2015) Tract profiles of the cerebellar white matter pathways in children and adolescents. Cerebellum 14(6):613–623. https://doi.org/10.1007/s12311-015-0652-1
Nooner KB, Colcombe SJ, Tobe RH, Mennes M, Benedict MM, Moreno AL, Panek LJ, Brown S, Zavitz ST, Li Q, Sikka S, Gutman D, Bangaru S, Schlachter RT, Kamiel SM, Anwar AR, Hinz CM, Kaplan MS, Rachlin AB, Adelsberg S, Cheung B, Khanuja R, Yan C, Craddock CC, Calhoun V, Courtney W, King M, Wood D, Cox CL, Kelly AM, Di Martino A, Petkova E, Reiss PT, Duan N, Thomsen D, Biswal B, Coffey B, Hoptman MJ, Javitt DC, Pomara N, Sidtis JJ, Koplewicz HS, Castellanos FX, Leventhal BL, Milham MP (2012) The NKI-rockland sample: a model for accelerating the pace of discovery science in psychiatry. Front Neurosci 6:152. https://doi.org/10.3389/fnins.2012.00152
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh Inventory. Neuropsychologia 9:97–113
O’Reilly JX, Croxson PL, Jbabdi S, Sallet J, Noonan MP, Mars RB, Browning PG, Wilson CR, Mitchell AS, Miller KL, Rushworth MF, Baxter MG (2013) Causal effect of disconnection lesions on interhemispheric functional connectivity in rhesus monkeys. Proc Natl Acad Sci USA 110(34):13982–13987. https://doi.org/10.1073/pnas.1305062110
Palesi F, De Rinaldis A, Castellazzi G, Calamante F, Muhlert N, Chard D, Tournier JD, Magenes G, D’Angelo E, Gandini Wheeler-Kingshott CAM (2017) Contralateral cortico-ponto-cerebellar pathways reconstruction in humans in vivo: implications for reciprocal cerebro-cerebellar structural connectivity in motor and non-motor areas. Sci Rep 7(1):12841. https://doi.org/10.1038/s41598-017-13079-8
Propper RE, O’Donnell LJ, Whalen S, Tie Y, Norton IH, Suarez RO, Zollei L, Radmanesh A, Golby AJ (2010) A combined fMRI and DTI examination of functional language lateralization and arcuate fasciculus structure: effects of degree versus direction of hand preference. Brain Cogn 73(2):85–92. https://doi.org/10.1016/j.bandc.2010.03.004
Rodrigo S, Oppenheim C, Chassoux F, Golestani N, Cointepas Y, Poupon C, Semah F, Mangin JF, Le Bihan D, Meder JF (2007) Uncinate fasciculus fiber tracking in mesial temporal lobe epilepsy. Initial findings. Eur Radiol 17(7):1663–1668. https://doi.org/10.1007/s00330-006-0558-x
Roland JL, Snyder AZ, Hacker CD, Mitra A, Shimony JS, Limbrick DD, Raichle ME, Smyth MD, Leuthardt EC (2017) On the role of the corpus callosum in interhemispheric functional connectivity in humans. Proc Natl Acad Sci USA 114(50):13278–13283. https://doi.org/10.1073/pnas.1707050114
Ruddy KL, Leemans A, Carson RG (2017) Transcallosal connectivity of the human cortical motor network. Brain Struct Funct 222(3):1243–1252. https://doi.org/10.1007/s00429-016-1274-1
Scholz J, Klein MC, Behrens TE, Johansen-Berg H (2009) Training induces changes in white-matter architecture. Nat Neurosci 12(11):1370–1371. https://doi.org/10.1038/nn.2412
Shadmehr R (2017) Distinct neural circuits for control of movement vs. holding still. J Neurophysiol 117(4):1431–1460. https://doi.org/10.1152/jn.00840.2016
Smith SM (2002) Fast robust automated brain extraction. Hum Brain Mapp 17(3):143–155. https://doi.org/10.1002/hbm.10062
Sokolov AA, Miall RC, Ivry RB (2017) The cerebellum: adaptive prediction for movement and cognition. Trends Cogn Sci 21(5):313–332. https://doi.org/10.1016/j.tics.2017.02.005
Uddin LQ (2013) Complex relationships between structural and functional brain connectivity. Trends Cogn Sci 17(12):600–602. https://doi.org/10.1016/j.tics.2013.09.011
Uddin LQ, Mooshagian E, Zaidel E, Scheres A, Margulies DS, Kelly AM, Shehzad Z, Adelstein JS, Castellanos FX, Biswal BB, Milham MP (2008) Residual functional connectivity in the split-brain revealed with resting-state functional MRI. NeuroReport 19(7):703–709. https://doi.org/10.1097/WNR.0b013e3282fb8203
Uddin LQ, Supekar KS, Ryali S, Menon V (2011) Dynamic reconfiguration of structural and functional connectivity across core neurocognitive brain networks with development. J Neurosci 31(50):18578–18589. https://doi.org/10.1523/JNEUROSCI.4465-11.2011
Vias C, Dick AS (2017) Cerebellar contributions to language in typical and atypical development: a review. Dev Neuropsychol 42(6):404–421. https://doi.org/10.1080/87565641.2017.1334783
Wedeen VJ, Wang RP, Schmahmann JD, Benner T, Tseng WY, Dai G, Pandya DN, Hagmann P, D’Arceuil H, de Crespigny AJ (2008) Diffusion spectrum magnetic resonance imaging (DSI) tractography of crossing fibers. NeuroImage 41(4):1267–1277. https://doi.org/10.1016/j.neuroimage.2008.03.036
Yeh FC, Verstynen TD, Wang Y, Fernandez-Miranda JC, Tseng WY (2013) Deterministic diffusion fiber tracking improved by quantitative anisotropy. PLoS One 8(11):e80713. https://doi.org/10.1371/journal.pone.0080713
Acknowledgements
The authors thank Anouk Scheres and Scott Squire for assistance with data collection.
Funding
This work was supported by a University of Miami Gabelli Senior Scholar Award, an award from the Canadian Institute for Advanced Research, and National Institute of Mental Health [R01MH107549] to LQU. We also acknowledge the Nathan Kline Institute Enhanced Rockland Sample for the availability of age-matched comparison participants, collected with support from R01MH094639.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This article does not contain any studies with animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nomi, J.S., Marshall, E., Zaidel, E. et al. Diffusion weighted imaging evidence of extra-callosal pathways for interhemispheric communication after complete commissurotomy. Brain Struct Funct 224, 1897–1909 (2019). https://doi.org/10.1007/s00429-019-01864-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-019-01864-2