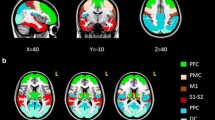
Abstract
Characterization of the microstructural properties and topography of the human corpus callosum (CC) is key to understanding interhemispheric neural communication and brain function. In this work, we tested the hypothesis that high-resolution T1 relaxometry at high field has adequate sensitivity and specificity for characterizing microstructural properties of the human CC, and elucidating the structural connectivity of the callosal fibers to the cortices of origin. The high-resolution parametric T1 images acquired from healthy subjects (N = 16) at 7 T clearly showed a consistent T1 distribution among individuals with substantial variation along the human CC axis, which is highly similar to the spatial patterns of myelin density and myelinated axon size based on the histology study. Compared to the anterior part of the CC, the posterior midbody and splenium had significantly higher T1 values. In conjunction with T1-based classification method, the splenial T1 values were decoded more reliably compared to a conventional partitioning method, showing a much higher T1 value in the inferior splenium than in the middle/superior splenium. Moreover, the T1 profile of the callosal subdivision represented the topology of the fiber connectivity to the projected cortical regions: the fibers in the posterior midbody and inferior splenium with a higher T1 (inferring a larger axon size) were mainly connected to motor–sensory and visual cortical areas, respectively; in contrast, the fibers in the anterior/posterior CC with a lower T1 (inferring a smaller axon size) were primarily connected to the frontal/parietal–temporal areas. These findings indicate that high-resolution T1 relaxometry imaging could provide a complementary and robust neuroimaging tool, useful for exploring the complex tissue properties and topographic organization of the human corpus callosum.




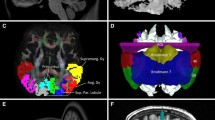
e, f Reprinted with the permission from the reference (Aboitiz et al. 1992a)

d Reprinted with the permission from the reference (Aboitiz and Montiel 2003)


Similar content being viewed by others
References
Aboitiz F, Montiel J (2003) One hundred million years of interhemispheric communication: the history of the corpus callosum. Braz J Med Biol Res 36 (4):409–420
Aboitiz F, Scheibel AB, Fisher RS, Zaidel E (1992a) Fiber composition of the human corpus callosum. Brain Res 598(1–2):143–153
Aboitiz F, Scheibel AB, Fisher RS, Zaidel E (1992b) Individual differences in brain asymmetries and fiber composition in the human corpus callosum. Brain Res 598(1–2):154–161
Alexander DC, Hubbard PL, Hall MG, Moore EA, Ptito M, Parker GJ, Dyrby TB (2010) Orientationally invariant indices of axon diameter and density from diffusion MRI. Neuroimage 52(4):1374–1389. https://doi.org/10.1016/j.neuroimage.2010.05.043
Andersson JL, Skare S, Ashburner J (2003) How to correct susceptibility distortions in spin-echo echo-planar images: application to diffusion tensor imaging. Neuroimage 20(2):870–888. https://doi.org/10.1016/S1053-8119(03)00336-7
Assaf Y, Blumenfeld-Katzir T, Yovel Y, Basser PJ (2008) AxCaliber: a method for measuring axon diameter distribution from diffusion MRI. Magn Reson Med 59(6):1347–1354. https://doi.org/10.1002/mrm.21577
Assaf Y, Alexander DC, Jones DK, Bizzi A, Behrens TE, Clark CA, Cohen Y, Dyrby TB, Huppi PS, Knoesche TR, Lebihan D, Parker GJ, Poupon C, consortium C, Anaby D, Anwander A, Bar L, Barazany D, Blumenfeld-Katzir T, De-Santis S, Duclap D, Figini M, Fischi E, Guevara P, Hubbard P, Hofstetter S, Jbabdi S, Kunz N, Lazeyras F, Lebois A, Liptrot MG, Lundell H, Mangin JF, Dominguez DM, Morozov D, Schreiber J, Seunarine K, Nava S, Poupon C, Riffert T, Sasson E, Schmitt B, Shemesh N, Sotiropoulos SN, Tavor I, Zhang HG, Zhou FL (2013) The CONNECT project: combining macro- and micro-structure. Neuroimage 80:273–282. https://doi.org/10.1016/j.neuroimage.2013.05.055
Behrens TE, Berg HJ, Jbabdi S, Rushworth MF, Woolrich MW (2007) Probabilistic diffusion tractography with multiple fibre orientations: what can we gain? Neuroimage 34(1):144–155. https://doi.org/10.1016/j.neuroimage.2006.09.018
Berlucchi G (1972) Anatomical and physiological aspects of visual functions of corpus callosum. Brain Res 37(2):371–392
Björnholm L, Nikkinen J, Kiviniemi V, Nordström T, Niemelä S, Drakesmith M, Evans JC, Pike GB, Veijola J, Paus T (2017) Structural properties of the human corpus callosum: multimodal assessment and sex differences. Neuroimage 152:108–118. https://doi.org/10.1016/j.neuroimage.2017.02.056
Budd JM, Kisvarday ZF (2012) Communication and wiring in the cortical connectome. Front Neuroanat 6:42. https://doi.org/10.3389/fnana.2012.00042
Callaghan PT, Eccles CD, Xia Y (1988) NMR microscopy of dynamic displacements: k-space and q-space imaging. J Phys E Sci Instrum 21(8):820
Caminiti R, Ghaziri H, Galuske R, Hof PR, Innocenti GM (2009) Evolution amplified processing with temporally dispersed slow neuronal connectivity in primates. Proc Natl Acad Sci USA 106(46):19551–19556. https://doi.org/10.1073/pnas.0907655106
Caminiti R, Carducci F, Piervincenzi C, Battaglia-Mayer A, Confalone G, Visco-Comandini F, Pantano P, Innocenti GM (2013) Diameter, length, speed, and conduction delay of callosal axons in macaque monkeys and humans: comparing data from histology and magnetic resonance imaging diffusion tractography. J Neurosci 33(36):14501–14511. https://doi.org/10.1523/JNEUROSCI.0761-13.2013
Chenevert TL, Brunberg JA, Pipe JG (1990) Anisotropic diffusion in human white matter: demonstration with MR techniques in vivo. Radiology 177(2):401–405. https://doi.org/10.1148/radiology.177.2.2217776
de Lacoste MC, Kirkpatrick JB, Ross ED (1985) Topography of the human corpus callosum. J Neuropathol Exp Neurol 44(6):578–591
Dougherty RF, Ben-Shachar M, Bammer R, Brewer AA, Wandell BA (2005a) Functional organization of human occipital-callosal fiber tracts. Proc Natl Acad Sci USA 102(20):7350–7355. https://doi.org/10.1073/pnas.0500003102
Dougherty RF, Ben-Shachar M, Deutsch G, Potanina P, Bammer R, Wandell BA (2005b) Occipital–callosal pathways in children: validation and atlas development. Ann N Y Acad Sci 1064:98–112. https://doi.org/10.1196/annals.1340.017
Fabri M, Pierpaoli C, Barbaresi P, Polonara G (2014) Functional topography of the corpus callosum investigated by DTI and fMRI. World J Radiol 6(12):895–906. https://doi.org/10.4329/wjr.v6.i12.895
Ford MC, Alexandrova O, Cossell L, Stange-Marten A, Sinclair J, Kopp-Scheinpflug C, Pecka M, Attwell D, Grothe B (2015) Tuning of Ranvier node and internode properties in myelinated axons to adjust action potential timing. Nat Commun 6:8073. https://doi.org/10.1038/ncomms9073
Garwood M, DelaBarre L (2001) The return of the frequency sweep: designing adiabatic pulses for contemporary NMR. J Magn Reson 153(2):155–177. https://doi.org/10.1006/jmre.2001.2340
Gelman N, Gorell JM, Barker PB, Savage RM, Spickler EM, Windham JP, Knight RA (1999) MR imaging of human brain at 3.0 T: preliminary report on transverse relaxation rates and relation to estimated iron content. Radiology 210(3):759–767. https://doi.org/10.1148/radiology.210.3.r99fe41759
Gelman N, Ewing JR, Gorell JM, Spickler EM, Solomon EG (2001) Interregional variation of longitudinal relaxation rates in human brain at 3.0 T: relation to estimated iron and water contents. Magn Reson Med 45(1):71–79
Genc S, Malpas CB, Ball G, Silk TJ, Seal ML (2018) Age, sex, and puberty related development of the corpus callosum: a multi-technique diffusion MRI study. Brain Struct Funct 223(6):2753–2765. https://doi.org/10.1007/s00429-018-1658-5
Good CD, Johnsrude IS, Ashburner J, Henson RN, Friston KJ, Frackowiak RS (2001) A voxel-based morphometric study of ageing in 465 normal adult human brains. Neuroimage 14(1 Pt 1):21–36
Haacke EM, Cheng NY, House MJ, Liu Q, Neelavalli J, Ogg RJ, Khan A, Ayaz M, Kirsch W, Obenaus A (2005) Imaging iron stores in the brain using magnetic resonance imaging. Magn Reson Imaging 23(1):1–25. https://doi.org/10.1016/j.mri.2004.10.001
Hallgren B, Sourander P (1958) The effect of age on the non-haemin iron in the human brain. J Neurochem 3(1):41–51. doi:https://doi.org/10.1111/j.1471-4159.1958.tb12607.x
Hargreaves BA, Cunningham CH, Nishimura DG, Conolly SM (2004) Variable-rate selective excitation for rapid MRI sequences. Magn Reson Med 52(3):590–597. https://doi.org/10.1002/mrm.20168
Harkins KD, Xu J, Dula AN, Li K, Valentine WM, Gochberg DF, Gore JC, Does MD (2016) The microstructural correlates of T 1 in white matter. Magn Reson Med 75(3):1341–1345. https://doi.org/10.1002/mrm.25709
Harkins Kevin D, Xu J, Dula Adrienne N, Li K, Valentine William M, Gochberg Daniel F, Gore John C, Does Mark D (2016) The microstructural correlates of T 1 in white matter. Magn Reson Med 75(3):1341–1345. https://doi.org/10.1002/mrm.25709 doi
Hofer S, Frahm J (2006) Topography of the human corpus callosum revisited—comprehensive fiber tractography using diffusion tensor magnetic resonance imaging. Neuroimage 32(3):989–994. https://doi.org/10.1016/j.neuroimage.2006.05.044
Hofer S, Wang X, Roeloffs V, Frahm J (2015) Single-shot T 1 mapping of the corpus callosum: a rapid characterization of fiber bundle anatomy. Front Neuroanat 9:57. https://doi.org/10.3389/fnana.2015.00057
Huang H, Zhang J, Jiang H, Wakana S, Poetscher L, Miller MI, van Zijl PC, Hillis AE, Wytik R, Mori S (2005) DTI tractography based parcellation of white matter: application to the mid-sagittal morphology of corpus callosum. Neuroimage 26(1):195–205. https://doi.org/10.1016/j.neuroimage.2005.01.019
Innocenti GM, Caminiti R, Hof PR (2010) Fiber composition in the planum temporale sector of the corpus callosum in chimpanzee and human. Brain Struct Funct 215(2):123–128. https://doi.org/10.1007/s00429-010-0274-9
Jensen JH, Helpern JA, Ramani A, Lu H, Kaczynski K (2005) Diffusional kurtosis imaging: the quantification of non-Gaussian water diffusion by means of magnetic resonance imaging. Magn Reson Med 53(6):1432–1440. https://doi.org/10.1002/mrm.20508
Kontis D, Catani M, Cuddy M, Walshe M, Nosarti C, Jones D, Wyatt J, Rifkin L, Murray R, Allin M (2009) Diffusion tensor MRI of the corpus callosum and cognitive function in adults born preterm. Neuroreport 20(4):424–428. https://doi.org/10.1097/WNR.0b013e328325a8f9
Lamantia AS, Rakic P (1990) Cytological and quantitative characteristics of four cerebral commissures in the rhesus monkey. J Comp Neurol 291(4):520–537. https://doi.org/10.1002/cne.902910404
Langkammer C, Krebs N, Goessler W, Scheurer E, Ebner F, Yen K, Fazekas F, Ropele S (2010) Quantitative MR imaging of brain iron: a postmortem validation study. Radiology 257(2):455–462. https://doi.org/10.1148/radiol.10100495
Le Bihan D, Breton E, Lallemand D, Grenier P, Cabanis E, Laval-Jeantet M (1986) MR imaging of intravoxel incoherent motions: application to diffusion and perfusion in neurologic disorders. Radiology 161(2):401–407. https://doi.org/10.1148/radiology.161.2.3763909
Lee B-Y, Zhu X-H, Li X, Chen W (2014) Quantitative assessment of microstructure properties of human corpus callosum using parametric T 1 and Myelin imaging. Proc Intl Soc Mag Reson Med 2014:32–36
Li X, Bolan PJ, Ugurbil K, Metzger GJ (2015) Measuring renal tissue relaxation times at 7 T. NMR Biomed 28(1):63–69. https://doi.org/10.1002/nbm.3195
Liewald D, Miller R, Logothetis N, Wagner HJ, Schuz A (2014) Distribution of axon diameters in cortical white matter: an electron-microscopic study on three human brains and a macaque. Biol Cybern 108(5):541–557. https://doi.org/10.1007/s00422-014-0626-2
Moeller S, Yacoub E, Olman CA, Auerbach E, Strupp J, Harel N, Uğurbil K (2010) Multiband multislice GE-EPI at 7 T, with 16-fold acceleration using partial parallel imaging with application to high spatial and temporal whole-brain fMRI. Magn Reson Med 63(5):1144–1153. https://doi.org/10.1002/mrm.22361
Myers RE (1959) Localization of function in the corpus callosum. Visual gnostic transfer. Arch Neurol 1:74–77
Narr KL, Thompson PM, Sharma T, Moussai J, Cannestra AF, Toga AW (2000) Mapping morphology of the corpus callosum in schizophrenia. Cereb Cortex 10(1):40–49
Park HJ, Kim JJ, Lee SK, Seok JH, Chun J, Kim DI, Lee JD (2008) Corpus callosal connection mapping using cortical gray matter parcellation and DT-MRI. Hum Brain Mapp 29(5):503–516. https://doi.org/10.1002/hbm.20314
Payne BR (1990) Function of the corpus callosum in the representation of the visual field in cat visual cortex. Vis Neurosci 5(2):205–211
Putnam MC, Steven MS, Doron KW, Riggall AC, Gazzaniga MS (2010) Cortical projection topography of the human splenium: hemispheric asymmetry and individual differences. J Cogn Neurosci 22(8):1662–1669. https://doi.org/10.1162/jocn.2009.21290
Rooney WD, Johnson G, Li X, Cohen ER, Kim SG, Ugurbil K, Springer CS Jr (2007) Magnetic field and tissue dependencies of human brain longitudinal 1H2O relaxation in vivo. Magn Reson Med 57(2):308–318. https://doi.org/10.1002/mrm.21122
Saenz M, Fine I (2010) Topographic organization of V1 projections through the corpus callosum in humans. Neuroimage 52(4):1224–1229. https://doi.org/10.1016/j.neuroimage.2010.05.060
Sereno MI, Lutti A, Weiskopf N, Dick F (2013) Mapping the human cortical surface by combining quantitative T(1) with retinotopy. Cereb Cortex 23(9):2261–2268. https://doi.org/10.1093/cercor/bhs213
Sigalovsky IS, Fischl B, Melcher JR (2006) Mapping an intrinsic MR property of gray matter in auditory cortex of living humans: a possible marker for primary cortex and hemispheric differences. Neuroimage 32(4):1524–1537. https://doi.org/10.1016/j.neuroimage.2006.05.023
Sisti HM, Geurts M, Gooijers J, Heitger MH, Caeyenberghs K, Beets IA, Serbruyns L, Leemans A, Swinnen SP (2012) Microstructural organization of corpus callosum projections to prefrontal cortex predicts bimanual motor learning. Learn Mem 19(8):351–357. https://doi.org/10.1101/lm.026534.112
Sotiropoulos SN, Jbabdi S, Xu J, Andersson JL, Moeller S, Auerbach EJ, Glasser MF, Hernandez M, Sapiro G, Jenkinson M, Feinberg DA, Yacoub E, Lenglet C, Van Essen DC, Ugurbil K, Behrens TE, Consortium WU-MH (2013) Advances in diffusion MRI acquisition and processing in the Human Connectome Project. Neuroimage 80:125–143. https://doi.org/10.1016/j.neuroimage.2013.05.057
Stikov N, Perry LM, Mezer A, Rykhlevskaia E, Wandell BA, Pauly JM, Dougherty RF (2011) Bound pool fractions complement diffusion measures to describe white matter micro and macrostructure. Neuroimage 54(2):1112–1121. https://doi.org/10.1016/j.neuroimage.2010.08.068
Stikov N, Campbell JS, Stroh T, Lavelee M, Frey S, Novek J, Nuara S, Ho MK, Bedell BJ, Dougherty RF, Leppert IR, Boudreau M, Narayanan S, Duval T, Cohen-Adad J, Picard PA, Gasecka A, Cote D, Pike GB (2015a) In vivo histology of the myelin g-ratio with magnetic resonance imaging. Neuroimage 118:397–405. https://doi.org/10.1016/j.neuroimage.2015.05.023
Stikov N, Campbell JS, Stroh T, Lavelee M, Frey S, Novek J, Nuara S, Ho MK, Bedell BJ, Dougherty RF, Leppert IR, Boudreau M, Narayanan S, Duval T, Cohen-Adad J, Picard PA, Gasecka A, Cote D, Pike GB (2015b) Quantitative analysis of the myelin g-ratio from electron microscopy images of the macaque corpus callosum. Data Brief 4:368–373. https://doi.org/10.1016/j.dib.2015.05.019
Stuber C, Morawski M, Schafer A, Labadie C, Wahnert M, Leuze C, Streicher M, Barapatre N, Reimann K, Geyer S, Spemann D, Turner R (2014) Myelin and iron concentration in the human brain: a quantitative study of MRI contrast. Neuroimage 93 Pt 1:95–106. https://doi.org/10.1016/j.neuroimage.2014.02.026
Thapaliya K, Vegh V, Bollmann S, Barth M (2017) Assessment of microstructural signal compartments across the corpus callosum using multi-echo gradient recalled echo at 7 T. Neuroimage. https://doi.org/10.1016/j.neuroimage.2017.11.029
Tomasi S, Caminiti R, Innocenti GM (2012) Areal differences in diameter and length of corticofugal projections. Cereb Cortex 22(6):1463–1472. https://doi.org/10.1093/cercor/bhs011
Tuch DS, Reese TG, Wiegell MR, Makris N, Belliveau JW, Wedeen VJ (2002) High angular resolution diffusion imaging reveals intravoxel white matter fiber heterogeneity. Magn Reson Med 48(4):577–582. https://doi.org/10.1002/mrm.10268
Ugurbil K, Xu J, Auerbach EJ, Moeller S, Vu AT, Duarte-Carvajalino JM, Lenglet C, Wu X, Schmitter S, Van de Moortele PF, Strupp J, Sapiro G, De Martino F, Wang D, Harel N, Garwood M, Chen L, Feinberg DA, Smith SM, Miller KL, Sotiropoulos SN, Jbabdi S, Andersson JL, Behrens TE, Glasser MF, Van Essen DC, Yacoub E, Consortium WU-MH (2013) Pushing spatial and temporal resolution for functional and diffusion MRI in the Human Connectome Project. Neuroimage 80:80–104. https://doi.org/10.1016/j.neuroimage.2013.05.012
Vu AT, Auerbach E, Lenglet C, Moeller S, Sotiropoulos SN, Jbabdi S, Andersson J, Yacoub E, Ugurbil K (2015) High resolution whole brain diffusion imaging at 7T for the Human Connectome Project. Neuroimage 122:318–331. https://doi.org/10.1016/j.neuroimage.2015.08.004
Wahl M, Lauterbach-Soon B, Hattingen E, Jung P, Singer O, Volz S, Klein JC, Steinmetz H, Ziemann U (2007) Human motor corpus callosum: topography, somatotopy, and link between microstructure and function. J Neurosci 27(45):12132–12138. https://doi.org/10.1523/JNEUROSCI.2320-07.2007
Wang X, Zhu XH, Zhang Y, Chen W (2016) In vivo parametric T 1/R 1 imaging correlation with myelin density and microstructure properties of rat corpus callosum. In: Proceedings of international society for magnetic resonance in medicine, p 1465
Waxman SG, Bennett MV (1972) Relative conduction velocities of small myelinated and non-myelinated fibres in the central nervous system. Nat New Biol 238(85):217–219
Witelson SF (1989) Hand and sex differences in the isthmus and genu of the human corpus callosum. A postmortem morphological study. Brain 112(Pt 3):799–835
Wright PJ, Mougin OE, Totman JJ, Peters AM, Brookes MJ, Coxon R, Morris PE, Clemence M, Francis ST, Bowtell RW, Gowland PA (2008) Water proton T 1 measurements in brain tissue at 7, 3, and 1.5 T using IR-EPI, IR-TSE, and MPRAGE: results and optimization. MAGMA 21(1–2):121–130. https://doi.org/10.1007/s10334-008-0104-8
Yarnykh VL (2007) Actual flip-angle imaging in the pulsed steady state: a method for rapid three-dimensional mapping of the transmitted radiofrequency field. Magn Reson Med 57(1):192–200. https://doi.org/10.1002/mrm.21120
Zhang H, Hubbard PL, Parker GJ, Alexander DC (2011) Axon diameter mapping in the presence of orientation dispersion with diffusion MRI. Neuroimage 56(3):1301–1315. https://doi.org/10.1016/j.neuroimage.2011.01.084
Acknowledgements
This work was supported by NIH Grants RO1 NS070839 and MH111413; R24 MH106049, S10 RR026783, U01 EB026978, P41 EB015894, and P30NS076408 and the W.M. Keck Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval
All procedures performed in the studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Lee, BY., Zhu, XH., Li, X. et al. High-resolution imaging of distinct human corpus callosum microstructure and topography of structural connectivity to cortices at high field. Brain Struct Funct 224, 949–960 (2019). https://doi.org/10.1007/s00429-018-1804-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-018-1804-0