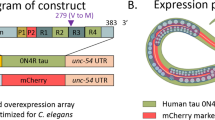
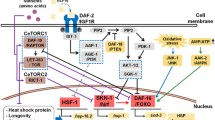
Abstract
Alzheimer’s disease (AD) is the major cause of dementia in the United States. At the cellular level, the brains of AD patients are characterized by extracellular dense plaques and intracellular neurofibrillary tangles whose major components are the β-amyloid peptide and tau, respectively. The β-amyloid peptide is a cleavage product of the amyloid precursor protein (APP); mutations in APP have been correlated with a small number of cases of familial Alzheimer’s disease. APP is the canonical member of the APP family, whose functions remain unclear. The nematode Caenorhabditis elegans, one of the premier genetic workhorses, is being used in a variety of ways to address the functions of APP and determine how the β-amyloid peptide and tau can induce toxicity. First, the function of the C. elegans APP-related gene, apl-1, is being examined. Although different organisms may use APP and related proteins, such as APL-1, in different functional contexts, the pathways in which they function and the molecules with which they interact are usually conserved. Second, components of the γ-secretase complex and their respective functions are being revealed through genetic analyses in C. elegans. Third, to address questions of toxicity, onset of degeneration, and protective mechanisms, different human β-amyloid peptide and tau variants are being introduced into C. elegans and the resultant transgenic lines examined. Here, we summarize how a simple system such as C. elegans can be used as a model to understand APP function and suppression of β-amyloid peptide and tau toxicity in higher organisms.


Similar content being viewed by others
References
Abbott AL, Alvarez-Saavedra E, Miska EA, Lau NC, Bartel DP, Horvitz HR et al (2005) The let-7 MicroRNA family members mir-48, mir-84, and mir-241 function together to regulate developmental timing in Caenorhabditis elegans. Dev Cell 9(3):403–414. doi:S1534-5807(05)00289-3
Abrahante JE, Daul AL, Li M, Volk ML, Tennessen JM, Miller EA et al (2003) The Caenorhabditis elegans hunchback-like gene lin-57/hbl-1 controls developmental time and is regulated by microRNAs. Dev Cell 4(5):625–637. doi:S1534580703001278
Arduengo PM, Appleberry OK, Chuang P, L’Hernault SW (1998) The presenilin protein family member SPE-4 localizes to an ER/Golgi derived organelle and is required for proper cytoplasmic partitioning during Caenorhabditis elegans spermatogenesis. J Cell Sci 111:3645–3654
Arya U, Dwivedi H, Subramaniam JR (2009) Reserpine ameliorates Abeta toxicity in the Alzheimer’s disease model in Caenorhabditis elegans. Exp Gerontol 44:462–466
Association (2008) 2008 Alzheimer’s disease facts and figures. Alzheimers Dement 4(2):110–133
Barnes NY, Li L, Yoshikawa K, Schwartz LM, Oppenheim RW, Milligan CE (1998) Increased production of amyloid precursor protein provides a substrate for caspase-3 in dying motoneurons. J Neurosci 18(15):5869–5880
Barsyte D, Lovejoy DA, Lithgow GJ (2001) Longevity and heavy metal resistance in daf-2 and age-1 long-lived mutants of Caenorhabditis elegans. FASEB J 15(3):627–634. doi:10.1096/fj.99-0966com
Bleuler M, Stoll WA (1955) Clinical use of reserpine in psychiatry: comparison with chlorpromazine. Ann N Y Acad Sci 61(1):167–173
Brandt R, Gergou A, Wacker I, Fath T, Hutter H (2009) A Caenorhabditis elegans model of tau hyperphosphorylation: induction of developmental defects by transgenic overexpression of Alzheimer’s disease-like modified tau. Neurobiol Aging 30:22–33
Brion JP, Hanger DP, Bruce MT, Couck AM, Flament-Durand J, Anderton BH (1991) Tau in Alzheimer neurofibrillary tangles. N- and C-terminal regions are differentially associated with paired helical filaments and the location of a putative abnormal phosphorylation site. Biochem J 273(Pt 1):127–133
Brookmeyer R, Johnson E, Ziegler-Graham K, Arrighi HM (2007) Forecasting the global burden of Alzheimer’s disease. Alzheimer’s Dement 3(3):186–191
Byerly L, Cassada RC, Russell RL (1976) The life cycle of the nematode Caenorhabditis elegans. I. Wild-type growth and reproduction. Dev Biol 51(1):23–33. doi:0012-1606(76)90119-6
Cabrejo L, Guyant-Marechal L, Laquerriere A, Vercelletto M, De la Fourniere F, Thomas-Anterion C et al (2006) Phenotype associated with APP duplication in five families. Brain 129(Pt 11):2966–2976
Campion D, Dumanchin C, Hannequin D, Dubois B, Belliard S, Puel M et al (1999) Early-onset autosomal dominant Alzheimer disease: prevalence, genetic heterogeneity, and mutation spectrum. Am J Hum Genet 65(3):664–670. doi:S0002-9297(07)62317-9
Carmine-Simmen K, Proctor T, Tschape J, Poeck B, Triphan T, Strauss R et al (2009) Neurotoxic effects induced by the Drosophila amyloid-beta peptide suggest a conserved toxic function. Neurobiol Dis 33(2):274–281. doi:S0969-9961(08)00263-5
Carroll PM, Fitzgerald K (2003) Model organisms in drug discovery. Wiley, New York
Chartier-Harlin MC, Crawford F, Houlden H, Warren A, Hughes D, Fidani L et al (1991) Early-onset Alzheimer’s disease caused by mutations at codon 717 of the beta-amyloid precursor protein gene. Nature 353(6347):844–846
Chen YZ (2004) APP induces neuronal apoptosis through APP-BP1-mediated downregulation of beta-catenin. Apoptosis 9(4):415–422. doi:10.1023/B:APPT.0000031447.05354.9f
Chui DH, Tanahashi H, Ozawa K, Ikeda S, Checler F, Ueda O et al (1999) Transgenic mice with Alzheimer presenilin 1 mutations show accelerated neurodegeneration without amyloid plaque formation. Nat Med 5(5):560–564 10.1038/8438
Cinar HN, Sweet KL, Hosemann KE, Earley K, Newman AP (2001) The SEL-12 presenilin mediates induction of the Caenorhabditis elegans uterine pi cell fate. Dev Biol 237:173–182
Cohen E, Bieschke J, Perciavalle RM, Kelly JW, Dillin A (2006) Opposing activities protect against age-onset proteotoxicity. Science 313:1604–1610
Curtis-Prior P, Vere D, Fray P (1999) Therapeutic value of Ginkgo biloba in reducing symptoms of decline in mental function. J Pharm Pharmacol 51(5):535–541
Daigle I, Li C (1993) apl-1, a Caenorhabditis elegans gene encoding a protein related to the human beta-amyloid protein precursor. Proc Natl Acad Sci USA 90(24):12045–12049
Davies L, Wolska B, Hilbich C, Multhaup G, Martins R, Simms G et al (1988) A4 amyloid protein deposition and the diagnosis of Alzheimer’s disease: prevalence in aged brains determined by immunocytochemistry compared with conventional neuropathologic techniques. Neurology 38(11):1688–1693
De Strooper B, Annaert W, Cupers P, Saftig P, Craessaerts K, Mumm JS et al (1999) A presenilin-1-dependent gamma-secretase-like protease mediates release of Notch intracellular domain. Nature 398(6727):518–522 10.1038/19083
Delacourte A, Defossez A (1986) Alzheimer’s disease: tau proteins, the promoting factors of microtubule assembly, are major components of paired helical filaments. J Neurol Sci 76(2–3):173–186
Dernburg AF, Zalevsky J, Colaiacovo MP, Villeneuve AM (2000) Transgene-mediated cosuppression in the C. elegans germ line. Genes Dev 14(13):1578–1583
Drake J, Link CD, Butterfield DA (2003) Oxidative stress precedes fibrillar deposition of Alzheimer’s disease amyloid beta-peptide (1–42) in a transgenic Caenorhabditis elegans model. Neurobiol Aging 24(3):415–420
Eimer S (2003) Analysis and suppression of mutant presenilin sel-12 in Caenorhabditis elegans. Dissertation, Ludwig-Maximilians-Universitaet Muenchen, Muenchen
Ermekova KS, Zambrano N, Linn H, Minopoli G, Gertler F, Russo T et al (1997) The WW domain of neural protein FE65 interacts with proline-rich motifs in Mena, the mammalian homolog of Drosophila enabled. J Biol Chem 272(52):32869–32877
Fay DS, Fluet A, Johnson CJ, Link CD (1998) In vivo aggregation of beta-amyloid peptide variants. J Neurochem 71(4):1616–1625
Fay DS, Stanley HM, Han M, Wood WB (1999) A Caenorhabditis elegans homologue of hunchback is required for late stages of development but not early embryonic patterning. Dev Biol 205(2):240–253. doi:S0012-1606(98)99096-0
Florez-McClure ML, Hohsfield LA, Fonte G, Bealor MT, Link CD (2007) Decreased insulin-receptor signaling promotes the autophagic degradation of beta-amyloid peptide in C. elegans. Autophagy 3:569–580
Fonte V, Kapulkin V, Taft A, Fluet A, Friedman D, Link CD (2002) Interaction of intracellular beta amyloid peptide with chaperone proteins. Proc Natl Acad Sci USA 99:9439–9444
Fonte V, Kipp DR, Yerg J 3rd, Merin D, Forrestal M, Wagner E et al (2008) Suppression of in vivo beta-amyloid peptide toxicity by overexpression of the HSP-16.2 small chaperone protein. J Biol Chem 283(2):784–791
Francis R, McGrath G, Zhang J, Ruddy DA, Sym M, Apfeld J et al (2002) aph-1 and pen-2 are required for Notch pathway signaling, gamma-secretase cleavage of betaAPP, and presenilin protein accumulation. Dev Cell 3:85–97
Galvan V, Zhang J, Gorostiza OF, Banwait S, Huang W, Ataie M et al (2008) Long-term prevention of Alzheimer’s disease-like behavioral deficits in PDAPP mice carrying a mutation in Asp664. Behav Brain Res 191(2):246–255. doi:S0166-4328(08)00180-0
Gervais FG, Xu D, Robertson GS, Vaillancourt JP, Zhu Y, Huang J et al (1999) Involvement of caspases in proteolytic cleavage of Alzheimer’s amyloid-beta precursor protein and amyloidogenic A beta peptide formation. Cell 97(3):395–406. doi:S0092-8674(00)80748-5
Glenner GG, Wong CW (1984) Alzheimer’s disease and Down’s syndrome: sharing of a unique cerebrovascular amyloid fibril protein. Biochem Biophys Res Commun 122(3):1131–1135
Goate A, Chartier-Harlin MC, Mullan M, Brown J, Crawford F, Fidani L et al (1991) Segregation of a missense mutation in the amyloid precursor protein gene with familial Alzheimer’s disease. Nature 349(6311):704–706
Goedert M, Spillantini MG, Jakes R, Rutherford D, Crowther RA (1989) Multiple isoforms of human microtubule-associated protein tau: sequences and localization in neurofibrillary tangles of Alzheimer’s disease. Neuron 3(4):519–526
Goedert M, Baur CP, Ahringer J, Jakes R, Hasegawa M, Spillantini MG et al (1996) PTL-1, a microtubule-associated protein with tau-like repeats from the nematode Caenorhabditis elegans. J Cell Sci 109:2661–2672
Gordon P, Hingula L, Krasny ML, Swienckowski JL, Pokrywka NJ, Raley-Susman KM (2008) The invertebrate microtubule-associated protein PTL-1 functions in mechanosensation and development in Caenorhabditis elegans. Dev Genes Evol 218(10):541–551
Gouras GK, Tsai J, Naslund J, Vincent B, Edgar M, Checler F et al (2000) Intraneuronal Abeta42 accumulation in human brain. Am J Pathol 156(1):15–20
Goutte C, Hepler W, Mickey KM, Priess JR (2000) aph-2 encodes a novel extracellular protein required for GLP-1-mediated signaling. Development 127(11):2481–2492
Goutte C, Tsunozaki M, Hale VA, Priess JR (2002) APH-1 is a multipass membrane protein essential for the Notch signaling pathway in Caenorhabditis elegans embryos. Proc Natl Acad Sci USA 99:775–779
Gralle M, Ferreira ST (2007) Structure and functions of the human amyloid precursor protein: the whole is more than the sum of its parts. Prog Neurobiol 82(1):11–32. doi:S0301-0082(07)00016-0
Greenwald I (2005) LIN-12/Notch signaling in C. elegans. WormBook, ed. The C. elegans Research Community, WormBook. doi:10.1895/wormbook.1.10.1, http://www.wormbook.org
Greenwald IS, Sternberg PW, Horvitz HR (1983) The lin-12 locus specifies cell fates in Caenorhabditis elegans. Cell 34(2):435–444. doi:0092-8674(83)90377-X
Grundke-Iqbal I, Iqbal K, Tung YC, Quinlan M, Wisniewski HM, Binder LI (1986) Abnormal phosphorylation of the microtubule-associated protein tau (tau) in Alzheimer cytoskeletal pathology. Proc Natl Acad Sci USA 83(13):4913–4917
Gu Y, Chen F, Sanjo N, Kawarai T, Hasegawa H, Duthie M et al (2003) APH-1 interacts with mature and immature forms of presenilins and nicastrin and may play a role in maturation of presenilin/nicastrin complexes. J Biol Chem 278:7374–7380
Guthrie CR, Schellenberg GD, Kraemer BC (2009) SUT-2 potentiates tau-induced neurotoxicity in Caenorhabditis elegans. Hum Mol Genet 18(10):1825–1838
Gutierrez-Zepeda A, Santell R, Wu Z, Brown M, Wu Y, Khan I et al (2005) Soy isoflavone glycitein protects against beta amyloid-induced toxicity and oxidative stress in transgenic Caenorhabditis elegans. BMC Neurosci 6:54
Haass C, Selkoe DJ (1993) Cellular processing of beta-amyloid precursor protein and the genesis of amyloid beta-peptide. Cell 75(6):1039–1042
Hardy J (2009a) APP mutations table. http://www.alzforum.org/res/com/mut/app/table1.asp
Hardy J (2009b) Presenilin-1 mutations table. http://www.alzforum.org/res/com/mut/pre/table1.asp. Accessed 14 Apr 2009
Hardy J (2009c) Presenilin-2 mutations table. http://www.alzforum.org/res/com/mut/pre/table2.asp. Accessed 14 Apr 2009
Hardy J, Allsop D (1991) Amyloid deposition as the central event in the aetiology of Alzheimer’s disease. Trends Pharmacol Sci 12(10):383–388
Heber S, Herms J, Gajic V, Hainfellner J, Aguzzi A, Rulicke T et al (2000) Mice with combined gene knock-outs reveal essential and partially redundant functions of amyloid precursor protein family members. J Neurosci 20(21):7951–7963
Hebert LE, Scherr PA, Bienias JL, Bennett DA, Evans DA (2003) Alzheimer disease in the US population: prevalence estimates using the 2000 census. Arch Neurol 60(8):1119–1122
Hedgecock EM, Culotti JG, Hall DH (1990) The unc-5, unc-6, and unc-40 genes guide circumferential migrations of pioneer axons and mesodermal cells on the epidermis in C. elegans. Neuron 4(1):61–85. doi:0896-6273(90)90444-K
Henderson ST, Johnson TE (2001) daf-16 integrates developmental and environmental inputs to mediate aging in the nematode Caenorhabditis elegans. Curr Biol 11:1975–1980
Herms J, Anliker B, Heber S, Ring S, Fuhrmann M, Kretzschmar H et al (2004) Cortical dysplasia resembling human type 2 lissencephaly in mice lacking all three APP family members. EMBO J 23(20):4106–4115
Honda Y, Honda S (1999) The daf-2 gene network for longevity regulates oxidative stress resistance and Mn-superoxide dismutase gene expression in Caenorhabditis elegans. Faseb J 13(11):1385–1393
Hornsten A, Lieberthal J, Fadia S, Malins R, Ha L, Xu X et al (2007) APL-1, a Caenorhabditis elegans protein related to the human beta-amyloid precursor protein, is essential for viability. Proc Natl Acad Sci USA 104(6):1971–1976
Horvitz HR (1999) Genetic control of programmed cell death in the nematode Caenorhabditis elegans. Cancer Res 59(7 Suppl):1701s–1706s
Hsiao KK, Borchelt DR, Olson K, Johannsdottir R, Kitt C, Yunis W et al (1995) Age-related CNS disorder and early death in transgenic FVB/N mice overexpressing Alzheimer amyloid precursor proteins. Neuron 15:1203–1218
Hsu A-L, Murphy CT, Kenyon C (2003) Regulation of aging and age-related disease by DAF-16 and heat-shock factor. Science 300(5622):1142–1145. doi:10.1126/science.1083701
Hubbard EJ, Wu G, Kitajewski J, Greenwald I (1997) sel-10, a negative regulator of lin-12 activity in Caenorhabditis elegans, encodes a member of the CDC4 family of proteins. Genes Dev 11(23):3182–3193
Hulette CM, Pericak-Vance MA, Roses AD, Schmechel DE, Yamaoka LH, Gaskell PC et al (1999) Neuropathological features of frontotemporal dementia and Parkinsonism linked to chromosome 17q21-22 (FTDP-17): Duke family 1684. J Neuropathol Exp Neurol 58(8):859–866
Hunt-Newbury R, Viveiros R, Johnsen R, Mah A, Anastas D, Fang L et al (2007) High-throughput in vivo analysis of gene expression in Caenorhabditis elegans. PLoS Biol 5(9):e237. doi:07-PLBI-RA-0103
Jacobsen JS, Wu CC, Redwine JM, Comery TA, Arias R, Bowlby M et al (2006) Early-onset behavioral and synaptic deficits in a mouse model of Alzheimer’s disease. Proc Natl Acad Sci USA 103(13):5161–5166
Jarriault S, Greenwald I (2005) Evidence for functional redundancy between C. elegans ADAM proteins SUP-17/Kuzbanian and ADM-4/TACE. Dev Biol 287(1):1–10. doi:S0012-1606(05)00542-7
Jeon M, Gardner HF, Miller EA, Deshler J, Rougvie AE (1999) Similarity of the C. elegans developmental timing protein LIN-42 to circadian rhythm proteins. Science 286(5442):1141–1146
Kang J, Lemaire HG, Unterbeck A, Salbaum JM, Masters CL, Grzeschik KH et al (1987) The precursor of Alzheimer’s disease amyloid A4 protein resembles a cell-surface receptor. Nature 325(6106):733–736
Kelly SM, Pabit SA, Kitchen CM, Guo P, Marfatia KA, Murphy TJ et al (2007) Recognition of polyadenosine RNA by zinc finger proteins. Proc Natl Acad Sci USA 104(30):12306–12311
Kenyon C, Chang J, Gensch E, Rudner A, Tabtiang R (1993) A C. elegans mutant that lives twice as long as wild type. Nature 366(6454):461–464
Kidd M (1964) Alzheimer’s disease—an electron microscopical study. Brain 87:307–320
Kimberly WT, LaVoie MJ, Ostaszewski BL, Ye W, Wolfe MS, Selkoe DJ (2003) Gamma-secretase is a membrane protein complex comprised of presenilin, nicastrin, Aph-1, and Pen-2. Proc Natl Acad Sci USA 100(11):6382–6387. doi:10.1073/pnas.1037392100
Kokel M, Borland CZ, DeLong L, Horvitz HR, Stern MJ (1998) clr-1 encodes a receptor tyrosine phosphatase that negatively regulates an FGF receptor signaling pathway in Caenorhabditis elegans. Genes Dev 12(10):1425–1437
Kosik KS, Joachim CL, Selkoe DJ (1986) Microtubule-associated protein tau (tau) is a major antigenic component of paired helical filaments in Alzheimer disease. Proc Natl Acad Sci USA 83(11):4044–4048
Kraemer BC, Schellenberg GD (2007) SUT-1 enables tau-induced neurotoxicity in C. elegans. Hum Mol Genet 16:1959–1971
Kraemer BC, Zhang B, Leverenz JB, Thomas JH, Trojanowski JQ, Schellenberg GD (2003) Neurodegeneration and defective neurotransmission in a Caenorhabditis elegans model of tauopathy. Proc Natl Acad Sci USA 100:9980–9985
Krigman MR, Feldman RG, Bensch K (1965) Alzheimer’s presenile dementia. A histochemical and electron microscopic study. Lab Invest 14:381–396
Kumar MS, Erkeland SJ, Pester RE, Chen CY, Ebert MS, Sharp PA et al (2008) Suppression of non-small cell lung tumor development by the let-7 microRNA family. Proc Natl Acad Sci USA 105(10):3903–3908
Kuo YM, Beach TG, Sue LI, Scott S, Layne KJ, Kokjohn TA et al (2001) The evolution of A beta peptide burden in the APP23 transgenic mice: implications for A beta deposition in Alzheimer disease. Mol Med 7(9):609–618
Kwon J (2008) Tau mutations table. http://www.alzforum.org/res/com/mut/tau/table1.asp
L’Hernault SW, Arduengo PM (1992) Mutation of a putative sperm membrane protein in Caenorhabditis elegans prevents sperm differentiation but not its associated meiotic divisions. J Cell Biol 119:55–68
LaFerla FM, Tinkle BT, Bieberich CJ, Haudenschild CC, Jay G (1995) The Alzheimer’s A beta peptide induces neurodegeneration and apoptotic cell death in transgenic mice. Nat Genet 9(1):21–30
LaFerla FM, Troncoso JC, Strickland DK, Kawas CH, Jay G (1997) Neuronal cell death in Alzheimer’s disease correlates with apoE uptake and intracellular Abeta stabilization. J Clin Invest 100(2):310–320. doi:10.1172/JCI119536
Le Bars PL, Katz MM, Berman N, Itil TM, Freedman AM, Schatzberg AF (1997) A placebo-controlled, double-blind, randomized trial of an extract of Ginkgo biloba for dementia. North American EGb Study Group. Jama 278(16):1327–1332
Le Bars PL, Kieser M, Itil KZ (2000) A 26-week analysis of a double-blind, placebo-controlled trial of the ginkgo biloba extract EGb 761 in dementia. Dement Geriatr Cogn Disord 11(4):230–237
Levitan D, Greenwald I (1995) Facilitation of lin-12-mediated signalling by sel-12, a Caenorhabditis elegans S182 Alzheimer’s disease gene. Nature 377(6547):351–354
Levitan D, Greenwald I (1998) Effects of SEL-12 presenilin on LIN-12 localization and function in Caenorhabditis elegans. Development 125(18):3599–3606
Levitan D, Doyle TG, Brousseau D, Lee MK, Thinakaran G, Slunt HH et al (1996) Assessment of normal and mutant human presenilin function in Caenorhabditis elegans. Proc Natl Acad Sci USA 93:14940–14944
Levitan D, Yu G, St George Hyslop P, Goutte C (2001) APH-2/nicastrin functions in LIN-12/Notch signaling in the Caenorhabditis elegans somatic gonad. Dev Biol 240:654–661
Levy-Lahad E, Wasco W, Poorkaj P, Romano DM, Oshima J, Pettingell WH et al (1995a) Candidate gene for the chromosome 1 familial Alzheimer’s disease locus. Science 269(5226):973–977
Levy-Lahad E, Wijsman EM, Nemens E, Anderson L, Goddard KA, Weber JL et al (1995b) A familial Alzheimer’s disease locus on chromosome 1. Science 269(5226):970–973
Lewis J, Dickson DW, Lin WL, Chisholm L, Corral A, Jones G et al (2001) Enhanced neurofibrillary degeneration in transgenic mice expressing mutant tau and APP. Science 293(5534):1487–1491
Li X, Greenwald I (1996) Membrane topology of the C. elegans SEL-12 presenilin. Neuron 17:1015–1021
Li X, Greenwald I (1997) HOP-1, a Caenorhabditis elegans presenilin, appears to be functionally redundant with SEL-12 presenilin and to facilitate LIN-12 and GLP-1 signaling. Proc Natl Acad Sci USA 94(22):12204–12209
Li ZW, Stark G, Gotz J, Rulicke T, Gschwind M, Huber G et al (1996) Generation of mice with a 200-kb amyloid precursor protein gene deletion by Cre recombinase-mediated site-specific recombination in embryonic stem cells. Proc Natl Acad Sci USA 93(12):6158–6162
Li QX, Maynard C, Cappai R, McLean CA, Cherny RA, Lynch T et al (1999) Intracellular accumulation of detergent-soluble amyloidogenic A beta fragment of Alzheimer’s disease precursor protein in the hippocampus of aged transgenic mice. J Neurochem 72(6):2479–2487
Li YM, Lai MT, Xu M, Huang Q, DiMuzio-Mower J, Sardana MK et al (2000) Presenilin 1 is linked with gamma-secretase activity in the detergent solubilized state. Proc Natl Acad Sci USA 97(11):6138–6143. doi:10.1073/pnas.110126897
Li J, Pauley AM, Myers RL, Shuang R, Brashler JR, Yan R et al (2002) SEL-10 interacts with presenilin 1, facilitates its ubiquitination, and alters A-beta peptide production. J Neurochem 82(6):1540–1548
Lin K, Dorman JB, Rodan A, Kenyon C (1997) daf-16: An HNF-3/forkhead family member that can function to double the life-span of Caenorhabditis elegans. Science 278(5341):1319–1322
Lin K, Hsin H, Libina N, Kenyon C (2001) Regulation of the Caenorhabditis elegans longevity protein DAF-16 by insulin/IGF-1 and germline signaling. Nat Genet 28:139–145
Link CD (1995) Expression of human beta-amyloid peptide in transgenic Caenorhabditis elegans. Proc Natl Acad Sci USA 92:9368–9372
Link CD (2006) C. elegans models of age-associated neurodegenerative diseases: lessons from transgenic worm models of Alzheimer’s disease. Exp Gerontol 41:1007–1013
Link CD, Johnson CJ, Fonte V, Paupard M, Hall DH, Styren S et al (2001) Visualization of fibrillar amyloid deposits in living, transgenic Caenorhabditis elegans animals using the sensitive amyloid dye, X-34. Neurobiol Aging 22:217–226
Link CD, Taft A, Kapulkin V, Duke K, Kim S, Fei Q et al (2003) Gene expression analysis in a transgenic Caenorhabditis elegans Alzheimer’s disease model. Neurobiol Aging 24:397–413
Lorenzo A, Yankner BA (1996) Amyloid fibril toxicity in Alzheimer’s disease and diabetes. Ann N Y Acad Sci 777:89–95
Luo L, Tully T, White K (1992) Human amyloid precursor protein ameliorates behavioral deficit of flies deleted for Appl gene. Neuron 9(4):595–605
Luo Y, Smith JV, Paramasivam V, Burdick A, Curry KJ, Buford JP et al (2002) Inhibition of amyloid-beta aggregation and caspase-3 activation by the Ginkgo biloba extract EGb761. Proc Natl Acad Sci USA 99:12197–12202
Luse SA, Smith KR Jr (1964) The ultrastructure of senile plaques. Am J Pathol 44(4):553–563
MacMorris M, Kumar M, Lasda E, Larsen A, Kraemer B, Blumenthal T (2007) A novel family of C. elegans snRNPs contains proteins associated with trans-splicing. RNA 13(4):511–520
Magara F, Muller U, Li ZW, Lipp HP, Weissmann C, Stagljar M et al (1999) Genetic background changes the pattern of forebrain commissure defects in transgenic mice underexpressing the beta-amyloid-precursor protein. Proc Natl Acad Sci USA 96(8):4656–4661
Mahn K, Borras C, Knock GA, Taylor P, Khan IY, Sugden D et al (2005) Dietary soy isoflavone induced increases in antioxidant and eNOS gene expression lead to improved endothelial function and reduced blood pressure in vivo. Faseb J 19(12):1755–1757
Mann DM, Jones D, South PW, Snowden JS, Neary D (1992) Deposition of amyloid beta protein in non-Alzheimer dementias: evidence for a neuronal origin of parenchymal deposits of beta protein in neurodegenerative disease. Acta Neuropathol 83(4):415–419
Masters CL, Simms G, Weinman NA, Multhaup G, McDonald BL, Beyreuther K (1985) Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proc Natl Acad Sci USA 82(12):4245–4249
McColl G, Roberts BR, Gunn AP, Perez KA, Tew DJ, Masters CL et al (2009) The Caernorhabditis elegans Abeta1-42 model of Alzheimer’s disease predominantly expresses Abeta3-42. J Biol Chem 284(34):22697–22702
McDermott JB, Aamodt S, Aamodt E (1996) ptl-1, a Caenorhabditis elegans gene whose products are homologous to the tau microtubule-associated proteins. Biochemistry 35(29):9415–9423
Mello C, Fire A (1995) DNA transformation. Methods Cell Biol 48:451–482
Metzger RR, Brown JM, Sandoval V, Rau KS, Elwan MA, Miller GW et al (2002) Inhibitory effect of reserpine on dopamine transporter function. Eur J Pharmacol 456(1–3):39–43
Miklossy J, Taddei K, Suva D, Verdile G, Fonte J, Fisher C et al (2003) Two novel presenilin-1 mutations (Y256S and Q222H) are associated with early-onset Alzheimer’s disease. Neurobiol Aging 24(5):655–662
Mirra SS, Murrell JR, Gearing M, Spillantini MG, Goedert M, Crowther RA et al (1999) Tau pathology in a family with dementia and a P301L mutation in tau. J Neuropathol Exp Neurol 58(4):335–345
Mix JA, Crews WD Jr (2002) A double-blind, placebo-controlled, randomized trial of Ginkgo biloba extract EGb 761 in a sample of cognitively intact older adults: neuropsychological findings. Hum Psychopharmacol 17(6):267–277
Miyasaka T, Ding Z, Gengyo-Ando K, Oue M, Yamaguchi H, Mitani S et al (2005) Progressive neurodegeneration in C. elegans model of tauopathy. Neurobiol Dis 20(2):372–383. doi:S0969-9961(05)00079-3
Mucke L, Masliah E, Johnson WB, Ruppe MD, Alford M, Rockenstein EM et al (1994) Synaptotrophic effects of human amyloid beta protein precursors in the cortex of transgenic mice. Brain Res 666(2):151–167. doi:0006-8993(94)90767-6
Murphy CT, McCarroll SA, Bargmann CI, Fraser A, Kamath RS, Ahringer J et al (2003) Genes that act downstream of DAF-16 to influence the lifespan of Caenorhabditis elegans. Nature 424(6946):277–283
Murrell J, Farlow M, Ghetti B, Benson MD (1991) A mutation in the amyloid precursor protein associated with hereditary Alzheimer’s disease. Science 254(5028):97–99
Napolitano F, D’Angelo F, Bimonte M, Perrina V, D’Ambrosio C, Scaloni A et al (2008) A differential proteomic approach reveals an evolutionary conserved regulation of Nme proteins by Fe65 in C. elegans and mouse. Neurochem Res 33(12):2547–2555. doi:10.1007/s11064-008-9683-z
Nikolaev A, Mclaughlin T, O’Leary DD, Tessier-Lavigne M (2009) APP binds DR6 to trigger axon pruning and neuron death via distinct caspases. Nature 457:981–989
Niwa R, Zhou F, Li C, Slack FJ (2008) The expression of the Alzheimer’s amyloid precursor protein-like gene is regulated by developmental timing microRNAs and their targets in Caenorhabditis elegans. Dev Biol 315(2):418–425
Nixon RA, Wegiel J, Kumar A, Yu WH, Peterhoff C, Cataldo A et al (2005) Extensive involvement of autophagy in Alzheimer disease: an immuno-electron microscopy study. J Neuropathol Exp Neurol 64(2):113–122
Nunan J, Small DH (2000) Regulation of APP cleavage by alpha-, beta- and gamma-secretases. FEBS Lett 483(1):6–10. doi:S0014-5793(00)02076-7
Oddo S, Caccamo A, Kitazawa M, Tseng BP, LaFerla FM (2003a) Amyloid deposition precedes tangle formation in a triple transgenic model of Alzheimer’s disease. Neurobiol Aging 24(8):1063–1070
Oddo S, Caccamo A, Shepherd JD, Murphy MP, Golde TE, Kayed R et al (2003b) Triple-transgenic model of Alzheimer’s disease with plaques and tangles: intracellular Abeta and synaptic dysfunction. Neuron 39(3):409–421
Ogg S, Paradis S, Gottlieb S, Patterson GI, Lee L, Tissenbaum HA et al (1997) The Fork head transcription factor DAF-16 transduces insulin-like metabolic and longevity signals in C. elegans. Nature 389(6654):994–999
Oken BS, Storzbach DM, Kaye JA (1998) The efficacy of Ginkgo biloba on cognitive function in Alzheimer disease. Arch Neurol 55(11):1409–1415
Okochi M, Eimer S, Bottcher A, Baumeister R, Romig H, Walter J et al (2000) A loss of function mutant of the presenilin homologue SEL-12 undergoes aberrant endoproteolysis in Caenorhabditis elegans and increases abeta 42 generation in human cells. J Biol Chem 275(52):40925–40932. doi:10.1074/jbc.M005254200
Perez RG, Zheng H, Van der Ploeg LH, Koo EH (1997) The beta-amyloid precursor protein of Alzheimer’s disease enhances neuron viability and modulates neuronal polarity. J Neurosci 17(24):9407–9414
Pike CJ, Burdick D, Walencewicz AJ, Glabe CG, Cotman CW (1993) Neurodegeneration induced by beta-amyloid peptides in vitro: the role of peptide assembly state. J Neurosci 13(4):1676–1687
Ponte P, Gonzalez-DeWhitt P, Schilling J, Miller J, Hsu D, Greenberg B et al (1988) A new A4 amyloid mRNA contains a domain homologous to serine proteinase inhibitors. Nature 331(6156):525–527. doi:10.1038/331525a0
Reinhart BJ, Slack FJ, Basson M, Pasquinelli AE, Bettinger JC, Rougvie AE et al (2000) The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans. Nature 403(6772):901–906. doi:10.1038/35002607
Ring S, Weyer SW, Kilian SB, Waldron E, Pietrzik CU, Filippov MA et al (2007) The secreted beta-amyloid precursor protein ectodomain APPs alpha is sufficient to rescue the anatomical, behavioral, and electrophysiological abnormalities of APP-deficient mice. J Neurosci 27(29):7817–7826
Rogaev EI, Sherrington R, Rogaeva EA, Levesque G, Ikeda M, Liang Y et al (1995) Familial Alzheimer’s disease in kindreds with missense mutations in a gene on chromosome 1 related to the Alzheimer’s disease type 3 gene. Nature 376(6543):775–778
Rosen DR, Martin-Morris L, Luo LQ, White K (1989) A Drosophila gene encoding a protein resembling the human beta-amyloid protein precursor. Proc Natl Acad Sci USA 86(7):2478–2482
Roush SF, Slack FJ (2009) Transcription of the C. elegans let-7 microRNA is temporally regulated by one of its targets, hbl-1. Dev Biol. doi:S0012-1606(09)01062-8
Rovelet-Lecrux A, Hannequin D, Raux G, Le Meur N, Laquerriere A, Vital A et al (2006) APP locus duplication causes autosomal dominant early-onset Alzheimer disease with cerebral amyloid angiopathy. Nat Genet 38(1):24–26
Sabo SL, Ikin AF, Buxbaum JD, Greengard P (2001) The Alzheimer amyloid precursor protein (APP) and FE65, an APP-binding protein, regulate cell movement. J Cell Biol 153(7):1403–1414
Schmitt FA, Davis DG, Wekstein DR, Smith CD, Ashford JW, Markesbery WR (2000) “Preclinical” AD revisited: neuropathology of cognitively normal older adults. Neurology 55(3):370–376
Selkoe DJ (1999) Translating cell biology into therapeutic advances in Alzheimer’s disease. Nature 399(suppl 6738):A23–A31
Selkoe DJ (2001) Alzheimer’s disease: genes, proteins, and therapy. Physiol Rev 81:741–766
Sherrington R, Rogaev EI, Liang Y, Rogaeva EA, Levesque G, Ikeda M et al (1995) Cloning of a gene bearing missense mutations in early-onset familial Alzheimer’s disease. Nature 375(6534):754–760
Slack FJ, Basson M, Liu Z, Ambros V, Horvitz HR, Ruvkun G (2000) The lin-41 RBCC gene acts in the C. elegans heterochronic pathway between the let-7 regulatory RNA and the LIN-29 transcription factor. Mol Cell 5(4):659–669. doi:S1097-2765(00)80245-2
Sleegers K, Brouwers N, Gijselinck I, Theuns J, Goossens D, Wauters J et al (2006) APP duplication is sufficient to cause early onset Alzheimer’s dementia with cerebral amyloid angiopathy. Brain 129(Pt 11):2977–2983
Slunt HH, Thinakaran G, Von Koch C, Lo AC, Tanzi RE, Sisodia SS (1994) Expression of a ubiquitous, cross-reactive homologue of the mouse beta-amyloid precursor protein (APP). J Biol Chem 269(4):2637–2644
Smith JV, Luo Y (2003) Elevation of oxidative free radicals in Alzheimer’s disease models can be attenuated by Ginkgo biloba extract EGb 761. J Alzheimers Dis 5(4):287–300
Smith JV, Burdick AJ, Golik P, Khan I, Wallace D, Luo Y (2002) Anti-apoptotic properties of Ginkgo biloba extract EGb 761 in differentiated PC12 cells. Cell Mol Biol (Noisy-le-grand) 48(6):699–707
Spillantini MG, Bird TD, Ghetti B (1998) Frontotemporal dementia and Parkinsonism linked to chromosome 17: a new group of tauopathies. Brain Pathol 8(2):387–402
Sprecher CA, Grant FJ, Grimm G, O’Hara PJ, Norris F, Norris K et al (1993) Molecular cloning of the cDNA for a human amyloid precursor protein homolog: evidence for a multigene family. Biochemistry 32(17):4481–4486
Srivastava D, Arya U, SoundaraRajan T, Dwivedi H, Kumar S, Subramaniam JR (2008) Reserpine can confer stress tolerance and lifespan extension in the nematode C. elegans. Biogerontology 9(5):309–316
Steen E, Terry BM, Rivera EJ, Cannon JL, Neely TR, Tavares R et al (2005) Impaired insulin and insulin-like growth factor expression and signaling mechanisms in Alzheimer’s disease—is this type 3 diabetes? J Alzheimers Dis 7(1):63–80
Steinbach JP, Muller U, Leist M, Li ZW, Nicotera P, Aguzzi A (1998) Hypersensitivity to seizures in beta-amyloid precursor protein deficient mice. Cell Death Differ 5(10):858–866
Struhl G, Greenwald I (1999) Presenilin is required for activity and nuclear access of Notch in Drosophila. Nature 398(6727):522–525. doi:10.1038/19091
Sulston JE, White JG (1980) Regulation and cell autonomy during postembryonic development of Caenorhabditis elegans. Dev Biol 78:577–597
Tanzi RE, McClatchey AI, Lamperti ED, Villa-Komaroff L, Gusella JF, Neve RL (1988) Protease inhibitor domain encoded by an amyloid protein precursor mRNA associated with Alzheimer’s disease. Nature 331(6156):528–530. doi:10.1038/331528a0
Tax FE, Thomas JH, Ferguson EL, Horvitz HR (1997) Identification and characterization of genes that interact with lin-12 in Caenorhabditis elegans. Genetics 147(4):1675–1695
Terry RD, Gonatas NK, Weiss M (1964) Ultrastructural studies in Alzheimer’s presenile dementia. Am J Pathol 44:269–297
Thinakaran G, Borchelt DR, Lee MK, Slunt HH, Spitzer L, Kim G et al (1996) Endoproteolysis of presenilin 1 and accumulation of processed derivatives in vivo. Neuron 17(1):181–190. doi:S0896-6273(00)80291-3
Tremml P, Lipp HP, Muller U, Ricceri L, Wolfer DP (1998) Neurobehavioral development, adult openfield exploration and swimming navigation learning in mice with a modified beta-amyloid precursor protein gene. Behav Brain Res 95(1):65–76
Vakil RJ (1949) A clinical trial of Rauwolfia serpentina in essential hypertension. Br Heart J 11(4):350–355
von Koch CS, Zheng H, Chen H, Trumbauer M, Thinakaran G, van der Ploeg LH et al (1997) Generation of APLP2 KO mice and early postnatal lethality in APLP2/APP double KO mice. Neurobiol Aging 18(6):661–669
Walker GA, White TM, McColl G, Jenkins NL, Babich S, Candido EP et al (2001) Heat shock protein accumulation is upregulated in a long-lived mutant of Caenorhabditis elegans. J Gerontol A Biol Sci Med Sci 56:B281–B287
Wang Y, Ha Y (2004) The X-ray structure of an antiparallel dimer of the human amyloid precursor protein E2 domain. Mol Cell 15:343–353
Wasco W, Bupp K, Magendantz M, Gusella JF, Tanzi RE, Solomon F (1992) Identification of a mouse brain cDNA that encodes a protein related to the Alzheimer disease-associated amyloid beta protein precursor. Proc Natl Acad Sci USA 89:10758–10762
Wasco W, Gurubhagavatula S, Paradis MD, Romano DM, Sisodia SS, Hyman BT et al (1993a) Isolation and characterization of APLP2 encoding a homologue of the Alzheimer’s associated amyloid beta protein precursor. Nat Genet 5(1):95–100. doi:10.1038/ng0993-95
Wasco W, Peppercorn J, Tanzi RE (1993b) Search for the genes responsible for familial Alzheimer’s disease. Ann N Y Acad Sci 695:203–208
Wen C, Metzstein MM, Greenwald I (1997) SUP-17, a Caenorhabditis elegans ADAM protein related to Drosophila KUZBANIAN, and its role in LIN-12/NOTCH signalling. Development 124(23):4759–4767
Wen C, Levitan D, Li X, Greenwald I (2000) spr-2, a suppressor of the egg-laying defect caused by loss of sel-12 presenilin in Caenorhabditis elegans, is a member of the SET protein subfamily. Proc Natl Acad Sci USA 97:14524–14529
Westlund B, Parry D, Clover R, Basson M, Johnson CD (1999) Reverse genetic analysis of Caenorhabditis elegans presenilins reveals redundant but unequal roles for sel-12 and hop-1 in Notch-pathway signaling. Proc Natl Acad Sci USA 96(5):2497–2502
Winter E (1991) Effects of an extract of Ginkgo biloba on learning and memory in mice. Pharmacol Biochem Behav 38(1):109–114
Winter JC (1998) The effects of an extract of Ginkgo biloba, EGb 761, on cognitive behavior and longevity in the rat. Physiol Behav 63(3):425–433
Wirths O, Multhaup G, Czech C, Blanchard V, Moussaoui S, Tremp G et al (2001) Intraneuronal Abeta accumulation precedes plaque formation in beta-amyloid precursor protein and presenilin-1 double-transgenic mice. Neurosci Lett 306(1–2):116–120. doi:S0304-3940(01)01876-6
Withee J, Galligan B, Hawkins N, Garriga G (2004) Caenorhabditis elegans WASP and Ena/VASP proteins play compensatory roles in morphogenesis and neuronal cell migration. Genetics 167(3):1165–1176. doi:10.1534/genetics.103.025676
Wittenburg N, Eimer S, Lakowski B, Röhrig S, Rudolph C, Baumeister R (2000) Presenilin is required for proper morphology and function of neurons in C. elegans. Nature 406:306–309
Wolfe MS, Xia W, Ostaszewski BL, Diehl TS, Kimberly WT, Selkoe DJ (1999) Two transmembrane aspartates in presenilin-1 required for presenilin endoproteolysis and gamma-secretase activity. Nature 398(6727):513–517. doi:10.1038/19077
Wu Y, Luo Y (2005) Transgenic C. elegans as a model in Alzheimer’s research. Curr Alzheimer Res 2(1):37–45
Wu G, Hubbard EJ, Kitajewski JK, Greenwald I (1998) Evidence for functional and physical association between Caenorhabditis elegans SEL-10, a Cdc4p-related protein, and SEL-12 presenilin. Proc Natl Acad Sci USA 95:15787–15791
Wu Y, Wu Z, Butko P, Christen Y, Lambert MP, Klein WL et al (2006) Amyloid-beta-induced pathological behaviors are suppressed by Ginkgo biloba extract EGb 761 and ginkgolides in transgenic Caenorhabditis elegans. J Neurosci 26:13102–13113
Xu K, Tavernarakis N, Driscoll M (2001) Necrotic cell death in C. elegans requires the function of calreticulin and regulators of Ca(2+) release from the endoplasmic reticulum. Neuron 31(6):957–971. doi:S0896-6273(01)00432-9
Yankner BA, Duffy LK, Kirschner DA (1990) Neurotrophic and neurotoxic effects of amyloid beta protein: reversal by tachykinin neuropeptides. Science 250(4978):279–282
Yatin SM, Varadarajan S, Link CD, Butterfield DA (1999) In vitro and in vivo oxidative stress associated with Alzheimer’s amyloid beta-peptide (1–42). Neurobiol Aging 20:325–330 (discussion 339–342)
Ye Y, Lukinova N, Fortini ME (1999) Neurogenic phenotypes and altered Notch processing in Drosophila Presenilin mutants. Nature 398(6727):525–529. doi:10.1038/19096
Yoshikai S, Sasaki H, Doh-ura K, Furuya H, Sakaki Y (1990) Genomic organization of the human amyloid beta-protein precursor gene. Gene 87(2):257–263
Yu G, Nishimura M, Arawaka S, Levitan D, Zhang L, Tandon A et al (2000) Nicastrin modulates presenilin-mediated notch/glp-1 signal transduction and betaAPP processing. Nature 407:48–54
Yu WH, Kumar A, Peterhoff C, Shapiro Kulnane L, Uchiyama Y, Lamb BT et al (2004) Autophagic vacuoles are enriched in amyloid precursor protein-secretase activities: implications for beta-amyloid peptide over-production and localization in Alzheimer’s disease. Int J Biochem Cell Biol 36(12):2531–2540. doi:10.1016/j.biocel.2004.05.010
Yu WH, Cuervo AM, Kumar A, Peterhoff CM, Schmidt SD, Lee JH et al (2005) Macroautophagy-a novel beta-amyloid peptide-generating pathway activated in Alzheimer’s disease. J Cell Biol 171(1):87–98. doi:jcb.200505082
Zambrano N, Bimonte M, Arbucci S, Gianni D, Russo T, Bazzicalupo P (2002) feh-1 and apl-1, the Caenorhabditis elegans orthologues of mammalian Fe65 and beta-amyloid precursor protein genes, are involved in the same pathway that controls nematode pharyngeal pumping. J Cell Sci 115(Pt 7):1411–1422
Zheng H, Koo E (2006) The amyloid precursor protein: beyond amyloid. Mol Neurodegener 1:5
Zheng H, Jiang M, Trumbauer ME, Sirinathsinghji DJ, Hopkins R, Smith DW et al (1995) beta-Amyloid precursor protein-deficient mice show reactive gliosis and decreased locomotor activity. Cell 81(4):525–531
Zheng WH, Bastianetto S, Mennicken F, Ma W, Kar S (2002) Amyloid beta peptide induces tau phosphorylation and loss of cholinergic neurons in rat primary septal cultures. Neuroscience 115(1):201–211. doi:S0306452202004049
Acknowledgments
We wish to thank Casey Brander for help with the figures and lab members for helpful discussions. This work was supported by grants from the Alzheimer’s Association, National Institutes Health, and National Science Foundation (CL) and a National Institutes of Health RCMI grant to City College.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ewald, C.Y., Li, C. Understanding the molecular basis of Alzheimer’s disease using a Caenorhabditis elegans model system. Brain Struct Funct 214, 263–283 (2010). https://doi.org/10.1007/s00429-009-0235-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-009-0235-3