Abstract
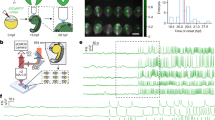
The developing mammalian heart initiates spontaneous contractions shortly after the myocardium differentiates from the splanchnic mesoderm. The precise timing and mode of the onset of heartbeat, however, have not been statistically described in mice. We analyzed the patterns of contractive activity in video-recorded heart regions ranging from the pre-somite stage to day 10.5 (E10.5). The first sign was detected at the 3-somite stage (E8.25), when a few cardiac myocytes constituted small contracting groups on both sides of the heart tube. Fluctuations of the basal [Ca2+]i level were detected in dormant 3-somite-stage hearts, indicating the initiation of electrical activity before visible contractions. After weak and irregular contractions at the 3-somite stage, the contractions were almost coordinated as early as the 4-somite stage. Unidirectional blood flow through the atrioventricular canal was established around the 20-somite stage at E9.25, correlated with the development of the endocardial cushion. We propose that not only the endocardial cushion but also coordinated contractions are essential for unidirectional flow, because induced bradycardia due to short exposure at room temperature caused regurgitation at E10.5 when otherwise highly organized flow was observed. These findings complement and extend earlier observations on functional heart development, providing a reference for fundamental research on mammalian cardiogenesis.



Similar content being viewed by others
References
Egashira K, Nishii K, Nakamura K, Kumai M, Morimoto S, Shibata Y (2004) Conduction abnormality in gap junction protein connexin45-deficient embryonic stem cell-derived cardiac myocytes. Anat Rec 280A:973–979
Franco D, Lamers WH, Moorman AFM (1998) Patterns of expression in the developing myocardium: towards a morphologically integrated transcriptional model. Cardiovasc Res 38:25–53
Goss CM (1938) The first contractions of the heart in rat embryos. Anat Rec 70:505–524
Hirota A, Kamino K, Komuro H, Sakai T, Yada T (1985) Early events in development of electrical activity and contraction in embryonic rat heart assessed by optical recording. J Physiol 369:209–227
Hogan B, Beddington R, Costantini F, Lacy E (1994) Manipulating the mouse embryo: a laboratory manual. Cold Spring Harbor, Cold Spring Harbor, New York
Huang GY, Linask KK (1998) Doppler echocardiographic analysis of effects of tribromoethanol anesthesia on cardiac function in the mouse embryo: a comparison with pentobarbital. Lab Anim Sci 48:206–209
Ji RP, Phoon CKL, Aristizábal O, McGrath KE, Palis J, Turnbull DH (2003) Onset of cardiac function during early mouse embryogenesis coincides with entry of primitive erythroblasts into the embryo proper. Circ Res 92:133–135
Kamino K (1991) Optical approaches to ontogeny of electrical activity and related functional organization during early heart development. Physiol Rev 71:53–91
Kaufman MH, Navaratnam V (1981) Early differentiation of the heart in mouse embryos. J Anat 133:235–246
Kumai M, Nishii K, Nakamura K, Takeda N, Suzuki M, Shibata Y (2000) Loss of connexin45 causes a cushion defect in early cardiogenesis. Development 127:3501–3512
Linask KK, Han MD, Artman M, Ludwig CA (2001) Sodium–calcium exchanger (NCX-1) and calcium modulation: NCX protein expression patterns and regulation of early heart development. Dev Dyn 221:249–264
McGrath KE, Koniski AD, Malik J, Palis J (2003) Circulation is established in a stepwise pattern in the mammalian embryo. Blood 101:1669–1676
Navaratnam V, Kaufman MH, Skepper JN, Barton S, Guttridge KM (1986) Differentiation of the myocardial rudiment of mouse embryos: an ultrastructural study including freeze-fracture replication. J Anat 146:65–85
Nishii K, Kumai M, Shibata Y (2001) Regulation of the epithelial–mesenchymal transformation through gap junction channels in heart development. Trends Cardiovasc Med 11:213–218
Nishii K, Kumai M, Egashira K, Miwa T, Hashizume K, Miyano Y, Shibata Y (2003) Mice lacking connexin45 conditionally in cardiac myocytes display embryonic lethality similar to that of germline knockout mice without endocardial cushion defect. Cell Commun Adhesion 10:365–369
Phoon CKL, Ji RP, Aristizábal O, Worrad DM, Zhou B, Baldwin HS, Turnbull DH (2004) Embryonic heart failure in NFATc1 -/- mice: novel mechanistic insights from in utero ultrasound biomicroscopy. Circ Res 95:92–99
Rentschler S, Vaidya DM, Tamaddon H, Degenhardt K, Sassoon D, Morley GE, Jalife J, Fishman GI (2001) Visualization and functional characterization of the developing murine cardiac conduction system. Development 128:1785–1792
Rentschler S, Zander J, Meyers K, France D, Levine R, Porter G, Rivkees SA, Morley GE, Fishman GI (2002) Neuregulin-1 promotes formation of the murine cardiac conduction system. Proc Natl Acad Sci USA 99:10464–10469
Rothenberg F, Efimov IR, Watanabe M (2004) Functional imaging of the embryonic pacemaking and cardiac conduction system over the past 150 years: technologies to overcome the challenges. Anat Rec 280A:980–989
Virágh S, Challice CE (1977) The development of the conduction system in the mouse embryo heart: I. The first embryonic A–V conduction pathway. Dev Biol 56:382–396
Acknowledgements
We thank Kanako Hashizume and Yumi Miyano for their technical assistance, Mamiko Kamizono and Takeyo Omura for their care of the experimental animals, and Dr. Sachio Morimoto (of the Department of Clinical Pharmacology) for allowing us to use the [Ca2+]i-recording equipment. This work was supported by Grants-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (No. 16790126 to K.N. and No. 17390052 to Y.S.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Nishii, K., Shibata, Y. Mode and determination of the initial contraction stage in the mouse embryo heart. Anat Embryol 211, 95–100 (2006). https://doi.org/10.1007/s00429-005-0065-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-005-0065-x